The following is the established format for referencing this article:
Schaffner, F. C., M. T. Hallworth, M. F. Jimenez, I. Rodríguez-Colón, and N. E. Seavy. 2023. Light-level tracking of Northern Waterthrush (Parkesia noveboracensis): lessons learned from wintering ground deployments. Journal of Field Ornithology 94(4):5.ABSTRACT
The limited understanding of spatial and temporal patterns across the full annual cycle of long-distance migrants remains a crucial gap in the ability to describe their life history and inform conservation efforts. The Northern Waterthrush (Parkesia noveboracensis), for example, winters in the Caribbean and northern South America and breeds across eastern North America. Little information exists pertaining to their migratory routes, behavior, or migratory connectivity. Here, we describe migratory patterns derived from light-level geolocators deployed on Northern Waterthrushes wintering in Puerto Rico. We found that birds stopped in the northern Greater Antilles before migrating along the Atlantic coast to their breeding sites as far north as Quebec, Canada. Variability in transition events (sunrise/sunset) due to shading of the light sensor led to large uncertainty in breeding location estimates. However, we were able to use the raw light-level data to infer the phenology of migration. Northern Waterthrushes departed Puerto Rico on 24 April (± 8.1 d) and arrived on the breeding grounds on 17 May (± 10.4 d), suggesting that spring migration lasts approximately 23 (± 3.6) d. We also used raw light-level data during the breeding season to infer nesting behavior, which allowed us to identify individuals as breeding females and describe their nesting phenology. Together, these results offer new information about the migratory routes for Northern Waterthrushes and demonstrate how light-level geolocators deployed on the wintering grounds can be used to sex monochromatic species and infer life history information during the breeding season.
RESUMEN
La comprensión limitada de los patrones espaciales y temporales para migrantes de larga distancia a través del ciclo anual completo se mantiene como un vacío crucial en la habilidad de describir su historia natural e informar los esfuerzos de conservación. Parkesia noveboracensis, por ejemplo, pasa el invierno en el Caribe y el norte de Sur América y se reproduce a lo largo del este de Norte América. Existe poca información sobre sus rutas migratorias, comportamiento o conectividad migratoria. Aquí, describimos los patrones derivados de geolocalizadores de nivel de luz instalados en P. noveboracensis pasando el invierno en Puerto Rico. Encontramos que las aves paran en el norte de las Antillas Mayores antes de migrar a lo largo de la costa Atlántica a sus sitios de reproducción, tan al norte como Quebec, Canadá. La variabilidad en la transición de eventos (amanecer/atardecer) debido a la sombra sobre el sensor de luz derivó en una incertidumbre amplia en la estimación de la ubicación de las zonas de reproducción. Sin embargo, pudimos utilizar los datos crudos del nivel de luz para inferir la fenología de la migración. Los individuos de P. noveboracensis partieron de Puerto Rico el 24 de abril (± 8.1 d) y llegaron a la zona de reproducción el 17 de mayo (± 10.4 d), sugiriendo que la migración de primavera tiene una duración aproximada de 23 (± 3.6) d. Utilizamos también los datos crudos de los niveles de luz durante la temporada de reproducción para inferir sus comportamientos de anidación, lo que nos permitió identificar los individuos como hembras reproductivas y describir su fenología de anidación. Conjuntamente, estos resultados proporcionan nueva información sobre las rutas migratorias de Parkesia noveboracensis y demuestra, como los geolocalizadores de nivel de luz instalado en zonas de invierno pueden ser usados en especies sexualmente monocromáticas e inferir información de historia natural durante la temporada reproductiva.
INTRODUCTION
The Caribbean islands, and particularly the Greater Antilles, are important overwintering destinations for many neotropical migrants, and research on these migrants has greatly advanced the understanding of overwintering ecology (Albert et al. 2020). One important finding has been the recognition that conditions on the wintering grounds can have carry-over effects on the breeding grounds (Harrison et al. 2011, Rockwell et al. 2012). These islands also provide important stopover sites during the long-distance flights of many migratory birds (Bayly et al. 2018). These findings underline the importance of obtaining more information on migratory movements between the breeding grounds and overwintering areas.
The Northern Waterthrush (Parkesia noveboracensis) is a long-distance migrant that nests in North America as far north as the Yukon Territory and migrates to spend the non-breeding season in Central America, northern South America, and throughout the Caribbean islands (Whitaker and Eaton 2020). Stable isotope data from wintering birds in Puerto Rico suggest that these birds likely breed in eastern North America (Langin et al. 2009, Schaffner et al. 2017a,b). However, no tracking studies have demonstrated more specific information about the breeding destinations and behavior of the birds that winter in or transit through Puerto Rico.
Northern Waterthrushes use a variety of vegetation types in the Caribbean, including mangroves and dry forests (Smith et al. 2010). Across these habitat types, more dominant individuals, usually males, are more likely to defend high-quality territories in red mangroves (and remain on those territories day and night), can maintain better body condition, and begin pre-migratory fat deposition earlier in the spring than do less dominant individuals, often females and subadults of both sexes (Smith et al. 2010). These results suggest that wintering habitat quality likely has carry-over effects on migration phenology and reproductive success. However, one challenge to understanding these patterns is that males and females have similar plumage characteristics, and wing measurements can only reliably be used to sex the largest and smallest individuals (Covino 2015). Thus, tracking Northern Waterthrushes, especially both sexes, across the annual cycle could both provide insights into migratory connectivity and identify potential carry-over effects of winter habitat quality.
One tool for tracking small migratory birds is light-level geolocation (the use of daylight measurements to track animal movements; McKinnon and Love 2018). The location data derived from light-level data can provide information about migratory phenology, migratory routes (when migration does not coincide with the spring or fall equinoxes), and breeding or non-breeding destinations. However, using light-level data to estimate locations or other behavioral information may be complicated when birds move into and out of shaded environments (Lisovski et al. 2012). In addition to location information, light-level data have been used to infer behavioral information such as documenting Northern Flicker cavity use throughout the year (Gow et al. 2015) and quantifying the reproductive behavior of shorebirds (Burger et al. 2012, Loktionov et al. 2015, Bulla et al. 2016, Smith et al. 2020). The ability to use light-level data to understand nesting phenology and reproductive success is potentially relevant for studying carry-over effects, yet there are relatively few examples of the use of light-level data to collect information beyond location data for passerine birds.
Thus, to evaluate the utility of light-level data for describing the migratory ecology of the Northern Waterthrush, we deployed geolocators on wintering Northern Waterthrushes in Puerto Rico. In addition to estimating geographic locations, we explored the potential to infer breeding behavior and nesting phenology. Our results provide an example of how light-level geolocators can be used to obtain information beyond just location, especially when female birds are tagged on wintering grounds.
METHODS
Study site
We captured Northern Waterthrushes and deployed geolocators at Jobos Bay National Estuarine Research Reserve (hereafter Jobos Bay) in the municipality of Salinas, southeastern Puerto Rico (17°57′27″ N, 66°13′20″ W). Jobos Bay contains mangrove forests, hypersaline lagoons, salt flats, coral reefs, dry forests, and seagrass bed ecosystems, most of them protected (Rodríguez-Colón 2012, Williams et al. 2013). This entire area is classified as a subtropical dry forest within the Holdridge Life Zones (Ewel and Whitmore 1973).
