The following is the established format for referencing this article:
Ferreira, D., L. Figueira, F. Cunha, and L. E. Lopes. 2023. Molt strategy and delayed plumage maturation in the Lined Seedeater. Journal of Field Ornithology 94(3):9.ABSTRACT
Lined Seedeaters Sporophila lineola, an intra-tropical migratory songbird, exhibit extensive phenotypic variation, with characteristic black-and-white male and brownish female plumages. In this study, we investigated whether variation in male plumage represents delayed plumage maturation, as reported for many other Sporophila seedeaters. We used data on molt and plumage from a seven-year-long study of color-banded Lined Seedeaters in southeastern Brazil. We also gathered molt and plumage data from museum collections and citizen-science platforms to identify which molts occur outside the breeding grounds. Our findings show that Lined Seedeaters follow a complex basic strategy, but the possibility that some individuals exhibit a complex alternate strategy, which is a common strategy among congeners, cannot be ruled out. Preformative molt and fresh formative plumage were recorded within the breeding grounds in the last months of the breeding season. Prebasic molt also start on the breeding grounds and probably continue during migration to the wintering grounds. Observed phenotypic variation in plumage of Lined Seedeater males is a product of delayed plumage maturation. Breeding males in female-like plumage are formative individuals in their first breeding season. All monitored males acquired black-and-white definitive plumage after their first breeding season, during the second pre-basic molt, but we found limited evidence that some individuals may retain the brownish plumage for more than one cycle. Descriptions presented here advance our understanding of Sporophila molt strategies that can be used in future studies focusing on the evolutionary and ecological underpinnings of plumage variation.
RESUMEN
Sporophila lineola, una especie de ave cantora migratoria intra-tropical, muestra amplia variación fenotípica, con plumajes característicos blanco y negro en los machos y café en las hembras. En este estudio investigamos si la variación en el plumaje de los machos representa maduración retardada del plumaje, como es reportado para muchos otros semilleros del género Sporophila. Utilizamos datos de muda y del plumaje de un estudio de siete años de duración en individuos de Sporophila lineola anillados con anillos de color en el sureste de Brasil. También recolectamos datos de muda y plumaje de colecciones de museo y plataformas de ciencia ciudadana para identificar las mudas que ocurren fuera de las áreas de reproducción. Nuestros resultados muestran que Sporophila lineola sigue una estrategia compleja básica, pero la posibilidad que algunos individuos muestren una estrategia compleja alterna, la cual es una estrategia común en congéneres, no puede ser descartada. La muda preformativa y el plumaje formativo fresco fueron registrados en las áreas de reproducción en los últimos meses de la temporada reproductiva. La muda prebásica también inicia en las áreas de reproducción y probablemente continua durante la migración a los sitios de invierno. La variación fenotípica observada en el plumaje de los machos de Sporophila lineola es producto de maduración retardada del plumaje. Todos los machos monitoreados adquirieron el plumaje definitivo blanco y negros después de su primera temporada de reproducción, durante la segunda muda pre-básica, pero encontramos evidencia limitada que algunos individuos pueden retener el plumaje café por más de un ciclo. Las descripciones presentadas en este estudio avanzan nuestro entendimiento de las estrategias de muda en Sporophila que pueden ser usados en estudios futuros que se enfoquen en las bases evolutivas y ecológicas de la variación en el plumaje.
INTRODUCTION
Variation in molt-and-plumage sequences across age classes has been used to separate cohorts of temperate songbirds for decades (Pyle 1997). Despite recent advances in our knowledge about molt in tropical birds, we still lack fundamental information regarding the molt timing, duration, insertions, and extent for most Neotropical songbirds (Ryder and Wolfe 2009, Johnson and Wolfe 2017). Variation in molt-and-plumage sequences facilitate the delineation of cohorts, a necessary step toward elucidating demographic trends such as changes in age ratios, an index of recruitment and productivity. Additionally, variation in molt-and-plumage sequences are shaped by environmental pressures, with cascading effects on a species’ behavior. For example, small ant-following birds maintained strong flight by avoiding large gaps in their remiges through adaptively slowing the duration of their annual molts, a precursor to maintaining large home ranges (Wolfe et al. 2021). We now understand that timing, frequency, duration, and extent of molts have evolved in response to environmental and social constraints, thus influencing the evolution of dissimilar life-history strategies (Johnson and Wolfe 2017). As such, the lack of knowledge surrounding molt-and-plumage sequences in the tropics makes it challenging to further develop hypotheses aimed at explaining the diversity of life-history strategies observed within the tropics and across latitudes.
