The following is the established format for referencing this article:
Sharp, A. J., A. Contina, V. Ruiz-Gutiérrez, T. Scott Sillett, E. Bridge, E. Besozzi, J. Muller, J. Kelly, A. Given, and C. S. Rushing. 2023. The strength of migratory connectivity in Painted Buntings is spatial scale dependent and shaped by molting behavior. Journal of Field Ornithology 94(1):7.ABSTRACT
As migratory species move through the stages of their annual cycle, individuals often display variation in the degree to which they remain in proximity to one another, a phenomenon called migratory connectivity. We show scale dependence in the strength of migratory connectivity in Painted Buntings (Passerina ciris), a North American passerine with disjunct eastern and interior breeding populations. Based on light-level geolocator data from 112 individuals at 11 breeding sites, migratory connectivity between breeding and wintering grounds was strong at the range-wide scale, with interior and eastern Painted Buntings remaining separated throughout the annual cycle. Conversely, migratory connectivity within the eastern and interior populations was weak, with individuals from different breeding areas mixing extensively on winter quarters. We found weak migratory connectivity within populations as birds moved from the breeding grounds to the wintering grounds (breeding-to-winter), with individuals from different regions of each population mixing extensively on the wintering grounds. The interior population, however, displayed strong migratory connectivity as birds moved from the breeding grounds to the intermediate molting grounds (breeding-to-molting), with birds from different breeding sites showing contrasting migratory strategies during the molting period. Our results suggest that spatial scale dependence of migratory connectivity is likely to be a pervasive phenomenon, given that migratory routes and the likelihood of molt migration often differ among populations. When possible, researchers should be deliberate about the spatial design of tracking studies to reduce potential biases that can result from spatial scale-dependent migratory connectivity.
INTRODUCTION
Populations of migratory species move annually between areas used during breeding, molting, and wintering (Alerstam and Bäckman 2018). Migratory connectivity describes the extent to which individuals from a given population remain together as they move between phases of the annual cycle (Webster et al. 2002). Migratory connectivity is considered “strong” when individuals that are spatially associated during one period of the annual cycle (e.g., breeding) remain close together during other periods of the annual cycle (e.g., non-breeding), and is further strengthened if interpopulation mixing is low (Finch et al. 2017). In contrast, migratory connectivity is weakened when sympatric individuals in one stationary period of the annual cycle are allopatric in another stationary period and/or mix with individuals from other regions when transitioning from one stationary period to the next. Weak migratory connectivity, where individuals from different breeding populations overlap on the wintering grounds, appears to be common for migratory songbirds (Finch et al. 2017).
Understanding the extent to which breeding populations are connected to specific non-breeding and stopover areas can be critical to the conservation of migratory species, which experience different conditions and risks as they move among breeding and non-breeding areas throughout their annual cycles (Marra et al. 2015, Cooper et al. 2017). Quantifying the linkages among breeding and non-breeding regions is also necessary for inferring connections between demographic trends observed on the breeding ground and conditions on the non-breeding ground (Rushing et al. 2016, Taylor and Stutchbury 2016, Kramer et al. 2018, Hallworth et al. 2021). Despite its importance, migratory connectivity is poorly studied for most species, and when it has been studied, is often derived from sampling that is limited in its spatial scale or coverage of the annual cycle (but see Knight et al. 2018, 2021; Kramer et al. 2018, Skinner et al. 2022). For example, migratory connectivity studies based on band recoveries are frequently based on only two locations per individual: a single location from the breeding ground and a single location from the non-breeding season (Brown and Miller 2016, Moore and Krementz 2017). This approach precludes potentially significant conclusions about migratory connectivity at intermediate stages, such as migration or stopover (Hewson et al. 2016). Additionally, for many migratory species, band recovery rates are extremely low and samples sizes are insufficient for estimating migratory connectivity, even after decades of banding effort. The logistical limitations of tracking technology (expense, difficulty of recovering archival devices) also constrain the spatial scale of many studies, such that the available data represent only a limited portion of the species-wide breeding range (Siegel et al. 2016, Witynski and Bonter 2018, Burgess et al. 2020). Detailed accounts of spatial and temporal variation in migratory connectivity are therefore missing for many species (McKinnon and Love 2018).
One such species is the Painted Bunting (Passerina ciris), a small (~16 g), short- to medium-distance migrant and species of conservation concern (U.S. Fish and Wildlife Service 2008). Painted Buntings exist in two allopatric breeding populations separated by a 500-km gap within which there are only sparse and sporadic breeding records (Fig. 1) (Gilbert et al. 2019). The eastern population breeds within 15 km of the Atlantic Ocean from southern North Carolina to northern Florida and in the Piedmont region of central South Carolina and Georgia. The interior population breeds in Texas east to Mississippi, north into Kansas, and southwest into northern Mexico. The interior population contains approximately 60 times more individuals and occupies an area approximately 25 times larger than that of the eastern population (Sykes and Holzman 2005, Fink et al. 2021). Both populations have declined in abundance since 1970, but the eastern population has declined at a steeper rate (Sauer et al. 2013).