Within Jobos Bay, we captured Northern Waterthrushes at two sites that were approximately 2 km apart (Schaffner et al. 2017a). The first site was in secondary mixed dry forest that included a variety of both native and exotic trees (Rodríguez-Colón 2012). The second site was an area dominated by mesquite (Prosopis pallida) and previously used as cattle pasture (Rodríguez-Colón 2012). At both sites, Northern Waterthrushes and other migrants, as well as non-nesting residents, night-roost in red mangroves (Rhizophora mangle); just before dawn, they begin moving through black mangrove (Avicennia germinans) and into the adjacent mixed dry forest and mesquite woodland to forage. They reverse this movement in late afternoon and dusk to return to their red mangrove roosts, similar to behaviors observed elsewhere in Puerto Rico (Smith et al. 2008). For statistical analyses, we pooled the data from birds captured at the two sites.
The rainy season occurs from September through December, followed by a marked dry season from January through April (Rodríguez-Colón 2012). Migrant passerines use Jobos Bay during both the wetter and dryer periods, permitting us to refer to these migratory periods as rainy season and dry season. Our previous banding efforts in the area led us to hypothesize that there would be more transients (using our areas as stopover sites) during the rainy season, whereas birds captured during the dry season were more likely to be using the area as their primary wintering ground.
Field methods and geolocator deployment
We captured Northern Waterthrushes in mist-nets that were set up along existing roads and trails. From September 2015 through April 2016, a line of eight 12 m long 30-mm or 38-mm mesh mist nets was established at each site approximately twice per month, alternating between the two sites. The following year (September 2016 through April 2017) a similar protocol was employed using ten nets instead of eight and playing recorded calls of Northern Waterthrushes during the netting sessions. Nets were operated in two-day sessions: we set up nets in the afternoon on the first day and operated until shortly after sunset. On the following dawn, we returned and operated the nets until mid-morning (Rodríguez-Colón 2012, Schaffner et al. 2017a,b). We measured the wing chords of the birds that we captured along with other morphometrics such as bill and tarsus lengths and bird mass. We banded all Northern Waterthrushes (whether tagged with a geolocator or not) with a U.S. Geological Survey (USGS) aluminum band and one to three Darvic color bands.
We deployed 19 geolocators (Intigeo-P50B1-7-DIP, Migrate Technology, Cambridge, UK) during the rainy season (September to December 2015). Because of the weight of these geolocators (0.55 g), we did not deploy geolocators on any waterthrushes that weighed < 18 g during this period. In the dry season (February to April 2016), we deployed an additional 21 geolocators (Intigeo-P30Z-7-DIP, Migrate Technology). Because these geolocators were lighter (0.40 g), we were able to deploy geolocators on all waterthrushes we captured. Both models were mounted on the birds using a modification of the leg-loop harness method using either braided cord or plastic microtubing (Rappole and Tipton 1991, Streby et al. 2015).
Banding encounter data
We assessed banding and encounter data to determine if any recoveries of Northern Waterthrushes originally banded during the non-breeding season in Puerto Rico were subsequently recovered during the breeding season in the United States and Canada (1964–2019; data provided by the USGS Bird Banding Lab). We considered encounters between 20 June and 20 July to represent breeding locations.
Statistical analysis
Recapture rates
To test for differences in recapture rates between individuals with and without geolocators, we used a Pearson chi-squared test and compared the number of individuals that were recaptured to those not recaptured. Because we expected return rates to be lower for individuals tagged in the rainy season, we ran separate analyses for tagged birds in each season. We also pooled all birds, regardless of whether they were tagged with geolocators, and compared recapture rates between seasons. We conducted these tests in R (R Core Team 2021) and used an alpha of 0.05 for all tests.
Light-level data
Our initial inspection of the raw light-level records revealed large amounts of shading during some parts of the year, especially during light-dark transition periods at sunrise and sunset. Such shading leads to large uncertainty in location estimates, complicating location inferences drawn from light-level data, especially when the degree of shading changes throughout the year (Lisovski et al. 2012). Thus, we approached our analysis of the light-level data by first estimating locations (methods described below) to the best of our ability, and then extracting as much information about migration and phenology as possible.
Location estimates and migration phenology
We estimated locations from light-level data using the threshold method (Lisovski et al. 2020). The geolocators we used recorded light levels at 5-min intervals on a scale of 1–74,000 lux; we log-transformed the light values prior to analysis. On the log scale, we used a light threshold of 0.5 to identify sunrise and sunset (hereafter twilights). We identified twilights with the “twGeos” package (Lisovski et al. 2016) and automatically deleted or adjusted outlying twilights with the twilightAdjust function (with 30 min as a cut-off for outliers and 15 min as accepted variability for successive twilight events).
To convert sunrise and sunset events into locations, we used the “GeoLight” package (Lisovski and Hahn 2012) in R (R Core Team 2021). Although more sophisticated methods are available for estimating locations (Rakhimberdiev et al. 2017, Lisovski et al. 2020), our attempts to use those methods either failed because of excessive shading in the data or because they smoothed data with movement models, producing results that masked uncertainty in the raw estimates. Thus, we opted to use the simplest method possible to clearly illustrate the limitations of the location estimates.
For each bird, we determined the sun elevation value using a post-deployment calibration period from 5 d after the tag was deployed to 5 d before the bird departed on migration (mean calibration = 57 d, range 23 to 139 d). The sun elevation angles for these birds ranged from −4.7° to −3.0°. We used these sun elevation angles to calculate the latitude and longitude for each day. For the two tags that were deployed well before the spring equinox, we summarized the estimated locations at the tagging location using mean and standard deviation of latitude and longitude. We assumed that birds were on their nesting grounds between 20 June and 20 July and summarized the locations during this period using the mean and standard deviation of latitude and longitude and 95% kernel density estimates.
We estimated the departure from non-breeding sites and arrival at breeding sites by manually inspecting the variability in light levels across each individual’s data set. We plotted the twilight events at the deployment location and, to determine the departure from wintering sites, we identified the period when observed twilight events began to deviate from those at the deployment location (see Fig. 1 for example). During long over-water flights, light levels show notable periods of consistently high light-intensity values (DeLuca et al. 2015). Because Northern Waterthrush departing from Puerto Rico must make a long over-water flight, we also looked for prolonged light periods as indicators of departure dates. To determine the arrival at breeding sites, we noted when the twilight events stabilized after a period of rapid change in twilight events.
Sex determination and nesting phenology
For birds that use shaded nest sites, the nesting period can be identified by regular intermittent dark periods during daylight hours (Burger et al. 2012; Fig. 1). We visually inspected all light-level data for evidence of nesting. The nesting phenology information we derived spanned from roughly the onset of incubation until either the nest failed or the adult bird began spending enough time away from the nest that the clear day-time dark signal was no longer apparent, probably several days after the young hatched. Because it is generally reported that only female Northern Waterthrush incubate (Eaton 1957), we assumed that any light-level record with evidence of nesting was female. For the other tagged birds, the lack of a nesting light signature does not necessarily preclude the possibility of an individual being a non-breeding female or a female with a nesting attempt that failed before the clutch was complete. As such, we refer to these birds as “putative males.”
All data and R code are available on MoveBank at https://www.movebank.org/cms/webapp?gwt_fragment=page=studies,path=study1259878781 (study name: “Northern Waterthrush-Schaffner-Puerto Rico”).