Males of sexually dichromatic birds often display remarkable phenotypic variation in plumage coloration and patterns, with some males showing a female-like dull plumage, whereas other males exhibit characteristically brightly colored definitive plumage (Greene et al. 2000, Karubian 2002, Karubian et al. 2008). Such phenotypic variation is often age-related, with males acquiring the definitive plumage only after the first potential breeding season, a process known as delayed plumage maturation (Rohwer et al. 1980, Hawkins et al. 2012). Delayed plumage maturation is common strategy among many sexually dichromatic and monogamous songbirds (Rohwer et al. 1980, Thompson 1991, Greene et al. 2000), such as manakins (Schaedler et al. 2021), tanagers (Palmerio and Massoni 2009), and seedeaters. For example, males of the Tawny-bellied Seedeater (Sporophila hypoxantha; Facchinetti et al. 2011) and the Tropeiro Seedeater (S. beltoni) look like females in their first breeding season, with Tropeiro Seedeaters taking four years to attain the definitive plumage (Repenning 2012).
In Lined Seedeaters (Sporophila lineola), breeding males exhibit two distinct plumage types: a characteristic black-and-white plumage and a female-like brownish plumage (Oliveira et al. 2010, Ferreira and Lopes 2017). However, the process underlying this phenotypic variation in males’ breeding plumage remains unclear. To investigate whether Lined Seedeaters undergo delayed plumage maturation, we used data on molt and plumage from a color-banded population to describe the plumage sequence and molt strategy in the species. Given that Lined Seedeaters are migratory and are present in the study area only during the breeding season, we also used museum and citizen-science data to investigate the timing and location of molts outside the breeding grounds.
METHODS
Study site
We conducted a field study at the Campus Florestal of the Universidade Federal de Viçosa (19°52′51″S, 44°24′49″W), municipality of Florestal, state of Minas Gerais, southeastern Brazil. The study area is a 1500-ha mosaic of secondary semideciduous forest fragments and various kinds of open habitats (Lopes and Marçal 2016), including human-made pastures of African grasses (Panicum and Brachiaria), active and abandoned crops, gardens, and small ponds (Ferreira and Lopes 2017).
Study species
The Lined Seedeater is an intra-tropical migratory species common in various types of open habitats throughout much of South America (Ridgely and Tudor 2009). The population that breeds in southeastern Brazil appears to migrate through central Brazil to its wintering grounds in northern South America. These wintering grounds are possibly shared, to some extent, with the other two breeding populations of the species, one from the Chaco of northern Argentina and northwestern Paraguay, and one from the Caatinga of northeastern Brazil (Cunha et al. 2022). These two populations exhibit phenological differences in relation to the breeding and migration dates when compared to the southeastern population (Silva 1995, Cunha et al. 2022). The first Lined Seedeaters arrive in the study area in late November and laying dates span from early December to late April (Ferreira and Lopes 2017). Males of this socially monogamous species defend small territories (Martins et al. 2021) where females build the nests and incubate the eggs; both sexes feed the nestlings (Oliveira et al. 2010, Ferreira and Lopes 2017).
This species has marked sexual dimorphism. Most males are black above and white below, with a white coronal stripe, broad white malar stripe, black throat, white band at the base of remiges, and white rump patch (Hilty 2002, Ridgely and Tudor 2009). In contrast, females are brownish, with throat, chest, and flanks of yellowish buff, contrasting with a rather sharply defined buffy white center of breast and belly (Hilty 2002). Some males exhibit a female-like plumage (Ferreira and Lopes 2017).
Capture of birds and molt examination
This research is part of the Lined Seedeater Project, a long-term monitoring program in operation since 2014. We captured individuals with mist nets at feeding sites and breeding territories, and banded them with a unique color combination and a numbered metallic band. Whenever possible, we color-banded six-day-old nestlings.
Molt-and-plumage data were collected during four breeding seasons (2017–2021), for which we applied the WRP molt-based aging system (Wolfe et al. 2010, Johnson and Wolfe 2017, Pyle et al. 2021). For that, we documented plumage appearance and coloration, as well as feather shape. We searched for possible molt limits among the wing coverts, body, and flight feathers. For feather abrasion, we followed the scheme proposed by Rogers (1990). The distinction between females and the similar-looking brownish males was based on their behavior (i.e., males sing, whereas females do not) and on the presence of a brood patch (only females incubate; Ferreira and Lopes 2017). We took, whenever possible, pictures of each color-banded individual with the wing and tail opened. Additionally, we used project data from 2014 to 2016, which only report the breeding-male plumage color (black and white or brownish), without molt-and-plumage data. These data were used to better understand the changes in plumage appearance of marked birds that were recaptured or sighted multiple times across the years.
Museum specimens
To identify the geographic location and timing of molts outside our study area, we examined specimens of the Lined Seedeater at the Museu de Zoologia of the Universidade de São Paulo, São Paulo (MZUSP), Museu Nacional, Rio de Janeiro (MNRJ), Carnegie Museum of Natural History, Pittsburgh (CM), and Louisiana State University Museum of Natural Sciences, Baton Rouge (LSUMZ). We followed the same criteria of field-captured birds to age and described the plumage of each specimen. We established sexes from museum labels.
Citizen-science data
In May 2020, with the same objective of museum data, we searched for photographic records of the species in two of the most comprehensive citizen-science projects for Neotropical birds: eBird, through the Macaulay Library (https://www.macaulaylibrary.org) and WikiAves (http://www.wikiaves.com). In each photo, we looked at plumage patterns, colors, and signs of molt.