Painted Buntings also show complex variation in molting and migration strategies across their breeding range. Eastern Painted Buntings molt at the end of the breeding season (definitive prebasic molt) before beginning fall migration (Thompson 1991). In contrast, the interior population appears to molt at locations intermediate to their breeding and wintering areas (i.e., stopover molt migration) (Rohwer et al. 2005, Tonra and Reudink 2018). We do not know if all interior Painted Buntings are molt-migrants or if this strategy is undertaken only by individuals that breed in the most arid regions of the breeding range (Bridge et al. 2016, Pageau et al. 2020).
We used tracking data from archival light-level geolocators to test the hypothesis that the complex breeding distribution and variation in molting behavior would affect the strength of migratory connectivity in Painted Buntings at multiple spatial scales and at different stages of the annual cycle. Previous tracking studies have focused either solely on the eastern population (Rushing et al. 2021) or on a single breeding site of the interior population (Contina et al. 2013). Specifically, we estimated (1) range-wide breeding-to-winter migratory connectivity, (2) breeding-to-molt migratory connectivity within the interior population, and (3) breeding-to-winter migratory connectivity within both the eastern and interior populations. We predicted that the geographic distance between the disjunct breeding grounds and the Gulf of Mexico would promote strong range-wide breeding-to-winter migratory connectivity between populations. Given the limited area where interior buntings have been observed molting, we predicted that buntings from the interior population would mix extensively during molt, which would lead to weak breeding-to-molt migratory connectivity. We also predicted that the relatively small winter range of the eastern population (Rushing et al. 2021) would lead to weak breeding-to-winter migratory connectivity (Finch et al. 2017). Finally, we predicted that the comparatively larger winter range of the interior population (Battey et al. 2017) would be associated with stronger breeding-to-winter connectivity than that of the eastern population.
METHODS
Geolocator deployment and data analysis
We deployed light-level geolocators (hereafter “geolocators”) at 11 sites across the Painted Bunting’s breeding range. During the summers of 2017, 2018, and 2019, we deployed 295 geolocators (stalked model P50Z11-7-DIP, Migrate Technology Ltd, Coton, Cambridge, UK) on eastern Painted Buntings at nine sites (Table 1, Fig. 1; see Appendix 1 for detailed site information). In the first two years, we attempted to put tags only on after-second-year (ASY) males, which are easily identifiable by their characteristic colorful plumage (Thompson 1991). In cases where we could not capture enough ASY males, the remaining tags were deployed on second-year (SY) males (n = 12). In 2019, we put approximately half (47/100) of the geolocators on ASY females, and the remainder on ASY males. Despite their lack of bright plumage, females are readily aged via patterns of molt in the primary coverts (Pyle 1997). Birds were captured using mist nets and hanging wire traps. Geolocators were attached with leg-loop harnesses (Rappole and Tipton 1991) made with elastic jewelry cord, and were secured with crimp beads. Within the interior population, we deployed 215 geolocators in Oklahoma during the summers of 2010, 2011, and 2012 as part of a separate study (Table 1) (see Contina et al. 2013 for field methods) and 14 geolocators in Arkansas during summer 2018. The tracking results from a subset (19/28) of our Oklahoma birds were previously used by Contina et al. (2013, 2016) to study genes that may be responsible for migratory behavior, as well the relationship between migration and isotope patterns.
Geolocator analysis
Geolocator data consisted of light levels recorded at predetermined intervals for the duration of the tag’s battery life (eastern: range = 310–398 days, median = 364 days; interior: range = 45–324 days, median = 115 days). We used the R package SGAT (Wotherspoon et al. 2013) to generate location estimates from the raw light data (see Appendix 1 for details on geolocator analysis). Twilights were identified using the function “preprocessLight”, which is part of the R package TwGeos (Wotherspoon et al. 2016). We used SGAT to determine appropriate zenith angles for each bird during the stationary breeding period when individuals were at known locations. For eastern birds, the breeding stationary period was defined as 1 June to 1 August. The wintering stationary period was defined as 1 December to 1 February. For interior birds, the stationary breeding period began when the geolocator was deployed and ended when the individual departed the breeding ground. This flexible time frame was necessary because some interior birds had already reached the molting ground and the recording of locations had stopped before others had received their geolocator on the breeding ground. We defined departure as the first significant (> 2 degrees) and permanent longitudinal movement away from a known stationary location (see Appendix 1 for more details). To determine appropriate zenith angles at times of the year when location was unknown (the nonbreeding season), we used the Hill-Ekstrom calibration method (Lisovski et al. 2012, 2020), which works on the principle that the true zenith angle should result in the smallest variation in estimated latitudes. To reduce uncertainty in the estimated locations, we incorporated a range mask as a prior to disallow stationary locations over water or outside the known range of the Painted Bunting (range map shapefile: Fink et al. 2021). We took a weighted median of the location density estimate surface (generated using the “slice” function within SGAT) to generate a point-estimate location for each individual during the non-breeding stationary period(s). We used the straight-line distance between these estimated locations (breeding to molting to wintering) to generate conservative estimates of migration distance per individual. We used one-way ANOVA to test for differences in wintering locations (latitude and longitude) among birds from different breeding sites. For all ANOVA and t tests, we used α = 0.05 to determine significance.