RESULTS
Of the 40 geolocators deployed between September 2015 and April 2016, we recovered 11 the following year (Table 1). None of the recovered individuals showed any sign of injury from carrying the geolocator. The recapture rates for control birds (carrying bands only) were lower for birds initially captured during the rainy season (September–December; 25%) compared to recapture rates of individuals initially captured in the dry season (January–April; 43%, χ² = 4.32, df = 1, P = 0.038; Table 1). Because of this difference, we compared the recapture rates of tagged and untagged birds within each season. In the rainy season, the recapture rates of birds with bands and geolocators (11%) was lower than that of birds with only bands (25%), but the difference was not statistically significant (χ² = 1.07, df = 1, P = 0.301). For birds captured during the dry season, there was little difference between the recapture rates of birds with bands and geolocators (43%) and control birds (43%; χ² = 9.7519 × 10−31, df = 1, P = 1; Table 1). All of the recaptured geolocator-tagged birds were recovered in the same habitat from which they had been deployed, whereas three of the control birds moved between deployment and recovery. Of those that were recaptured in different habitat types, all moved from mesquite woodland to mixed dry forest.
Breeding locations, migration routes, and migration phenology
Of the 11 recovered tags, 9 provided useable data (Fig. 2). Of the nine birds with useable data, four individuals had breeding locations located in northern Quebec, Canada (Fig. 3). The other five individuals (two females and three putative males) had breeding location estimates to the east of the Atlantic seaboard over the Atlantic Ocean (Fig. 3). That these location estimates fell over the ocean indicated that the calibrated sun elevation angle from the non-breeding season was inconsistent from the sun elevation angle during the breeding season (McKinnon et al. 2013, Hallworth et al. 2015).
In our review of the USGS Bird Banding Lab data, we found that of 2531 Northern Waterthrushes banded in Puerto Rico between 1964 and 2019, only 4 were encountered away from the island. Three of the re-encounters occurred between 3 April and 11 May (presumably during migration) in the U.S. states of Maryland, New York, and Virginia. A fourth re-encounter occurred on 13 July, presumably when the bird was on the breeding grounds, in Massachusetts (latitude 42.75, longitude −71.75; Fig. 3).
Location estimates during migration indicated that birds initiated migration by flying west, evidenced by both the rapid shift in locations (Fig. 4) and the extended light periods suggesting migratory flights extended into the following day (Fig. 2). After stopping in the northern Greater Antilles (location estimates were not accurate enough to distinguish between stopovers in Cuba or the Bahamas), the birds moved more slowly northward along the Atlantic seaboard during migration (Fig. 4). The mean spring departure date was April 24 (± 8.1 d) and the mean breeding site arrival date was May 17 (± 10.4 d).
Sex determination and nesting phenology
Of the nine recovered geolocators, we determined that six (66%) were putative males and three (33%) were females based on light-level signatures consistent with incubation behavior (Fig. 2; Table 2). For the three females, the mean date of nesting behavior onset was June 1 (± 4.1 d) and the mean end date of nesting was June 15 (± 1.9 d). One bird exhibited nesting behavior for 5 d, suggesting that the nest was depredated. The other two exhibited nesting behavior for 19 and 21 d, suggesting that the nest survived long enough to hatch (Fig. 4). There was no evidence of renesting or second brooding.
DISCUSSION
Understanding spatial and temporal patterns of habitat use across the full annual cycle of migratory birds is crucial information in efforts to protect them (Webster et al. 2002). The breeding location estimates we generated add to the body of information about migratory connectivity of the Northern Waterthrush. Although the behavior and movement of non-breeding Northern Waterthrushes has been given considerable attention (Reitsma et al. 2002, Burson et al. 2005, Smith et al. 2008), fewer studies have sought to describe their full annual cycle and migration phenology.
Our results suggest that Northern Waterthrushes wintering in Puerto Rico migrate north along the eastern coast of North America and breed from the northeastern United States up into northern Quebec. Although, to our knowledge, our study is the first to use tracking devices to describe the migratory pathways of Northern Waterthrushes, our results are consistent with breeding latitudes that have been suggested by stable isotope analyses on this species (Langin et al. 2009, Schaffner et al. 2017b) as well as banding re-encounters compiled by the USGS Bird Banding Lab, which we reference in our results.
The estimates of breeding locations over the Atlantic Ocean illustrate the challenges of light-level geolocation when sun elevation angles derived from calibrations at the tagging locations are inconsistent with sun elevation angles recorded during other periods of the year (Hallworth et al. 2015). These inconsistencies can be attributed to differences in the light environment that can be created by bird behavior, vegetation structure, topography, weather, and other factors that vary through space or time (Porter and Smith 2013). Most commonly, greater shading during the breeding season would result in location estimates displaced south of the true breeding locations, and the northernmost location estimates are often most consistent with the true location (Porter and Smith 2013). In this case, the northern location estimates, as indicated by the kernel density estimates, fell across the region of Connecticut to Nova Scotia, and they included the area of Massachusetts, where a Northern Waterthrush banded in Puerto Rico was subsequently encountered during the breeding season (Fig. 3).
However, these data illustrate another challenge of light-level geolocation: locations that occur over the ocean are easily recognized as problematic, but for locations that fall on land, there is no easy way to recognize unrealistic locations. For the data sets that suggest breeding locations over the ocean, we also tried the Hill-Ekstrom calibration method (Lisovski et al. 2020) but were unable to identify a definitive sun elevation angle using this approach. Considering the location uncertainty associated with light-level geolocation, future tracking efforts using alternative tracking devices such as automated telemetry (e.g., Motus network) or archival global positioning system (GPS) tags may provide greater insights into migratory routes and breeding locations for Northern Waterthrushes.
Despite the limited accuracy of the location information, the light-level data provided other information about the annual cycle of these birds. Independent of the location estimates, we used raw light-level data to generate information about migratory and reproductive behavior that would not be accessible with archival GPS tags or automated telemetry. Using light-level data recorded by geolocators, we were able to determine that the Northern Waterthrushes we tracked departed their wintering locations in mid- to late April and arrived at their breeding grounds in mid- to late May. Additionally, we were able to determine that some birds began nesting in early June, which allowed us to identify them as breeding females. One nesting period was particularly short, which may reflect a failed nesting attempt, but there was no evidence of renesting. Northern Waterthrushes are not known to double brood, though they are known to renest after nests are lost to weather or predation (Whitaker and Eaton 2020). Although light levels have been used to infer reproductive behavior in previous studies (Burger et al. 2012, Loktionov et al. 2015, Smith et al. 2020), we suggest that it remains a largely underused aspect of geolocator data.
Because only female Northern Waterthrushes incubate, the light-level nesting data provide additional information that can be used to assign sexes of individuals captured on the wintering ground. Covino (2015) found that Northern Waterthrushes with wing chord < 72 mm or > 75.5 mm could be sexed as females and males with 95% accuracy. The wing chord of nearly all of our tracked birds fell at the cusp or within the range of overlap (Table 2), such that most of these individuals could not have been confidently sexed on the wintering grounds using morphological measurements alone. Thus, light-level records provide an additional source of information that can be used to help determine the sex of monomorphic species captured outside of the breeding season. The possibility of using this information highlights the importance of archiving raw light-level data in addition to derived location estimates in the event that further information can be extracted as new techniques become available (Kranstauber et al. 2011, Lisovski et al. 2020).