RESULTS
We captured and banded a total of 465 individuals between 2017 and 2021, including 184 males, 183 females, and 98 individuals of unknown sex. We took standardized pictures of 69% (n = 322) of those captured birds, with the wing and tail opened. Among the color-banded individuals, in the first three breeding seasons sampled for molts (2017–2020; n = 399), 11% (n = 44; females = 19, males = 25) were recaptured at least once in a subsequent breeding season. We also color-banded 220 nestlings during the four monitored breeding seasons. Of the 155 color-banded nestlings that successfully fledged during the first three breeding seasons (2017–2021), only four individuals (2.6%) were observed in a subsequent season, with one of them being recaptured.
Based on field, museum, and citizen-science data, we suggest that Lined Seedeaters follow the complex basic strategy, with members of the species performing two molts in their first cycle (a preformative and a prebasic) and one molt in all subsequent cycles (a prebasic; Fig. 1). We were, however, uncertain about the occurrence of a partial alternate plumage in some individuals, whose lesser coverts looked fresher than the other wing coverts, but not distinct in color or quality. Because of that, we maintain the possibility of at least some individuals performing a complex alternate strategy, with an inserted molt, the prealternate, in all cycles.
We used eight molt cycle–based age categories for classifying the captured birds, including codes to indicate uncertainty of cycle and plumage (M-FCF: at minimum in formative plumage, n = 56, photos = 33).
First cycle juvenile plumage
Birds in the first cycle juvenile plumage (FCJ, n = 17, photos = 7) were found at our study site from mid January to late April (and probably May for late fledglings). Birds in the FCJ exhibit a brownish plumage much like adult females, with no apparent distinctions between sexes at this age. They feature loosely textured body plumage, slightly duller in the breast (A, Fig. 2), primaries (B, Fig. 2) and rectrices (C, Fig. 2) are thinner, more tapered, and of lower quality (duller and of lower barb density) than in basic plumages. Gape flanges are swollen and yellowish white soon after fledging.
First cycle preformative molt
The first cycle preformative molt (FPF, n = 34, photos = 24) is partial. The FPF includes body feathers, lesser, median, and all greater wing coverts (D, Fig. 2). Some individuals in the FPF can also replace the tertials (S7-9, n = 3) and some rectrices (n = 4; E, Fig. 2), but we never recorded the central rectrices being replaced in the FPF. Formative wing coverts are dark brown with brownish edging, which differ from the duller juvenile feathers (D, Fig. 2). Slightly swollen gape flanges can still be seen in most individuals. Birds undergoing the preformative molt were recorded from February to April at our study site.
First cycle formative plumage
The first cycle formative plumage (FCF, n = 52, photos = 41) is similar in both sexes and can be identified by the presence of molt limits between juvenile and formative feathers. These molt limits occur between greater and primary coverts, and sometimes also between secondaries and tertials, and between formative and juvenile rectrices. Some individuals (n = 10) in fresh FCF were recorded by the end of the breeding season, from March to April (F, Fig. 2). Recapture of an individual color-banded as a nestling (G, Fig. 2) revealed that males can return to the breeding grounds to breed for the first time (n = 1, photos = 1) in the FCF (H, Fig. 2) exhibiting a female-like plumage until the end of their first molt cycle. Males and females recorded in FCF from December to February showed the retained juvenile feathers notably worn.
Definitive prebasic molt
We documented only four individuals going through the definitive prebasic molt in the study area, including three cases identified as definitive prebasic molt (DPB) and one in the second prebasic molt (SPB). These individuals showed moderate to heavy traces of body molt (A and B, Fig. 3) but no sign of remiges molt (C, Fig. 3). In the SPB, the newly replaced rectrices, some great coverts, and tertials were glossy black, contrasting against the retained brownish feathers (F, Fig. 3). The body plumage was mottled with black-and-white feathers, evidencing the transition to the definitive male plumage (D and E, Fig. 3). The prebasic molt was recorded by the end of the breeding season, in March and April. However, those few individuals undergoing the prebasic molt, which were unringed when captured, were not observed again. Therefore, we cannot be sure if they were part of the studied breeding population or if they were transient birds that bred somewhere else.
Definitive cycle basic
Most Lined Seedeaters captured were in their definitive cycle basic (DCB; males: n = 130, photos = 110; females: n = 162, photos = 85), with no apparent molt or molt limits, while exhibiting broad and truncated rectrices (A and C, Fig. 4). Individuals in DCB captured in November, when they were arriving to the study area, exhibited fresh plumage with no sign of differential abrasion. Males in DCB are black above and white below, with white patches on their head, wings, and rump (D, Fig. 4); females in DCB are brownish (B, Fig. 4). All breeding males that were color-banded in the female-like plumage and that we could recapture (n = 6) or observe (n = 9; A and B, Fig. 5) in a subsequent season returned to the study area in the black-and-white definitive basic cycle plumage (C and D, Fig. 5). These observations, together with the record of one individual in SPB, indicate that males acquire the definitive black-and-white basic plumage during the second prebasic molt (SPB; D, E, and F, Fig. 3); i.e., they present delayed plumage maturation.