Estimating the strength of migratory connectivity
We used the R package MigConnectivity (Cohen et al. 2018) to quantify the strength of migratory connectivity between stationary periods: breeding, molting (interior only), and winter. Although tracking data cannot prove the occurrence of molt, we use the term breeding-to-molting connectivity to describe migratory connectivity during the late summer/early autumn period when birds would have completed breeding but have not yet arrived on their final wintering ground. We did not examine breeding-to-molting migratory connectivity in the eastern population because eastern Painted Buntings remain on the breeding ground during this post-breeding period. The “estMC” function estimates the strength of migratory connectivity (MC) from geolocator data while accounting for uncertainty in the estimated locations (Cohen et al. 2018). Values of MC fall between -1 and 1, although real-world scenarios typically result in an MC value that falls between 0 and 1 (Cohen et al. 2018). Negative values indicate a propensity for birds from one region to spread out away from each other and toward birds from other regions as they transition from one stationary period to the next. Values near to 0 indicate that birds from all breeding regions mix uniformly on the non-breeding ground. Values near to 1 indicate that birds from individual breeding regions remain clustered together from one stationary period to the next and remain segregated from birds from other breeding regions.
MigConnectivity defines migratory connectivity at the population level, so that the user must define discrete breeding and non-breeding regions. For this purpose, we binned the wintering grounds into five regions: Mexico, Central America, Florida, Cuba, and the Bahamas. The later three regions were chosen because they are separated from each other by open water and thus form natural distinct regions. We chose Mexico and Central America as wintering regions to divide the western wintering range into approximately northern and southern halves. To quantify breeding-to-molting migratory connectivity within the interior population, we defined two molting regions: northwest Mexico, and a more eastern region that encompasses the area between the interior breeding sites and the Gulf of Mexico. These two regions were chosen to differentiate between two basic strategies during the molting season: (1) using the northwest Mexico monsoon region during the molting season, or (2) using an alternative strategy such as migrating directly to the wintering ground. MigConnectivity also requires the user to bin the breeding range into discrete regions. For the interior population, we used Oklahoma and Arkansas as two discrete breeding regions. In the eastern population, we binned the breeding range into three regions by latitude: a northern region (North Carolina sites), a central region (all South Carolina sites), and a southern region (Georgia and Florida sites). We examined range-wide (inter-population) breeding-to-winter migratory connectivity, breeding-to-molting migratory connectivity within the interior population, and breeding-to-winter migratory connectivity within each population (intra-population).
To improve estimates of migratory connectivity (both breeding-to-molting and breeding-to-winter), we accounted for differences in relative abundance between breeding sites in our estimate of MC (Cohen et al. 2018). Because relative abundance data were not available for our specific field sites, we divided the breeding range into polygons, with relative abundance within each polygon used as the abundance estimate for all breeding sites within that polygon. The eastern population was divided into three roughly equal-sized polygons corresponding to the regions described previously. For Oklahoma and Arkansas breeding sites, relative abundance was extracted from a rectangle measuring one degree latitude by one degree longitude around each site. Estimates of relative abundance in each region were derived from data that are publicly available from eBird (Fink et al. 2020), using the R package “ebirdst” (Auer et al. 2019).
“EstMC” requires a single position (i.e., “centroid”) for each bird during periods of the annual cycle when birds are in an unknown location, such as on the wintering or molting ground. For the wintering centroid, we used the point-estimate generated during the core of the wintering period (December and January). For the molting period (interior population only), the date range used to calculate the centroid varied by individual due to wide variation in when individuals arrived on the molting ground. For interior birds that did not do a prolonged stopover at a distinct molting location, we used a date range that best represented their behavior during the post-breeding molting period, such as moving directly to the wintering ground at the beginning or end of the post-breeding molting period. To estimate uncertainty in location estimations derived from light levels, we calculated the mean difference between known and estimated latitude and longitude during a window of time when it could be safely assumed that birds were still on the breeding ground (see Appendix 1 for details).
RESULTS
We recovered 87 of 295 (29%) geolocators from the eastern population, of which 82 (94%) had viable data (Table 1). Fifty-five of 229 (24%) geolocators were recovered from the interior population; 36 (65%) recorded viable data (34 from Oklahoma, two from Arkansas). We excluded five of the 34 Oklahoma individuals that were involved in an experimental treatment that potentially could have affected their migration (Contina et al. 2013). An additional Oklahoma tag was excluded because a malfunctioning geolocator clock may have resulted in erroneous locations. Of the remaining 28 Oklahoma tags, 20 collected data through the post-breeding molting period but stopped recording data before the bird reached the final winter destination (Table 1). Tags that stopped functioning in late summer while the individual was still well outside the known wintering range for Painted Buntings resulted in this classification. Eight Oklahoma tags and both Arkansas tags recorded data long enough to estimate the wintering location. All 82 viable geolocators from the eastern population operated long enough to allow us to determine wintering locations. Tags were determined to have operated long enough to allow us to identify wintering location if the data showed a distinct movement away from the breeding/molting ground that resulted in an apparent stationary period within the known wintering range of the Painted Bunting. Higher failure rates for the tags deployed on interior birds is attributable to using an early generation of light-level geolocators.