In contrast to many bird migration tracking studies that deploy tracking devices on birds during the breeding season, we deployed geolocators on Northern Waterthrush wintering grounds in Puerto Rico. The bias toward deploying tracking devices during the breeding season likely plays a role in the noted bias toward tracking males (McKinnon and Love 2018), as males tend to be easier to detect because they sing, respond more aggressively to audio playback, and exhibit higher site fidelity. The relatively few studies that have tracked both male and female birds have suggested that differential migration strategies may be overlooked and have potentially important conservation implications (Woodworth et al. 2016, Saino et al. 2017, Deakin et al. 2019). Deploying geolocators on wintering Northern Waterthrushes allowed us to track female birds (though unknowingly at the time) and gather information on their migratory and nesting phenology. Additionally, we demonstrate that raw light data from light-level geolocators can be used as additional evidence for sex determination when morphometrics such as a wing chord are inconclusive. Although our sample size limited our ability to compare male and female migration strategies, our results reinforce the idea that it remains an often-overlooked area of research with important conservation implications (McKinnon and Love 2018).
We found that birds banded or tagged with geolocators in the dry season (January–April) were more likely to be recaptured than those tagged or banded earlier in the year during the rainy season (September–December). We suspect that differences in recapture rates are related to status and ages of the birds passing through the area in these two time periods. Between September and December, we suspect more of the captures are transients (some of which may be bound for wintering sites in South America) and young birds that have a lower probability of survival. In contrast, later in the year, during the dry season, we were more likely to capture older birds that had established territories at the site (I. Rodríguez-Colón, unpublished data). It is possible that these individuals are more likely to survive to the following year and more likely to return to the same territories.
Although we did not find a significant difference between the recovery rate of birds with bands only and those with both bands and geolocators, there tended to be lower return rates of birds that were tagged with larger (0.55 g) geolocators during the rainy season. This finding, in conjunction with other evidence that tags with a larger relative load have a negative effect on return rates (Brlík et al. 2020), suggests that out of an abundance of caution, lighter tracking devices should be used when available. Given the stark contrast between seasonal return rates, we recommend that efforts to deploy tags on wintering Northern Waterthrushes focus efforts between January and April.
Our results illustrate that despite the limitations of light-level location in estimating locations of Northern Waterthrushes, it offers a unique opportunity to generate information about reproductive behavior in a manner that allows the information to be tied to migration timing and non-breeding season conditions. Expanding efforts to track migratory birds with tags deployed on their wintering grounds will provide an exciting opportunity to build an understanding of carry-over effects and other aspects of migratory bird biology.
RESPONSES TO THIS ARTICLE
Responses to this article are invited. If accepted for publication, your response will be hyperlinked to the article. To submit a response, follow this link. To read responses already accepted, follow this link.
ACKNOWLEDGMENTS
This study was conducted under the authorization of Animal Care and Use Committee Protocols A03-006-13 and A03-023-16 of the Ana G Méndez University System Office of Compliance. We thank Ian José Pérez Cruz, Mariangely Colón Cruz, and the many students and volunteers who assisted with this project, including Noraisha Bonilla, Jerry Agosto Díaz, Nicole Camacho Fontánez, Soely Luyando Flusa, Guillermo Plaza Rodríguez, Braiam Rosado, Roy Santiago Parilla, Andrea Carolina Torres López, Nangeshi Plaud Pabón, Kenneth Torres, Clarisse M. Carrasquillo, Waleska Vázquez-Carrero, E. Emilio Font, Giselle Caraballo, Liz Mari Rivera, Marie (Ellen) Pacheco, Keishla Rodriguez Graciani, Keila Martínez, Manuel Seda, Julio Salgado, Edwin González, Gerardo Silva, Suheilie Rodríguez, Luis Sanabria, Ryan A. Díaz Bonet, Vincente Vera, Cari López, Keysha Carrasquillo, and others, as well as Erik Johnson, James Fox, and Peter Olsson, who provided advice on geolocator deployment and data interpretation. Partial financial support was provided by the Blake-Nuttall Fund of the Nuttall Ornithological Club and the special fund for research of the Ana G Méndez University System. The manuscript was improved by comments from two anonymous reviewers.
DATA AVAILABILITY
The data/code that support the findings of this study are openly available in Movebank at https://www.movebank.org/cms/webapp?gwt_fragment=page=studies,path=study1259878781, reference number 1259878781.
LITERATURE CITED
Albert, S., J. D. Wolfe, J. Kellerman, T. Sherry, B. J. M. Stutchbury, N. J. Bayly and A. Ruiz-Sánchez. 2020. Habitat ecology of Nearctic–Neotropical migratory landbirds on the nonbreeding grounds. Ornithological Applications 122(4):duaa055. https://doi.org/10.1093/condor/duaa055
Bayly, N. J., K. V. Rosenberg, W. E. Easton, C. Gómez, J. Carlisle, D. N. Ewert, A. Drake, and L. Goodrich. 2018. Major stopover regions and migratory bottlenecks for Nearctic-Neotropical landbirds within the Neotropics: a review. Bird Conservation International 28(1):1-26. https://doi.org/10.1017/S0959270917000296
Brlík, V., J. Koleček, M. Burgess, S. Hahn, D. Humple, M. Krist, J. Ouwehand, E. L. Weiser, P. Adamík, J. A. Alves, D. Arlt, S. Barišic, D. Becker, E. J. Belda, V. Beran, C. Both, S. P. Bravo, M. Briedis, B. Chutný, D. Cikovic, N. W. Cooper, J. S. Costa, V. R. Cueto, T. Emmenegger, K. Fraser, O. Gilg, M. Guerrero, M. T. Hallworth, C. Hewson, F. Jiguet, J. A. Johnson, T. Kelly, D. Kishkinev, M. Leconte, T. Lislevand, S. Lisovski, C. López, K. P. McFarland, P. P. Marra, S. M. Matsuoka, P. Matyjasiak, C. M. Meier, B. Metzger, J. S. Monrós, R. Neumann, A. Newman, R. Norris, T. Pärt, V. Pavel, N. Perlut, M. Piha, J. Reneerkens, C. C. Rimmer, A. Roberto-Charron, C. Scandolara, N. Sokolova, M. Takenaka, D. Tolkmitt, H. van Oosten, A. H. J. Wellbrock, H. Wheeler, J. van der Winden, K. Witte, B. K. Woodworth, and P. Procházka. 2020. Weak effects of geolocators on small birds: a meta-analysis controlled for phylogeny and publication bias. Journal of Animal Ecology 89(1):207-220. https://doi.org/10.1111/1365-2656.12962