The two distinct phenotypes of breeding males observed in the study area are from different age classes: the most frequent are males in their definitive basic cycle that exhibit a black-and-white plumage; and the less common are males in their first cycle formative (and at minimum formative plumage for males not known to be in their first or later cycle), which exhibit a female-like brownish plumage (data from four breeding seasons, 2017–2021). Males in the definitive black-and-white plumage represented 83% (n = 123) of color-banded breeding males and were social fathers of 82% (n = 348) of the nests monitored. Males in female-like brownish plumage were 17% (n = 25) of color-banded breeding males and were social fathers of 18% (n = 76) of the nests monitored.
Museum specimens and variations
Field data and museum specimens suggest that some variation in when males acquire the definitive plumage may occur. We recorded six museum specimens (LSUMZ 125034, MZUSP 28986, MZUSP 28964, MZUSP 85417, MZUSP 79819, and MZUSP 38608, Appendix 1; A, Fig. 6) and twenty banded males (photos = 12; A, Fig. 7) in brownish plumage that showed all rectrices truncated, all feathers slightly abraded, and no apparent molt limits (B, Fig. 6). These brownish males differed from the much-worn plumage with clear molt limits of monitored males in the FCF (B, Fig. 7).
We captured nine males with a mix of black and brown feathers on their bodies, but with no signs of active molt. Three museum specimens (two collected in São Paulo and one in Amazonas; MZUSP 4364, MZUSP 54734, and MZUSP 19573, Appendix 1) showed the same pattern of plumage coloration. Intriguingly, in these individuals, the brownish coloration was mostly restricted to the edge of black feathers, a condition that is not typically observed in Lined Seedeaters, indicating that this was not a case of suspended molt (in which case the contrast would be between completely brown retained feathers and completely black replaced feathers). These distinctive, brown-edged feathers appear mostly in the body feathers and wing coverts (A, C, and D, Fig. 8), occurring less frequently in the flight feathers (B, Fig. 8).
Possible alternate plumage
An inserted molt within each molt cycle is termed prealternate. Among the monitored individuals, we occasionally observed what seems to be a subtle molt limit from a partial prealternate molt, i.e., first cycle alternate (FCA, n = 4, photos = 4) and definitive cycle alternate (DCA, n = 5, photos = 5; Fig. 9). This purported molt limit was characterized by the fresher and glossier lesser wing coverts, as well as by some fresher body feathers contrasting with worn-out greater and primary coverts and alula. Although this plumage pattern would indicate an alternate plumage resultant from a partial prealternate molt, we are not confident about it. We did not record individuals going through a first prealternate molt (FPA) or definitive prealternate molt (DPA) in the study area, which indicates that if these molts occur, they occurred in the wintering grounds or during migration. Moreover, a plumage, resulting from a second inserted molt within definitive cycles, was not detected.
Citizen-science data
Citizen-science photographs provided little expansion of the understanding of timing and location of molts in the Lined Seedeater. From the photographs available on online platforms, we identified 54 Lined Seedeaters in apparent molt activity or with a mix of black feathers contrasting against retained brown feathers. These records were from Brazil, Venezuela, and French Guiana, from both breeding and non-breeding grounds (Appendix 2). Most records were from Brazil (n = 50), covering the southeastern (n = 22), north (n = 12), south (n = 6), central-west (n = 6), and northeastern regions (n = 4). We could not identify with certainty the age classes of birds in most photographs, but some records clearly showed individuals in active molts (SPB, DPB, and UPU; n = 38). Most records of individuals in the prebasic molt were from the breeding grounds (n = 24; Goiás, Minas Gerais, Rio de Janeiro, São Paulo, Paraná, and Santa Catarina) rather than from the non-breeding grounds (n = 13), but this may be due to the larger number of bird watchers on the breeding grounds, i.e., sampling bias.
DISCUSSION
In this study, we used field, museum, and citizen-science data to describe the molt strategy and plumage sequence of Lined Seedeaters, detailing the process responsible for the phenotypic variation observed in breeding males. Lined Seedeaters follow a complex basic strategy, characterized by a preformative molt within the first cycle, followed by subsequent cycles exhibiting a prebasic molt. We also found limited evidence that some individuals in the same breeding population may follow the complex alternate strategy, with an inserted alternate molt in all cycles, a common strategy among members of the genus Sporophila. A complex alternate strategy has been reported for the Pearly-bellied Seedeater (S. pileata; Blaauw 1919), the Copper Seedeater (S. bouvreuil; Sick 1997), the Dark-throated Seedeater (S. ruficollis; Sick 1997), the Black-bellied Seedeater (S. melanogaster ; Sick 1997), the Variable Seedeater (S. corvina; Wolfe et al. 2009), the Tawny-bellied Seedeater (S. hypoxantha; Facchinetti et al. 2011), the Tropeiro Seedeater (S. beltoni; Repenning 2012), the Chestnut-bellied Seedeater (S. castaneiventris; Johnson and Wolfe 2017), the Gray Seedeater (S. intermedia; Moreno-Palacios et al. 2017), and the Chestnut-bellied Seed-finch (S. angolensis; Johnson and Wolfe 2017).