Geolocator data revealed a persistent division between the eastern and interior Painted Bunting populations (Fig. 1). We found no evidence that interior and eastern birds mix during the stationary non-breeding periods, which results in strong range-wide migratory connectivity (MC = 0.71, SE = 0.10) (Table 2). An MC value < 1 despite perfect separation between the two populations indicates intra-population mixing from breeding to winter (Cohen et al. 2018). In contrast, within both the eastern and interior Painted Bunting populations, we found low breeding-to-winter migratory connectivity (eastern population MC = -0.05, SE = 0.04; interior population MC = 0.03, SE = 0.22) (Table 2, Figs. 2 and 3). Individuals from all four eastern states (North Carolina, South Carolina, Georgia, and Florida) occurred in all three major wintering regions (Florida, the Bahamas, and Cuba). Individuals from the northernmost breeding site (North Carolina, n = 8) tended to migrate to the southern portion of the wintering ground, but an ANOVA test revealed no significant (P < 0.05) differences in wintering location (latitude and longitude) among the breeding sites (ANOVA latitude: DF = 7,72, F = 1.95, P = 0.07). However, when grouped by breeding state instead of specific breeding site (all states except for Florida, Oklahoma, and Arkansas had multiple breeding sites), birds from North Carolina did winter farther south than birds from South Carolina or Georgia (ANOVA latitude: DF = 3,76, F = 4.03, P = 0.01). No significant difference in winter latitude was detected between birds from North Carolina and Florida (t test, t = 1.82, DF = 18,47, P = 0.09). Birds from the interior population wintered from central Mexico south to Nicaragua, and there was no significant difference in wintering location between Arkansas and Oklahoma birds (ANOVA latitude: DF = 1,6, F = 0.571, P = 0.479).
Despite weak breeding-to-winter migratory connectivity within populations, breeding-to-molting migratory connectivity within the interior population was strong (MC = 0.81, SE = 0.16) (Table 2), indicating that individuals from Arkansas and Oklahoma remained segregated during the post-breeding molting period. During the post-breeding period, all Oklahoma breeders migrated to northwestern Mexico, a known molting region for western molt-migrants (Pyle et al. 2009). One Arkansas bird stayed within the breeding region during the post-breeding molting period before migrating around the Gulf of Mexico en route to its wintering ground in southern Mexico/northern Central America. The second Arkansas bird migrated directly from its breeding site to overwinter in southern Mexico, making its way south in short hops along the Gulf Coast.
DISCUSSION
We have shown that, as hypothesized, the strength of migratory connectivity can vary dramatically within a single species, depending on the spatial scale of inference and the periods of the annual cycle during which migratory connectivity is measured. As predicted, breeding-to-winter migratory connectivity of Painted Buntings was strong at the range-wide scale, with no evidence that eastern and interior populations mixed on the non-breeding grounds. In partial support of our predictions, breeding-to-winter migratory connectivity within both the eastern and interior populations was weak, with individuals from different breeding sites within each population mixing extensively on wintering grounds. Despite weak breeding-to-winter migratory connectivity, breeding-to-molting migratory connectivity was strong in the interior population, contrary to our prediction and demonstrating the importance of quantifying migratory connectivity at stages intermediate to breeding and winter (Cohen et al. 2019). Our results suggest that differences in molting strategy may drive strong breeding-to-molting migratory connectivity in molt-migrants like the Painted Bunting, but also that this strong migratory connectivity does not necessarily persist into the stationary wintering season. These results align with recent theoretical research that has examined the effect of spatial sampling design on estimates of migratory connectivity (Vickers et al. 2021).
Our results also highlight the need to consider how patterns of migratory connectivity change throughout the annual cycle. Despite weak breeding-to-winter migratory connectivity within the interior population, individuals breeding in Oklahoma and Arkansas diverged in migratory behavior following the breeding season, which resulted in strong migratory connectivity during the molting period. Although our study was not designed to quantify molting behavior per se, banding and stable isotope studies have confirmed that northwest Mexico is an important molting region for multiple molt-migrant species, including Painted Buntings (Rohwer et al. 2005, Pyle et al. 2009, Rohwer 2013). Conversely, our limited results from Arkansas suggest that buntings there molt on their breeding grounds like the eastern population. Our results also suggest that molting behavior had profound implications on migratory distance. On average, individuals from Oklahoma (n = 8) traveled nearly 1500 km farther than Arkansas birds (n = 2) to reach the wintering ground, and more than 2500 km farther than birds from the eastern population. This sizable difference in migratory distance provides support for the hypothesis that morphological differences observed in Painted Buntings (increasing wing length moving from east to west) are selected for and maintained by a divergence in migratory behavior during the non-breeding period (Battey et al. 2017). Despite differences in molting behavior, birds from Oklahoma and Arkansas do not appear to occupy different regions of the wintering range.