Bulla, M., M. Valcu, A. M. Dokter, A. G. Dondua, A. Kosztolányi, A. L. Rutten, B. Helm, B. K. Sandercock, B. Casler, B. J. Ens, C. S. Spiegel, C. J. Hassell, C. Küpper, C. Minton, D. Burgas, D. B. Lank, D. C. Payer, E. Y. Loktionov, E. Nol, E. Kwon, F. Smith, H. R. Gates, H. Vitnerová, H. Prüter, J. A. Johnson, J. J. H. St Clair, J.-F. Lamarre, J. Rausch, J. Reneerkens, J. R. Conklin, J. Burger, J. Liebezeit, J. Bêty, J. T. Coleman, J. Figuerola, J. C. E. W. Hooijmeijer, J. A. Alves, J. A. M. Smith, K. Weidinger, K. Koivula, K. Gosbell, K.-M. Exo, L. Niles, L. Koloski, L. McKinnon, L. Praus, M. Klassen, M.-A. Giroux, M. Sládeček, M. L. Boldenow, M. I. Goldstein, M. Šálek, N. Senner, N. Rönkä, N. Lecomte, O. Gilg, O. Vincze, O. W. Johnson, P. A. Smith, P. F. Woodard, P. S. Tomkovich, P. F. Battley, R. Bentzen, R. B. Lanctot, R. Porter, S. T. Saalfeld, S. Freeman, S. C. Brown, S. Yezerinac, T. Székely, T. Montalvo, T. Piersma, V. Loverti, V.-M. Pakanen, W. Tijsen, and B. Kempenaers. 2016. Unexpected diversity in socially synchronized rhythms of shorebirds. Nature 540:109-113. https://doi.org/10.1038/nature20563
Burger, J., L. J. Niles, R. R. Porter and A. D. Dey. 2012. Using geolocator data to reveal incubation periods and breeding biology in Red Knots Calidris canutus rufa. Wader Study Group Bulletin 119(1):26-36. https://www.waderstudygroup.org/article/1890/
Burson, S. L. III, L. R. Reitsma, and P. D. Hunt. 2005. Conservation implications of multiple habitat use by Northern Waterthrushes during the non-breeding season. Journal of Caribbean Ornithology 18:72-76. https://jco.birdscaribbean.org/index.php/jco/article/view/223
Covino, K. M. 2015. Determination of sex using morphometrics in the Northern Waterthrush (Parkesia noveboracensis) and Swainson’s Thrush (Catharus ustulatus). Wilson Journal of Ornithology 127(4):706-711. https://doi.org/10.1676/14-173.1
Deakin, Z., K. C. Hamer, R. B. Sherley, S. Bearhop, T. W. Bodey, B. L. Clark, W. J. Grecian, M. Gummery, J. Lane, G. Morgan, L. Morgan, R. A. Phillips, E. D. Wakefield, and S. C. Votier. 2019. Sex differences in migration and demography of a wide-ranging seabird, the northern gannet. Marine Ecology Progress Series 622:191-201. https://doi.org/10.3354/meps12986
DeLuca, W. V., B. K. Woodworth, C. C. Rimmer, P. P. Marra, P. D. Taylor, K. P. McFarland, S. A. Mackenzie, and R. D. Norris. 2015. Transoceanic migration by a 12 g songbird. Biology Letters 11(4):20141045. https://doi.org/10.1098/rsbl.2014.1045
Eaton, S. W. 1957. A life history study of Seiurus noveboracensis. Science Studies (St. Bonaventure University) 19:7-36.
Ewel, J. J., and J. L. Whitmore. 1973. The ecological life zones of Puerto Rico and the U.S. Virgin Islands. Research Paper ITF-018. U.S. Department of Agriculture Forest Service, Institute of Tropical Forestry, Rio Piedras, Puerto Rico. https://www.fs.usda.gov/research/treesearch/5551
Gow, E. A., K. L. Wiebe, and J. W. Fox. 2015. Cavity use throughout the annual cycle of a migratory woodpecker revealed by geolocators. Ibis 157(1):167-170. https://doi.org/10.1111/ibi.12206
Hallworth, M. T., T. S. Sillett, S. L. Van Wilgenburg, K. A. Hobson, and P. P. Marra. 2015. Migratory connectivity of a Neotropical migratory songbird revealed by archival light-level geolocators. Ecological Applications 25(2):336-347. https://doi.org/10.1890/14-0195.1
Harrison, X. A., J. D. Blount, R. Inger, D. R. Norris, and S. Bearhop. 2011. Carry-over effects as drivers of fitness differences in animals. Journal of Animal Ecology 80(1):4-18. https://doi.org/10.1111/j.1365-2656.2010.01740.x
Kranstauber, B., A. Cameron, R. Weinzerl, T. Fountain, S. Tilak, M. Wikelski, and R. Kays. 2011. The Movebank data model for animal tracking. Environmental Modelling and Software 26(6):834-835. https://doi.org/10.1016/j.envsoft.2010.12.005
Langin, K. M., P. P. Marra, Z. Németh, F. R. Moore, T. K. Kyser, and L. M. Ratcliffe. 2009. Breeding latitude and timing of spring migration in songbirds crossing the Gulf of Mexico. Journal of Avian Biology 40(3):309-316. https://doi.org/10.1111/j.1600-048X.2008.04496.x
Lisovski, S., S. Bauer, M. Briedis, S. C. Davidson, K. L. Dhanjal-Adams, M. T. Hallworth, J. Karagicheva, C. M. Meier, B. Merkel, J. Ouwehand, L. Pedersen, E. Rakhimberdiev, A. Roberto-Charron, N. E. Seavy, M. D. Sumner, C. M. Taylor, S. J. Wotherspoon, and E. S. Bridge. 2020. Light-level geolocator analyses: a user’s guide. Journal of Animal Ecology 89(1):221-236. https://doi.org/10.1111/1365-2656.13036
Lisovski, S., and S. Hahn. 2012. GeoLight - processing and analysing light-based geolocator data in R. Methods in Ecology and Evolution 3(6):1055-1059. https://doi.org/10.1111/j.2041-210X.2012.00248.x
Lisovski, S., C. M. Hewson, R. H. G. Klaassen, F. Korner-Nievergelt, M. W. Kristensen, and S. Hahn. 2012. Geolocation by light: accuracy and precision affected by environmental factors. Methods in Ecology and Evolution 3(3):603-612. https://doi.org/10.1111/j.2041-210X.2012.00185.x
Lisovski, S., S. Wotherspoon, and M. Sumner. 2016. TwGeos: basic data processing for light-level geolocation archival tags. R package version 0.1.2. https://github.com/slisovski/TwGeos
Loktionov, E. Y., P. S. Tomkovich, and R. R. Porter. 2015. Study of incubation, chick rearing and breeding phenology of Red Knots Calidris canutus rogersi in sub-Arctic Far Eastern Russia aided by geolocators. Wader Study 122(2):142-152. https://doi.org/10.18194/ws.00012
McKinnon, E. A., and O. P. Love. 2018. Ten years tracking the migrations of small landbirds: lessons learned in the golden age of bio-logging. Auk 135(4):834-856. https://doi.org/10.1642/AUK-17-202.1
McKinnon, E. A., C. Q. Stanley, K. C. Fraser, M. M. MacPherson, G. Casbourn, P. P. Marra, C. E. Studds, N. Diggs, and B. J. M. Stutchbury. 2013. Estimating geolocator accuracy for a migratory songbird using live ground-truthing in tropical forest. Animal Migration 2(2013):1-8. https://doi.org/10.2478/ami-2013-0001
Porter, R., and P. A. Smith. 2013. Techniques to improve the accuracy of location estimation using light-level geolocation to track shorebirds. Wader Study Group Bulletin 120(3):147-158. https://www.waderstudygroup.org/article/1280/
R Core Team. 2021. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.r-project.org/
Rakhimberdiev, E., A. Saveliev, T. Piersma, and J. Karagicheva. 2017. FLightR: an R package for reconstructing animal paths from solar geolocation loggers. Methods in Ecology and Evolution 8(11):1482-1487. https://doi.org/10.1111/2041-210X.12765
Rappole, J. H., and A. R. Tipton. 1991. New harness design for attachment of radio transmitters to small passerines. Journal of Field Ornithology 62(3):335-337. https://sora.unm.edu/node/51662
Reitsma, L., P. Hunt, S. L. Burson, and B. B. Steele. 2002. Site fidelity and ephemeral habitat occupancy: Northern Waterthrush use of Puerto Rican black mangroves during the nonbreeding season. Wilson Bulletin 114(1):99-105. https://doi.org/10.1676/0043-5643(2002)114[0099:SFAEHO]2.0.CO;2
Rockwell, S. M., C. I. Bocetti, and P. P. Marra. 2012. Carry-over effects of winter climate on spring arrival date and reproductive success in an endangered migratory bird, Kirtland’s Warbler (Setophaga kirtlandii). Auk 129(4):744-752. https://doi.org/10.1525/auk.2012.12003
Rodríguez-Colón, I. 2012. Inference of habitat connectivity via habitat use by resident and migratory birds between secondary dry forest and mangroves in Jobos Bay National Estuarine Research Reserve. Thesis. Universidad del Turabo, Gurabo, Puerto Rico.