In all Sporophila species studied to date, the prebasic molt is complete and the prealternate molt is partial, but the extension of the preformative molt varies from partial in Tawny-bellied Seedeaters (Facchinetti et al. 2011) to complete in Chestnut-bellied Seed Finches, Gray Seedeaters, and Variable Seedeaters (Wolfe et al. 2009, Johnson and Wolfe 2017, Moreno-Palacios et al. 2017). Our findings show that Lined Seedeaters follow a partial preformative molt, a complete prebasic molt, and that some individuals may also exhibit a partial prealternate molt.
The prebasic molt is considered homologous across all birds, but the occurrence of inserted molts is variable between groups, and is usually correlated to the species’ life history (Wolfe et al. 2021). Preformative molts are likely an adaptive response to replace the poor-quality juvenile feathers, which need to grow quickly due a high nest-predation risk experienced by tropical birds (Wolfe et al. 2021). Prealternate molts are commonly found in birds that inhabit sunny, grassy areas, characteristic of the Sporophila group, where they are exposed to strong wear from heightened solar exposure in an abrasive habitat (Howell 2010, Wolfe et al. 2021).
For most migratory Sporophila seedeaters, the timing and location of different molts remain little known. Understanding molt-and-plumage sequences in Lined Seedeaters is particularly difficult because these species have at least three breeding populations (Cunha et al. 2022). One population breeds in southeastern Brazil, one in the Chaco of northern Argentina and northwestern Paraguay, and another in the Caatinga of northeastern Brazil. These populations possibly share a wintering area to some extent, yet they show some phenological differences in the timing of breeding and migration (Cunha et al. 2022). Moreover, the migratory route and the boundaries of the wintering grounds for each population remain ambiguous (Silva 1995, Cunha et al. 2022). These characteristics, coupled with scarce records from northern South America, make interpretation of museum specimens and citizen science challenging.
Our few records of prebasic molts in the study area were from individuals captured once and never recorded again. Most citizen-science records of birds in prebasic molt were from the breeding grounds in southeastern Brazil. However, photographs of individuals and museum specimens in the prebasic molt also appear in the non-breeding grounds in the northern part of South America and on route to the wintering grounds (Silva 1995, Cunha et al. 2022). Thus, our data indicate that post-reproductive individuals start their prebasic molt outside their breeding territories, but possibly within their breeding grounds, and continue molting along their migratory route to the wintering grounds. Lined Seedeaters may utilize stopover sites along their migratory route where individuals molt and then suspend molt to continue their migration. However, it is necessary to conduct studies tracking individuals at a fine spatial scale to determine more precisely the location and timing of molting for each population.
As we observed in Lined Seedeaters, other congeneric long-distance migrants (Silva 1999, Repenning 2012), such as the Double-collared Seedeater (S. caerulescens) and the Tropeiro Seedeater, usually undergo their preformative and prebasic molts on the breeding grounds, but outside breeding territories (Ortiz and Capllonch 2007, Repenning 2012). Our records of Lined Seedeaters undergoing a preformative molt appear at the end of their breeding season, although a few individuals in a fresh formative plumage were also recorded at the same time, indicating that juveniles that hatched early in the breeding season may exhibit a concordantly earlier preformative molt. Field data indicate that juveniles perform their preformative molt within their breeding grounds, with some of them finishing this molt before migration. Last, we never recorded an individual undergoing a prealternate molt. If Lined Seedeaters indeed perform a prealternate molt, it probably occurs on their wintering grounds, as is usual in migrant songbirds (Howell 2010).
Our results show that phenotypic variation in Lined Seedeaters is a product of delayed plumage maturation, with males breeding in female-like plumage during their first breeding season, and only acquiring the black-and-white adult plumage during their second prebasic molt. Breeding in subadult plumage is a common strategy among seedeaters (Armani 1985, Ryder and Wolfe 2009), as suggested by anecdotal observations in the literature for at least 12 species of Sporophila: the Ruddy-breasted Seedeater (Miller 1932, Meyer de Schauensee 1952, Haverschmidt 1968, Olson 1968), the Slate-colored Seedeater (S. schistacea; Meyer de Schauensee 1952, Junge and Mees 1961), the Plumbeous Seedeater (S. plumbea ; Meyer de Schauensee 1952), the Variable Seedeater (Olson 1968), the Dark-throated Seedeater, the Marsh Seedeater (S. palustris ; Areta 2009), the Lesson’s Seedeater (S. bouvronides ; Hilty 2002), the White-collared Seedeater (S. torqueola; Skutch 1954), the Pearly-bellied Seedeater (Belton 1985), the Copper Seedeater (Sick 1967), the Chestnut-bellied Seed-Finch (Haverschmidt 1968, Wetmore et al. 1984, Johnson and Wolfe 2017), and the Tropeiro Seedeater (Repenning 2012). In these studies, breeding males were referred as exhibiting an “immature” (Meyer de Schauensee 1952) or “juvenile like” (Sick 1967) plumage.