Contrasting molting behavior between Oklahoma and Arkansas birds could reflect differences in both midsummer climate at these locations and genetics. Painted Buntings in the western portion of the breeding range experience an increasingly arid landscape by midsummer, whereas the more eastern breeding regions remain green and productive later into the summer and fall. Thus, molt-migration to the monsoonal regions of western Mexico is likely adaptive for interior Painted Buntings breeding in more arid regions. The westernmost breeding buntings may also be following ancestral routes of range expansion of the Passerina clade (Ruegg and Smith 2002, Shipley et al. 2013). Moreover, two genetically distinct groups exist within the interior population and one within the eastern population (Herr et al. 2011, Battey et al. 2017, Contina et al. 2019a, b). Thorough sampling across the breeding range, particularly in Arkansas (n = 2 individuals in this study), Louisiana, and Mississippi will improve our understanding of how climate and genetics interact to affect molt behavior and susceptibility of Painted Buntings to ongoing environmental change (Sykes et al. 2007, Contina et al. 2016, 2019a; Battey et al. 2017).
The scale-dependent strength of migratory connectivity in Painted Buntings has broader conservation implications. Because a large portion of the interior population migrates to a relatively small area to molt, efforts to conserve natural habitat in the monsoon region of northwest Mexico could have an outsized benefit to the species. Similarly, habitat destruction and other negative factors that occur in this region (e.g., illegal trapping) could be particularly detrimental, and could be a potential culprit if interior bunting populations begin to decline more rapidly. Weak breeding-to-winter migratory connectivity could benefit the eastern population, which occurs in an area that is 25 times smaller than that of the interior population, may be declining faster (Sauer et al. 2013), and faces intense pressure from development on both breeding and wintering areas (Napton et al. 2010, Jones et al. 2013). Weak migratory connectivity can help mediate the effect of habitat loss in one portion of the range, thereby promoting population resilience, at least in the near-term (Finch et al. 2017, Rushing et al. 2021). Strong range-wide migratory connectivity in Painted Buntings suggests that interior and eastern populations should be managed separately. Without immigration from the much larger interior population, eastern Painted Bunting populations do not appear to benefit from any type of “rescue effect” and are thus completely reliant on this increasingly fragmented coastal habitat.
In conclusion, our results emphasize the importance of considering the scale dependence of migratory connectivity. Although the Painted Bunting’s allopatric breeding populations and intraspecific variation in molting strategies appear to be unusual among North American migratory songbirds, the existence of population-specific migratory routes (Delmore et al. 2012, Stanley et al. 2015, Kramer et al. 2017) and well-known gradients in molt migration (Voelker and Rohwer 1998, Pageau et al. 2020) implies that scale-dependent migratory connectivity is a pervasive phenomenon. When possible, researchers should be deliberate about the spatial design of tracking studies to reduce potential biases that can result from scale-dependent migratory connectivity (Vickers et al. 2021).
RESPONSES TO THIS ARTICLE
Responses to this article are invited. If accepted for publication, your response will be hyperlinked to the article. To submit a response, follow this link. To read responses already accepted, follow this link.
AUTHOR CONTRIBUTIONS
AJS, CSR, VRG, AG, and TSS designed the study for the eastern population. ESB, JK, EB, and JM designed the study for the interior population. AJS analyzed movement data and drafted the manuscript. All authors contributed to subsequent revisions of the manuscript.
ACKNOWLEDGMENTS
We thank the following groups for their assistance in obtaining movement data: Airlie Gardens, Audubon South Carolina, Bald Head Island Conservancy, Carolina Beach State Park, the Town of Kiawah Island, and Little Talbot Island State Park. We thank Chris Sayers, Patricia Rodrigues, Max Witynski, Michelle Harris, and Anna Kennedy for their hard work in the field. Additional thanks are due to Chris Marsh, Charlie Winterbauer, Scott Coleman, Beth Darrow, and Kathleen Kelso for arranging access to field sites. Finally, we would like to thank Wesley Hochachka and two anonymous reviewers for their valuable editorial feedback on the manuscript.
DATA AVAILABILITY
Raw light levels, annotated twilights, daily location estimates, and relevant metadata are available online at Movebank.org (Range-wide Migratory Connectivity of Painted Buntings (Geolocator) https://www.movebank.org/cms/webapp?gwt_fragment=page%3Dstudies%2Cpath%3Dstudy1990489346%2Bfile
LITERATURE CITED
Alerstam, T., and J. Bäckman. 2018. Ecology of animal migration. Current Biology 28:R968-R972. https://doi.org/10.1016/j.cub.2018.04.043
Auer, T., D. Fink, and M. Strimas-Mackey. 2019. ebirdst: tools for loading, plotting, mapping and analysis of eBird status and trends data products.