Saino, N., R. Ambrosini, M. Caprioli, A. Romano, M. Romano, D. Rubolini, C. Scandolara, and F. Liechti. 2017. Sex-dependent carry-over effects on timing of reproduction and fecundity of a migratory bird. Journal of Animal Ecology 86(2):239-249. https://doi.org/10.1111/1365-2656.12625
Schaffner, F. C., I. Rodríguez-Colón, E. E. Font, and W. Vázquez-Carrero. 2017a. Initial deployment of archival light-level geolocators on Northern Waterthrushes (Parkesia noveboracensis) at Jobos Bay, Puerto Rico. Ambientis 2017:59-76.
Schaffner, F. C., I. Rodríguez-Colón, W. Vázquez-Carrero, and E. E. Font-Nicole. 2017b. Feather hydrogen stable isotopes reveal migratory and interhabitat connectivity of North American wintering songbirds in coastal secondary dry forest on the south coast of Puerto Rico. Ambientis 2017:39-58.
Smith, J. A. M., K. Regan, N. W. Cooper, L. Johnson, E. Olson, A. Green, J. Tash, D. C. Evers, and P. P. Marra. 2020. A green wave of saltmarsh productivity predicts the timing of the annual cycle in a long-distance migratory shorebird. Scientific Reports 10:20658. https://doi.org/10.1038/s41598-020-77784-7
Smith, J. A. M., L. R. Reitsma, and P. P. Marra. 2010. Moisture as a determinant of habitat quality for a nonbreeding Neotropical migratory songbird. Ecology 91(10):2874-2882. https://doi.org/10.1890/09-2212.1
Smith, J. A. M., L. R. Reitsma, L. L. Rockwood, and P. P. Marra. 2008. Roosting behavior of a Neotropical migrant songbird, the northern waterthrush Seiurus noveboracensis, during the non-breeding season. Journal of Avian Biology 39:460-465. https://doi.org/10.1111/j.0908-8857.2008.04227.x
Streby, H. M., T. L. McAllister, S. M. Peterson, G. R. Kramer, J. A. Lehman, and D. E. Andersen. 2015. Minimizing marker mass and handling time when attaching radio-transmitters and geolocators to small songbirds. Condor 117(2):249-255. https://doi.org/10.1650/CONDOR-14-182.1
Webster, M. S., P. P. Marra, S. M. Haig, S. Bensch, and R. T. Holmes. 2002. Links between worlds: unraveling migratory connectivity. Trends in Ecology and Evolution 17(2):76-83. https://doi.org/10.1016/S0169-5347(01)02380-1
Whitaker, D. M., and S. W. Eaton. 2020. Northern Waterthrush (Parkesia noveboracensis), version 1.0. In A. F. Poole, editor. Birds of the world. Cornell Lab of Ornithology, Ithaca, New York, USA. https://doi.org/https://doi.org/10.2173/bow.norwat.01
Williams, C. O., R. Lowrance, D. D. Bosch, J. R. Williams, E. Benham, A. Dieppa, R. Hubbard, E. Mas, T. Potter, D. Sotomayor, E. M. Steglich, T. Strickland, and R. G. Williams. 2013. Hydrology and water quality of a field and riparian buffer adjacent to a mangrove wetland in Jobos Bay watershed, Puerto Rico. Ecological Engineering 56:60-68. https://doi.org/10.1016/j.ecoleng.2012.09.005
Woodworth, B. K., A. E. M. Newman, S. P. Turbek, B. C. Dossman, K. A. Hobson, L. I. Wassenaar, G. W. Mitchell, N. T. Wheelwright, and D. R. Norris. 2016. Differential migration and the link between winter latitude, timing of migration, and breeding in a songbird. Oecologia 181:413-422. https://doi.org/10.1007/s00442-015-3527-8
Fig. 1
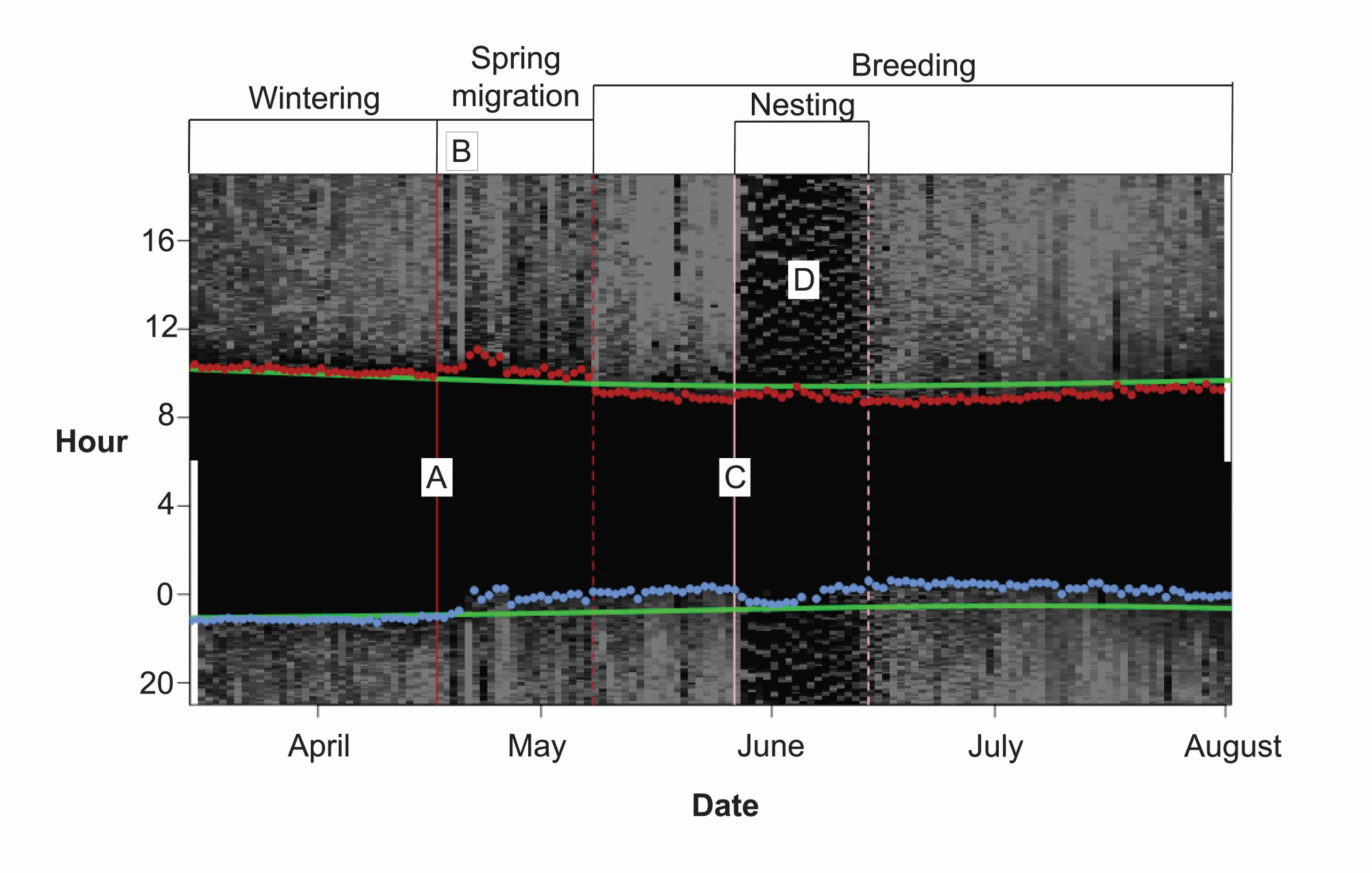
Fig. 1. Example light-level reading from geolocator tag X344, which was deployed on a Northern Waterthrush. The transitions between stages of the full annual cycle are annotated in the brackets above the reading. The departure of birds on spring migration (A) was identified by sunrise and sunset times (red and blue dots) deviating from sunrise/sunset times in Puerto Rico (green lines) and the multiple hours of high-intensity light values as birds made an extended daytime flight away from the island (B). The nesting period (C) was determined by identifying multiple days of alternating light and dark bouts indicative of incubation bouts during daylight hours (D).