Sporophila seedeaters exhibit marked interspecific variation in the timing to acquire the definitive adult plumage. We found that Lined Seedeaters usually acquire definitive plumage during the second prebasic molt, like Tawny-bellied Seedeaters (Facchinetti et al. 2011), and Gray Seedeaters (Moreno-Palacios et al. 2017). For some other congeneric species, however, like the Slate-colored Seedeater (Restall et al. 2006), the Ruddy-breasted Seedeater (Dickey and Van Rossem 1938), and the Tropeiro Seedeater (Repenning 2012), the definitive plumage might take as many as four cycles to be acquired. Intraspecific variation in the timing to acquire the definitive plumage has been reported for Ruddy-breasted Seedeaters (Dickey and Van Rossem 1938) and Dark-throated Seedeaters (Areta 2009). Therefore, the retention of the brownish plumage beyond the second basic molt may be a likely explanation for our observations of brownish males showing truncated flight feather tips, higher quality plumage, and no apparent molt limits. Yet, despite this evidence that some Lined Seedeaters may retain brownish plumage for more than one cycle, we emphasize that all males monitored to date have reached the black-and-white plumage in a subsequent breeding season.
This study revealed that delayed plumage maturation explains the phenotypic variation in breeding males of Lined Seedeaters. This common strategy among members of several bird families might be an adaptive life-history strategy rooted in the incapacity of young adults to compete with older adults (Hawkins et al. 2012). The female-like plumage may act as a signal of subordination to reduce aggression from adult males (Lyon and Montgomerie 1986, Schaedler et al. 2021) in feeding areas and can also help young males to move easily across the territory of adult males and gain access to territories and females (Rohwer et al. 1980). Such hypotheses cover topics as broad as sexual selection, alternative mating strategies, and population structure within species (Karubian 2002, Karubian et al. 2008, Webster et al. 2008, Marques-Santos et al. 2018). Therefore, with this paper we open an avenue of possibilities to investigate several hypotheses related to the evolution of life-history strategies in seedeaters.
RESPONSES TO THIS ARTICLE
Responses to this article are invited. If accepted for publication, your response will be hyperlinked to the article. To submit a response, follow this link. To read responses already accepted, follow this link.
ACKNOWLEDGMENTS
We thank the Lined Seedeater Project team for their help during data collection. We are thankful to Dr. Gretchen Wagner and Dr. Jared Wolfe for their comments on a previous version of the manuscript. This study was funded in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brasil (CAPES) – Finance Code 001 (research fellowship to D. F. F.). L. E. L. received a research fellowship from CNPq (309660/2017-3 and 316960/2021-7). UFV allowed us to conduct this study in the area under their care and permits were granted by ICMBio (61078-1) and CEUA-UFV (03/2018).
DATA AVAILABILITY
The data are available upon request from the corresponding author.
LITERATURE CITED
Areta, J. I. 2009. Paedomorphosis in Sporophila seedeaters. Bulletin of the British Ornithologists’ Club 129:98-103.
Armani, G. C. 1985. Guide des passereaux granivores Embérizinés. Societe Nouvelle des Editions Boubee, Paris, France.
Belton, W. 1985. Birds of Rio Grande do Sul, Brazil. Part 2. Formicariidae through Corvidae. Bulletin of the American Museum of Natural History 180:1-241.
Blaauw, F. E. 1919. On the eclipse plumage of Spermophila pileata. Ibis 1:83-84. https://doi.org/10.1111/j.1474-919X.1919.tb02875.x
Cunha, F. C. R., L. E. Lopes, and A. Selezneva. 2022. Revealing migration schedule and potential breeding grounds of Lined Seedeaters using citizen-science data. Emu - Austral Ornithology 122:167-175. https://doi.org/10.1080/01584197.2022.2105235
Dickey, D., and A. Van Rossem. 1938. Birds of El Salvador. Field Museum of Natural History Zoological Series 23:1-658. https://doi.org/10.5962/bhl.title.3068
Facchinetti, C., B. Mahler, A. G. Di Giacomo, and J. C. Reboreda. 2011. Stages of plumage maturation of the Tawny-bellied Seedeater: evidence of delayed plumage maturation and cryptic differentiation between juveniles and females. Condor 113:907-914. https://doi.org/10.1525/cond.2011.110010
Ferreira, D. F., and L. E. Lopes. 2017. Natural history of the Lined Seedeater Sporophila lineola (Aves: Thraupidae) in southeastern Brazil. Journal of Natural History 51:1425-1435. https://doi.org/10.1080/00222933.2017.1319518
Greene, E., B. E. Lyon, V. R. Muehter, L. Ratcliffe, S. J. Oliver, and P. T. Boag. 2000. Disruptive sexual selection for plumage coloration in a passerine bird. Nature 407:1000-1003. https://doi.org/10.1038/35039500
Haverschmidt, F. 1968. Birds of Suriname. Oliver and Boyd, London, UK.
Hawkins, G. L., G. E. Hill, and A. Mercadante. 2012. Delayed plumage maturation and delayed reproductive investment in birds. Biological Reviews 87:257-274. https://doi.org/10.1111/j.1469-185X.2011.00193.x
Hilty, S. L. 2002. Birds of Venezuela. Princeton University Press, Princeton, New Jersey, USA. https://doi.org/10.1515/9781400834099
Howell, S. N. G. 2010. Molt in North American Birds. Houghton Mifflin Harcourt, New York, New York, USA.
Johnson, E. I., and J. D. Wolfe. 2017. Molt in Neotropical birds: life history and aging criteria. CRC Press, Boca Raton, Florida, USA. https://doi.org/10.4324/9781315119755
Junge, G. C. A., and G. F. Mees. 1961. The avifauna of Trinidad and Tobago. Zoologische Verhandelingen 37:1-172.
Karubian, J. 2002. Costs and benefits of variable breeding plumage in the Red-backed Fairywren. Evolution 56:1673-1682. https://doi.org/10.1111/j.0014-3820.2002.tb01479.x
Karubian, J., T. S. Sillett, and M. S. Webster. 2008. The effects of delayed plumage maturation on aggression and survival in male Red-backed Fairywrens. Behavioral Ecology 19:508-516. https://doi.org/10.1093/beheco/arm159
Lopes, L. E., and B. F. Marçal. 2016. Avifauna do campus Florestal da Universidade Federal de Viçosa, Minas, Brasil. Atualidades Ornitológicas 193:41-56.
Lyon, B. E., and R. D. Montgomerie. 1986. Delayed plumage maturation in passerine birds: reliable signaling by subordinate males? Evolution 40:605-615. https://doi.org/10.1111/j.1558-5646.1986.tb00511.x