Battey, C. J., E. B. Linck, K. L. Epperly, C. French, D. L. Slager, P. W. Sykes, and J. Klicka. 2017. A migratory divide in the Painted Bunting (Passerina ciris). American Naturalist 191:259-268. https://doi.org/10.1086/695439
Bridge, E. S., J. D. Ross, A. J. Contina, and J. F. Kelly. 2016. Do molt-migrant songbirds optimize migration routes based on primary productivity? Behavioral Ecology 27:784-792. https://doi.org/10.1093/beheco/arv199
Brown, D., and G. Miller. 2016. Band recoveries reveal alternative migration strategies in American Robins. Animal Migration 3:35-47. https://doi.org/10.1515/ami-2016-0004
Burgess, M. D., T. Finch, J. A. Border, J. Castello, G. Conway, M. Ketcher, et al. 2020. Weak migratory connectivity, loop migration and multiple non-breeding site use in British breeding Whinchats Saxicola rubetra. Ibis 162:1292-1302. https://doi.org/10.1111/ibi.12825
Cohen, E. B., J. A. Hostetler, M. T. Hallworth, C. S. Rushing, T. S. Sillett, and P. P. Marra. 2018. Quantifying the strength of migratory connectivity. Methods in Ecology and Evolution 9:513-524. https://doi.org/10.1111/2041-210X.12916
Cohen, E. B., C. R. Rushing, F. R. Moore, M. T. Hallworth, J. A. Hostetler, M. G. Ramirez, and P. P. Marra. 2019. The strength of migratory connectivity for birds en route to breeding through the Gulf of Mexico. Ecography 42:658-669. https://doi.org/10.1111/ecog.03974
Contina, A., J. L. Alcantara, E. S. Bridge, J. D. Ross, W. F. Oakley, J. F. Kelly, and K. C. Ruegg. 2019a. Genetic structure of the Painted Bunting and its implications for conservation of migratory populations. Ibis 161:372-386. https://doi.org/10.1111/ibi.12641
Contina, A., R. A. Bay, V. Le Underwood, T. B. Smith, J. F. Kelly, E. S. Bridge, and K. C. Ruegg. 2019b. Characterization of SNP markers for the Painted Bunting (Passerina ciris) and their relevance in population differentiation and genome evolution studies. Conservation Genetics Resources 11:5-10. https://doi.org/10.1007/s12686-017-0950-7
Contina, A., E. S. Bridge, and J. F. Kelly. 2016. Exploring novel candidate genes from the Mouse Genome Informatics database: potential implications for avian migration research. Integrative Zoology 11:240-249. https://doi.org/10.1111/1749-4877.12199
Contina, A., E. S. Bridge, N. E. Seavy, J. M. Duckles, and J. F. Kelly. 2013. Using geologgers to investigate bimodal isotope patterns in Painted Buntings (Passerina ciris). Auk 130:265-272. https://doi.org/10.1525/auk.2013.13003
Cooper, N. W., M. T. Hallworth, and P. P. Marra. 2017. Light-level geolocation reveals wintering distribution, migration routes, and primary stopover locations of an endangered long-distance migratory songbird. Journal of Avian Biology 48:209-219. https://doi.org/10.1111/jav.01096
Delmore, K. E., J. W. Fox, and D. E. Irwin. 2012. Dramatic intraspecific differences in migratory routes, stopover sites and wintering areas, revealed using light-level geolocators. Proceedings of the Royal Society B: Biological Sciences 279:4582-4589. https://doi.org/10.1098/rspb.2012.1229
Finch, T., S. J. Butler, A. M. A. Franco, and W. Cresswell. 2017. Low migratory connectivity is common in long-distance migrant birds. Journal of Animal Ecology 86:662-673. https://doi.org/10.1111/1365-2656.12635
Fink, D., T. Auer, A. Johnston, V. Ruiz-Gutierrez, W. M. Hochachka, and S. Kelling. 2020. Modeling avian full annual cycle distribution and population trends with citizen science data. Ecological Applications 30:e02056. https://doi.org/10.1002/eap.2056
Fink, D., T. Auer, A. Johnston, M. Strimas-Mackey, O. Robinson, S. Ligocki, et al. 2021. eBird status and trends. Cornell Lab of Ornithology, Ithaca, New York. https://doi.org/10.2173/ebirdst.2021
Gilbert, N. A., H. E. Horne, and J. A. Trent. 2019. Mind the gap: the Painted Bunting (Passerina ciris) breeds in central Alabama and eastern Mississippi. Wilson Journal of Ornithology 131:147-152. https://doi.org/10.1676/18-61
Hallworth, M. T., E. Bayne, E. McKinnon, O. Love, J. A. Tremblay, B. Drolet, et al. 2021. Habitat loss on the breeding grounds is a major contributor to population declines in a long-distance migratory songbird. Proceedings of the Royal Society B: Biological Sciences 288:20203164. https://doi.org/10.1098/rspb.2020.3164
Herr, C. A., P. W. Sykes, and J. Klicka. 2011. Phylogeography of a vanishing North American songbird: the Painted Bunting (Passerina ciris). Conservation Genetics 12:1395-1410. https://doi.org/10.1007/s10592-011-0237-6