Fig. 2
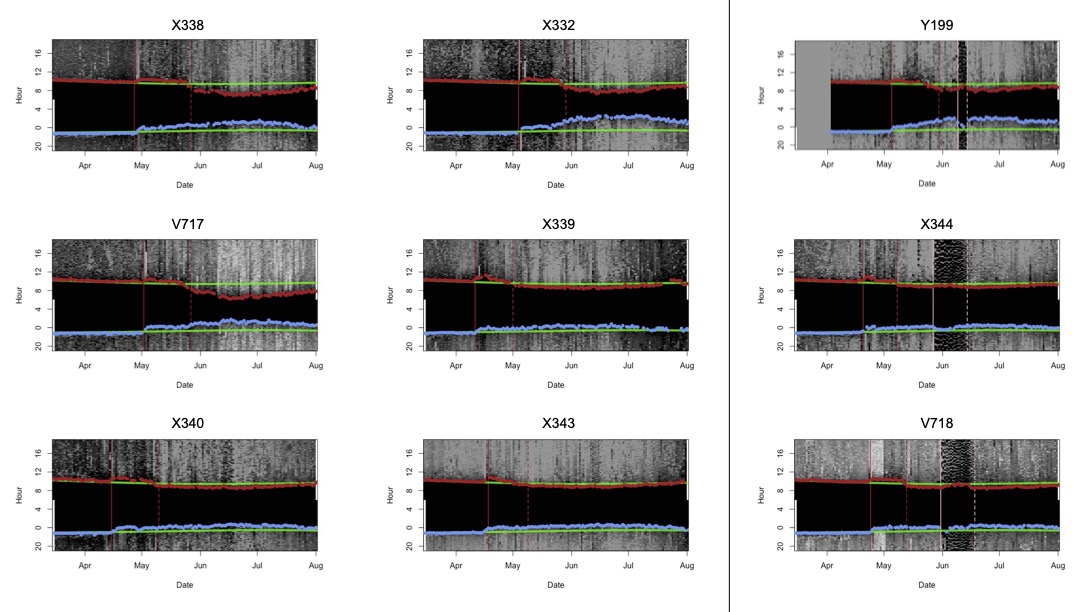
Fig. 2. Annotated light-level recordings from nine geolocators that were deployed on Northern Waterthrushes and used to determine the sex of each bird. Putative male birds are represented on the left panel, and female birds are represented on the right. Sunrise and sunset at the deployment location are plotted in green, with the observed twilight events plotted in red and blue.

Fig. 3
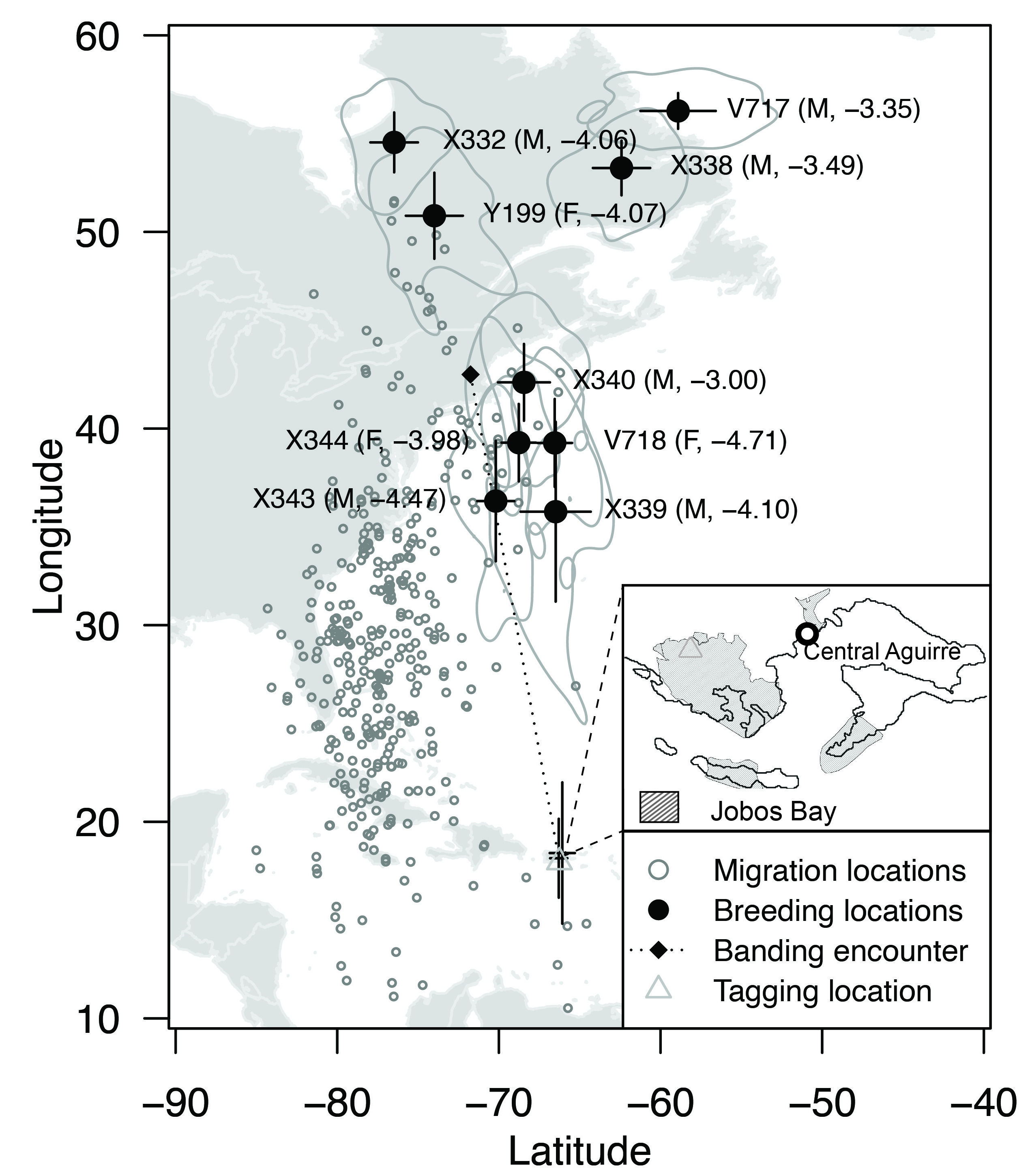
Fig. 3. Migration locations and average breeding locations for nine Northern Waterthrushes that were tagged with light-level geolocators. Standard deviations of latitude and longitude are denoted by black lines and 95% kernel density estimates are denoted by gray polygons. The presumed sex (M = male, F = female) and calibrated sun-elevation angle used in the analysis are given in parentheses. Maps for individual birds are in Fig. 4.