Marques-Santos, F., U. Wischhoff, J. J. Roper, and M. Rodrigues. 2018. Delayed plumage maturation explains differences in breeding performance of Saffron Finches. Emu - Austral Ornithology 118:323-333. https://doi.org/10.1080/01584197.2018.1450637
Martins, G. M., F. C. R. Cunha, and L. E. Lopes. 2021. Territorial behavior of the migratory Lined Seedeater during the breeding season. Ornithology Research 29:133-139. https://doi.org/10.1007/s43388-021-00065-y
Meyer de Schauensee, R. 1952. A review of the genus Sporophila. Proceedings of the Academy of Natural Sciences of Philadelphia 104:153-196. http://www.jstor.org/stable/4064459
Miller, A. H. 1932. Observations on some breeding birds of El Salvador, Central America. Condor 34:8-17. https://doi.org/10.2307/1363786
Moreno-Palacios, M., S. Losada-Prado, and M. Á. Echeverry-Gálvis. 2017. Secuencia de mudas y plumajes de Volatinia jacarina y Sporophila intermedia en el valle del Magdalena. Ornitología Colombiana 16:1-21.
Oliveira, L. S., L. M. S. Sousa, P. V. Davanço, and M. R. Francisco. 2010. Breeding behaviour of the Lined Seedeater (Sporophila lineola) in southeastern Brazil. Ornitologia Neotropical 21:251-261.
Olson, S. L. 1968. Possible breeding of some seedeaters in subadult plumage. Florida Naturalist 41:20.
Ortiz, D., and P. Capllonch. 2007. Distribución y migración de Sporophila c. caerulescens en Sudamérica. Revista Brasileira Ornitologia 15:377-385.
Palmerio, A. G., and V. Massoni. 2009. Reproductive biology of female Saffron Finches does not differ by the plumage of the mate. Condor 111:715-721. https://doi.org/10.1525/cond.2009.080044
Pyle, P. 1997. Identification guide to North American birds: a compendium of information on identifying, ageing, and sexing “near-passerines” and passerines in the hand. Slate Creek Press, Bolinas, California, USA.
Pyle, P., M. Gahbauer, E. I. Johnson, T. B. Ryder, and J. D. Wolfe. 2021. Application of a global age-coding system (“WRP”), based on molts and plumages, for use in demographic and other studies of birds. Ornithology 139:1-12. https://doi.org/10.1093/ornithology/ukab063
Repenning, M. 2012. História natural, com ênfase na biologia reprodutiva, de uma população migratória de Sporophila aff. plumbea (Aves, Emberizidae) do sul do Brasil. Pontifícia Universidade Católica do Rio Grande do Sul, Porto Alegre, Brazil.
Restall, R., C. Rodner, and M. Lentino. 2006. Birds of northern South America: an identification guide: plates and maps. Volume 2. Christopher Helm, London, UK.
Ridgely, R., and G. Tudor. 2009. Field guide to the songbirds of South America: the passerines. University of Texas Press, Austin, Texas, USA.
Rogers, D. I. 1990. The use of feather abrasion in moult studies. Corella 14:141-147.
Rohwer, S., S. D. Fretwell, and D. M. Niles. 1980. Delayed maturation in passerine plumages and the deceptive acquisition of resources. American Naturalist 115:400-437. https://www.jstor.org/stable/2460726 https://doi.org/10.1086/283569
Ryder, T. B., and J. D. Wolfe. 2009. The current state of knowledge on molt-and-plumage sequences in selected tropical families: a review. Ornitologia Neotropical 20:1-18.
Schaedler, L. M., L. U. Taylor, R. O. Prum, and M. Anciães. 2021. Constraint and function in the predefinitive plumages of manakins (Aves: Pipridae). Integrative and Comparative Biology 61:1363-1377. https://doi.org/10.1093/icb/icab063
Sick, H. 1967. “Bico de Ferro”: overlooked seedeater from Rio de Janeiro (Sporophila, Fringillidae, Aves). Anais da Academia Brasileira de Ciências 39:307-314.
Sick, H. 1997. Ornitologia brasileira. Nova Fronteira, Rio de Janeiro, Brazil.
Silva, J. D. 1995. Seasonal distribution of the Lined Seedeater Sporophila lineola. Bulletin of the British Ornithologists’ Club 115:14-21.
Silva, J. M. C. 1999. Seasonal movements and conservation of seedeaters of the genus Sporophila in South America. Studies in Avian Biology 19:272-280.
Skutch, A. F. 1954. Life histories of Central American birds. Families Fringillidae, Thraupidae, Icteridae, Parulidae, and Coerebidae. Pacific Coast Avifauna 31:1-448.
Thompson, C. W. 1991. The sequence of molts and plumages in Painted Buntings and implications for theories of delayed plumage maturation. Condor 93:209-235. https://doi.org/10.2307/1368938
Webster, M. S., C. W. Varian, and J. Karubian. 2008. Plumage color and reproduction in the Red-backed Fairywren: why be a dull breeder? Behavioral Ecology 19:517-524. https://doi.org/10.1093/beheco/arn015
Wetmore, A., R. F. Pasquier, and S. L. Olson. 1984. The birds of the Republic of Panamá, Part 4 - Passeriformes: Hirundinidae (swallows) to Fringillidae (finches). Smithsonian Institution Press, Washington, D.C., USA.
Wolfe, J. D., P. Pyle, and C. J. Ralph. 2009. Breeding seasons, molt patterns, and gender and age criteria for selected northeastern Costa Rican resident landbirds. Wilson Journal of Ornithology 121:556-567. https://www.jstor.org/stable/20616943 https://doi.org/10.1676/08-111.1
Wolfe, J. D., T. B. Ryder, and P. Pyle. 2010. Using molt cycles to categorize the age of tropical birds: an integrative new system. Journal of Field Ornithology 81:186-194. https://doi.org/10.1111/j.1557-9263.2010.00276.x
Wolfe, J. D., R. S. Terrill, E. I. Johnson, L. L. Powell, and T. B. Ryder. 2021. Ecological and evolutionary significance of molt in lowland Neotropical landbirds. Ornithology 138:1-13. https://doi.org/10.1093/ornithology/ukaa073
Fig. 1
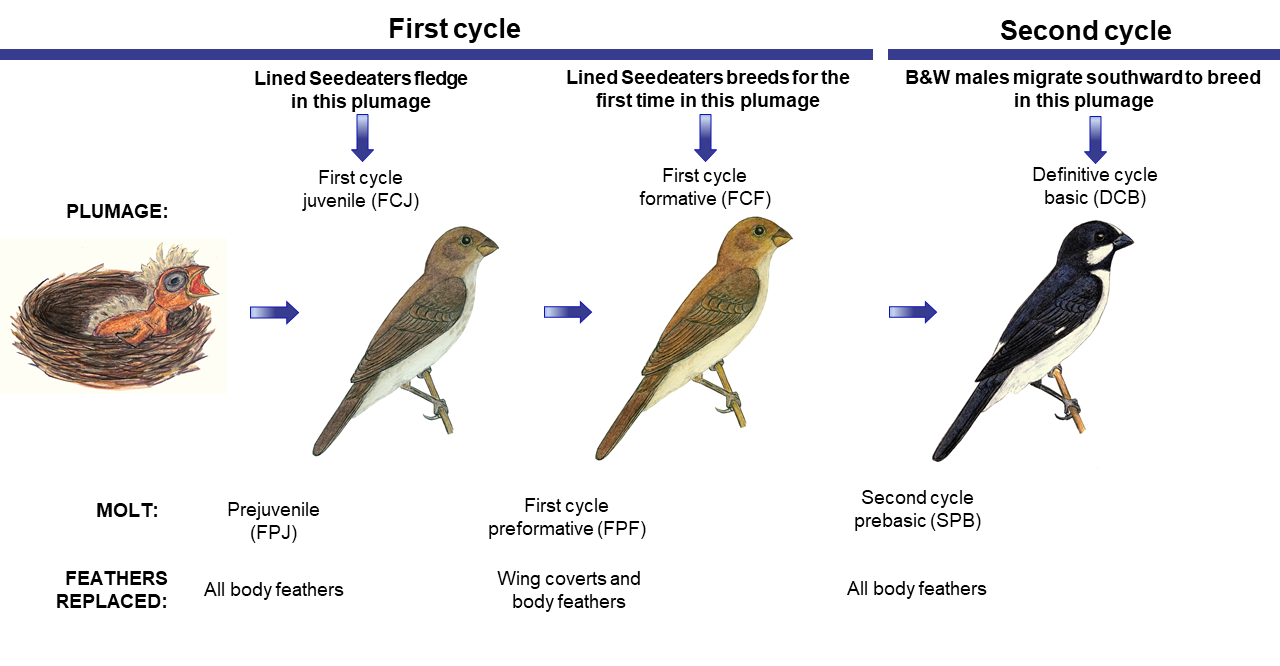
Fig. 1. Hypothesized molt cycle of Lined Seedeaters, a species that exhibits a complex basic strategy of molt. The species exhibit delayed plumage maturation, with males acquiring the definitive plumage during the second prebasic molt. Artwork by L. E. Lopes.