Hewson, C. M., K. Thorup, J. W. Pearce-Higgins, and P. W. Atkinson. 2016. Population decline is linked to migration route in the Common Cuckoo. Nature Communications 7:12296. https://doi.org/10.1038/ncomms12296
Jones, G., A. Snider, and S. Luo. 2013. Changes in the extent of North Carolina Barrier Island Maritime Forests 1988–2011: an assessment of past efforts at protection. Journal of Forestry 111:186-193. https://doi.org/10.5849/jof.12-022
Knight, E. C., A.-L. Harrison, A. L. Scarpignato, S. L. Van Wilgenburg, E. M. Bayne, J. W. Ng, et al. 2021. Comprehensive estimation of spatial and temporal migratory connectivity across the annual cycle to direct conservation efforts. Ecography 44:665-679. https://doi.org/10.1111/ecog.05111
Knight, S. M., D. W. Bradley, R. G. Clark, E. A. Gow, M. Bélisle, L. L. Berzins, et al. 2018. Constructing and evaluating a continent-wide migratory songbird network across the annual cycle. Ecological Monographs 88:445-460. https://doi.org/10.1002/ecm.1298
Kramer, G. R., D. E. Andersen, D. A. Buehler, P. B. Wood, S. M. Peterson, J. A. Lehman, et al. 2018. Population trends in (Vermivora warblers are linked to strong migratory connectivity. Proceedings of the National Academy of Sciences 115:E3192-E3200. https://doi.org/10.1073/pnas.1718985115
Kramer, G. R., H. M. Streby, S. M. Peterson, J. A. Lehman, D. A. Buehler, P. B. Wood, et al. 2017. Nonbreeding isolation and population-specific migration patterns among three populations of Golden-winged Warblers. Condor 119:108-121. https://doi.org/10.1650/CONDOR-16-143.1
Lisovski, S., S. Bauer, M. Briedis, S. C. Davidson, K. L. Dhanjal-Adams, M. T. Hallworth, et al. 2020. Light-level geolocator analyses: a user’s guide. Journal of Animal Ecology 89:221-236. https://doi.org/10.1111/1365-2656.13036
Lisovski, S., C. M. Hewson, R. H. G. Klaassen, F. Korner-Nievergelt, M. W. Kristensen, and S. Hahn. 2012. Geolocation by light: accuracy and precision affected by environmental factors. Methods in Ecology and Evolution 3:603-612. https://doi.org/10.1111/j.2041-210X.2012.00185.x
Marra, P. P., E. B. Cohen, S. R. Loss, J. E. Rutter, and C. M. Tonra. 2015. A call for full annual cycle research in animal ecology. Biology Letters 11:20150552. https://doi.org/10.1098/rsbl.2015.0552
McKinnon, E. A., and O. P. Love. 2018. Ten years tracking the migrations of small landbirds: lessons learned in the golden age of bio-logging. Auk 135:834-856. https://doi.org/10.1642/AUK-17-202.1
Moore, J. D., and D. G. Krementz. 2017. Migratory connectivity of American Woodcock using band return data. Journal of Wildlife Management 81:1063-1072. https://doi.org/10.1002/jwmg.21269
Napton, D. E., R. F. Auch, R. Headley, and J. Taylor. 2010. Land changes and their driving forces in the southeastern United States. Regional Environmental Change 10:37-53. https://doi.org/10.1007/s10113-009-0084-x
Pageau, C., C. M. Tonra, M. Shaikh, N. J. Flood, and M. W. Reudink. 2020. Evolution of moult-migration is directly linked to aridity of the breeding grounds in North American passerines. Biology Letters 16:20200155. https://doi.org/10.1098/rsbl.2020.0155
Pyle, P. 1997. Identification guide to North American birds. Part I: Columbidae to Ploceidae. Slate Creek Press, Point Reyes Station, California, USA.
Pyle, P., W. A. Leitner, L. Lozano-Angulo, F. Avilez-Teran, H. Swanson, E. G. Limón, and M. K. Chambers. 2009. Temporal, spatial, and annual variation in the occurrence of molt-migrant passerines in the Mexican monsoon region. Condor 111:583-590. https://doi.org/10.1525/cond.2009.090085
Rappole, J. H., and A. R. Tipton. 1991. New harness design for attachment of radio transmitters to small passerines (Nuevo Diseño de Arnés para Atar Transmisores a Passeriformes Pequeños). Journal of Field Ornithology 62:335-337.
Rohwer, S. 2013. Molt intensity and conservation of a molt migrant (Passerina ciris) in northwest Mexico: Intensidad de la Muda en un Migrante (Passerina ciris) y su Conservación en el Noroeste de México. Condor 115:421-433. https://doi.org/10.1525/cond.2013.120090
Rohwer, S., L. K. Butler, D. R. Froehlich, R. Greenberg, and P. P. Mara. 2005. Ecology and demography of east–west differences in molt scheduling of neotropical migrant passerines. Pages 87-105 in R. Greenberg and P. P. Marra, editors. Birds of two worlds: the ecology and evolution of migration. Johns Hopkins University Press, Baltimore, Maryland, USA.