Fig. 4
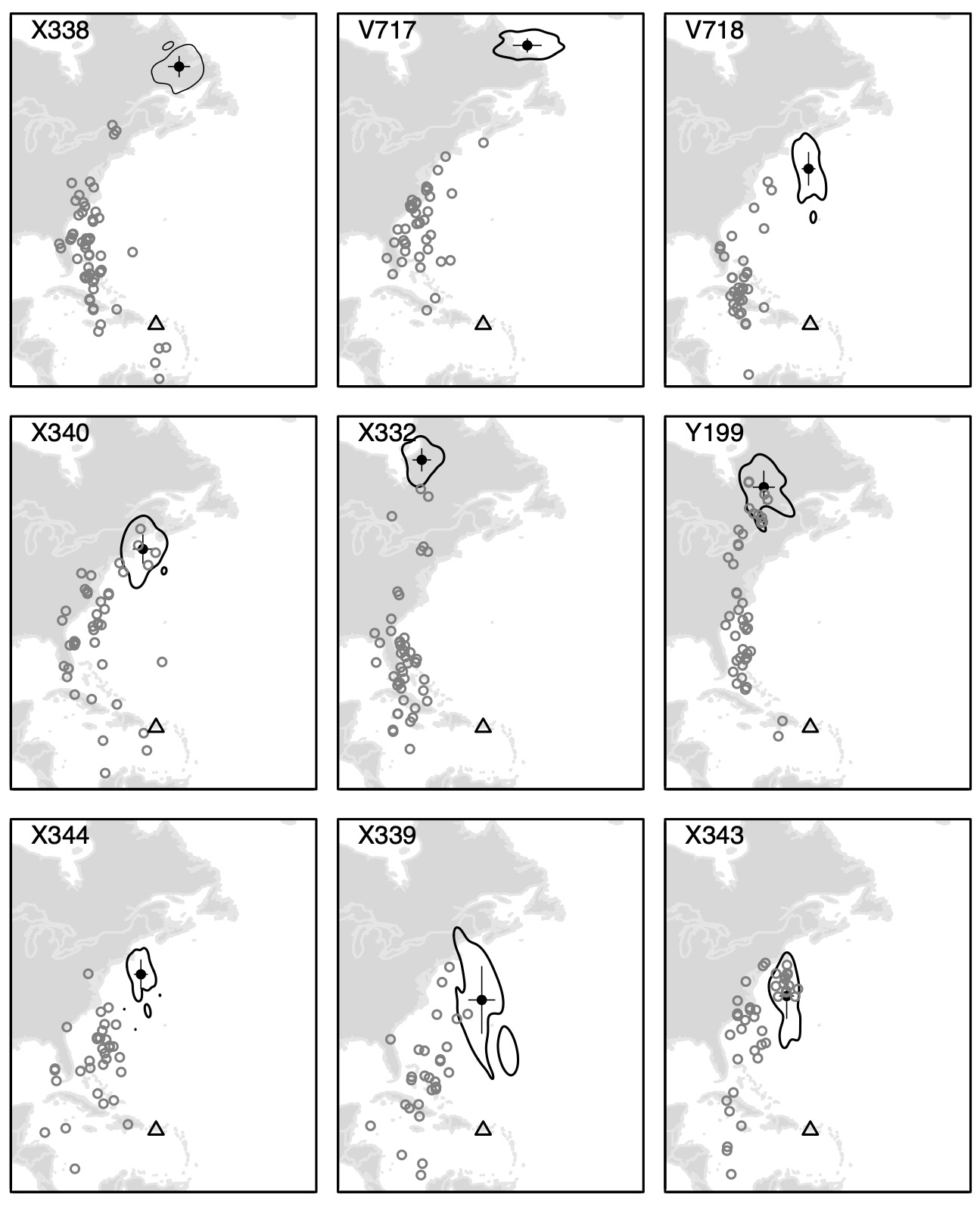
Fig. 4. Migration locations and average breeding locations for nine individual Northern Waterthrushes that were tagged with light-level geolocators.

Table 1
Table 1. Comparison of the deployment, recovery, and recapture rates for Northern Waterthrushes banded with either leg bands only or both geolocators and leg bands during the rainy and dry seasons. Birds were captured and fitted with geolocators or bands between September 2015 and April 2016. Geolocators were recovered and birds were recaptured in the year following deployment. Numbers refer to the number of birds captured or recaptured at least once, rather than the total number of encounters, given that some individuals were recaptured on multiple occasions.
| Deployment type | Number deployed | Number recaptured | Recapture rate (%) |
| Rainy season (September to December 2015) | |||
| Band only | 81 | 20 | 24.7 |
| Band and geolocator | 19 | 2 | 10.5 |
| Dry season (January to April 2016) | |||
| Band only | 61 | 26 | 42.7 |
| Band and geolocator | 21 | 9 | 42.9 |
Table 2
Table 2. Summary of the putative sex, wing chord length, deployment length, and tag model used for each bird tagged with a light-level geolocator.
| Bird | Wing cord length (mm) | Presumed sex | Deployment date (m/d/y) | Recovery date (m/d/y) | Days deployed | Tag model |
| V717† | NA | Male | 12/5/2015 | 10/7/2016 | 307 | Larger |
| V718 | 72 | Female | 12/6/2015 | 10/22/2016 | 321 | Larger |
| X340 | 73 | Female | 3/5/2016 | 10/22/2016 | 231 | Smaller |
| X338 | 75 | Male | 3/5/2016 | 4/2/2017 | 393 | Smaller |
| X343 | 71 | Male | 3/6/2016 | 10/7/2016 | 215 | Smaller |
| X332 | 75 | Male | 3/6/2016 | 2/5/2017 | 336 | Smaller |
| X339† | 75 | Male | 2/28/2016 | 3/11/2017 | 377 | Smaller |
| X344 | 72 | Female | 3/12/2016 | 10/16/2016 | 218 | Smaller |
| X346 | 69 | Unknown | 3/12/2016 | 11/11/2016 | 244 | Smaller |
| Y199 | 71 | Female | 4/2/2016 | 10/15/2016 | 196 | Smaller |
| X047 | 74 | Unknown | 4/3/2016 | 9/11/2016 | 161 | Larger |
| † Bird was captured at least once before the study duration. | ||||||