Fig. 2

Fig. 2. FCJ: first cycle juvenile (1 March 2019, sex: undetermined): (A) birds have more loosely textured body feathers and (B) thinner, more tapered primaries and (C) rectrices when compared to subsequent plumages. (D) FPF: first cycle preformative molt (17 March 2020, sex: undetermined): with replaced body feathers, lesser, median, and greater coverts and (E) rectrices in some cases. (F) FCF: first cycle formative (17 March 2020, sex: undetermined): fresh formative body feathers, including lesser, median, and greater coverts, with all retained juvenile rectrices in this case. (G) FPJ: prejuvenile molt (16 January 2018, sex: male): nestling banded when replacing the natal down feathers and (H) recaptured in the subsequent season, when breeding in the FCF: first cycle formative (2 January 2019).

Fig. 3

Fig. 3. DPB: definitive prebasic molt (A) (B) (27 April 2020, sex: male), red arrow indicates the traces of molt on the body feathers and (C) the abraded flight feathers not replaced yet. SPB: second prebasic molt (15 April 2020, sex: male): the red arrows indicate (D) (E) the fresh black-and-white feathers including (F) tertials and rectrices contrasting with worn brownish feathers indicate by white arrow.

Fig. 4

Fig. 4. DCB: definitive basic plumage (A) and (B): female on 5 January 2021; (C) and (D): male on 13 January 2021. Note relatively fresh plumage, without apparent molt limits and light wear at the remiges and truncated rectrices.

Fig. 5

Fig. 5. (A) and (B) FCF: first cycle formative male when first captured (1 March 2018) and (C) and (D) when recaptured (25 February 2019) in the DCB: definitive basic cycle in the subsequent season. Note the abrasion of retained juvenile feathers in FCF as indicated by the red arrows.

Fig. 6

Fig. 6. Male specimen in M-FCF: at minimum in formative plumage (25 October 2009, sex: male, MZUSP 85417) with a (A) higher quality plumage, no apparent molt limits and (B) truncated rectrices.

Fig. 7

Fig. 7. Variation in the brownish plumage observed in captured male Lined Seedeaters. Compare the rectrices shape and feather abrasion across the different brownish male’s age codes. (A) M-FCF: at minimum in formative plumage (6 January 2020) with fresh plumage, truncated rectrices, and absence of molt limits. (B) FCF: first cycle formative (13 January 2018) with worn central rectrices retained from the juvenile plumage indicate by the red arrow.

Fig. 8

Fig. 8. Males captured in the UCB: unknown cycle, basic plumage showing traces of brownish in (A) the body feathers and coverts but (B) absent in flight feathers (14 December 2020). (C) Note the brown-edged body feathers, coverts, (D) and alula (7 December 2017).

Fig. 9

Fig. 9. Possible DCA: definitive cycle alternate in Lined Seedeaters (6 January 2018, sex: female) with fresher lesser covers indicated by the red arrow.