Ruegg, K. C., and T. B. Smith. 2002. Not as the crow flies: a historical explanation for circuitous migration in Swainson’s Thrush (Catharus ustulatus). Proceedings of the Royal Society of London. Series B: Biological Sciences 269:1375-1381. https://doi.org/10.1098/rspb.2002.2032
Rushing, C.S., T. B. Ryder, and P. P. Marra. 2016. Quantifying drivers of population dynamics for a migratory bird throughout the annual cycle. Proceedings of the Royal Society B: Biological Sciences 283:20152846. https://doi.org/10.1098/rspb.2015.2846
Rushing, C. S., A. M. Van Tatenhove, A. Sharp, V. Ruiz-Gutierrez, M. C. Freeman, P. W. Sykes, Jr., et al. 2021. Integrating tracking and resight data enables unbiased inferences about migratory connectivity and winter range survival from archival tags. Ornithological Applications 123:1-14. https://doi.org/10.1093/ornithapp/duab010
Sauer, J. R., W. A. Link, J. E. Fallon, K. L. Pardieck, and D. J. Ziolkowski. 2013. The North American Breeding Bird Survey 1966–2011: summary analysis and species accounts. North American Fauna 79:1-32. https://doi.org/10.3996/nafa.79.0001
Shipley, J. R., A. Contina, N. Batbayar, E. S. Bridge, A. T. Peterson, and J. F. Kelly. 2013. Niche conservatism and disjunct populations: a case study with Painted Buntings (Passerina ciris). Auk 130:476-486. https://doi.org/10.1525/auk.2013.12151
Siegel, R. B., R. Taylor, J. F. Saracco, L. Helton, and S. Stock. 2016. GPS-tracking reveals non-breeding locations and apparent molt migration of a Black-headed Grosbeak. Journal of Field Ornithology 87:196-203. https://doi.org/10.1111/jofo.12149
Skinner, A. A., M. P. Ward, I. Souza-Cole, J. R. Wright, F. R. Thompson III, T. J. Benson, et al. 2022. High spatiotemporal overlap in the non-breeding season despite geographically dispersed breeding locations in the Eastern Whip-poor-will (Antrostomus vociferus). Diversity and Distributions 28:712-726. https://doi.org/10.1111/ddi.13477
Stanley, C. Q., E. A. McKinnon, K. C. Fraser, M. P. Macpherson, G. Casbourn, L. Friesen, et al. 2015. Connectivity of Wood Thrush breeding, wintering, and migration sites based on range-wide tracking. Conservation Biology 29:164-174. https://doi.org/10.1111/cobi.12352
Sykes, P. W., Jr., and S. Holzman. 2005. Current range of the eastern population of Painted Bunting (Passerina ciris). Part 1: breeding. North American Birds 59:4-17.
Sykes, P. W., Jr., S. Holzman, and E. E. Iñigo-Elias. 2007. Current range of the eastern population of Painted Bunting (Passerina ciris). Part II: winter range. North American Birds 61(3):378-406.
Taylor, C. M., and B. J. M. Stutchbury. 2016. Effects of breeding versus winter habitat loss and fragmentation on the population dynamics of a migratory songbird. Ecological Applications 26:424-437. https://doi.org/10.1890/14-1410
Thompson, C. W. 1991. The sequence of molts and plumages in Painted Buntings and implications for theories of delayed plumage maturation. Condor 93:209-235. https://doi.org/10.2307/1368938
Tonra, C. M., and M. W. Reudink. 2018. Expanding the traditional definition of molt-migration. Auk 135:1123-1132. https://doi.org/10.1642/AUK-17-187.1
United States Fish and Wildlife Service. 2008. Birds of conservation concern. United States Department of the Interior, Fish and Wildlife Service, Division of Migratory Bird Management, Arlington, Texas, USA.
Vickers, S. H., A. M. A. Franco, and J. J. Gilroy. 2021. Sensitivity of migratory connectivity estimates to spatial sampling design. Movement Ecology 9:16. https://doi.org/10.1186/s40462-021-00254-w
Voelker, G., and S. Rohwer. 1998. Contrasts in scheduling of molt and migration in eastern and western Warbling Vireos. Auk 115:142-155. https://doi.org/10.2307/4089119
Webster, M. S., P. P. Marra, S. M. Haig, S. Bensch, and R. T. Holmes. 2002. Links between worlds: unraveling migratory connectivity. Trends in Ecology & Evolution 17:76-83. https://doi.org/10.1016/S0169-5347(01)02380-1
Witynski, M. L., and D. N. Bonter. 2018. Crosswise migration by Yellow Warblers, Nearctic-Neotropical passerine migrants. Journal of Field Ornithology 89:37-46. https://doi.org/10.1111/jofo.12237
Wotherspoon, S. J., M. D. Sumner, and S. Lisovski. 2013. R package SGAT: solar/satellite geolocation for animal tracking. GitHub repository.
Wotherspoon, S. J., M. D. Sumner, and S. Lisovski. 2016. TwGeos: basic data processing for light-level geolocation archival tags.
Fig. 1
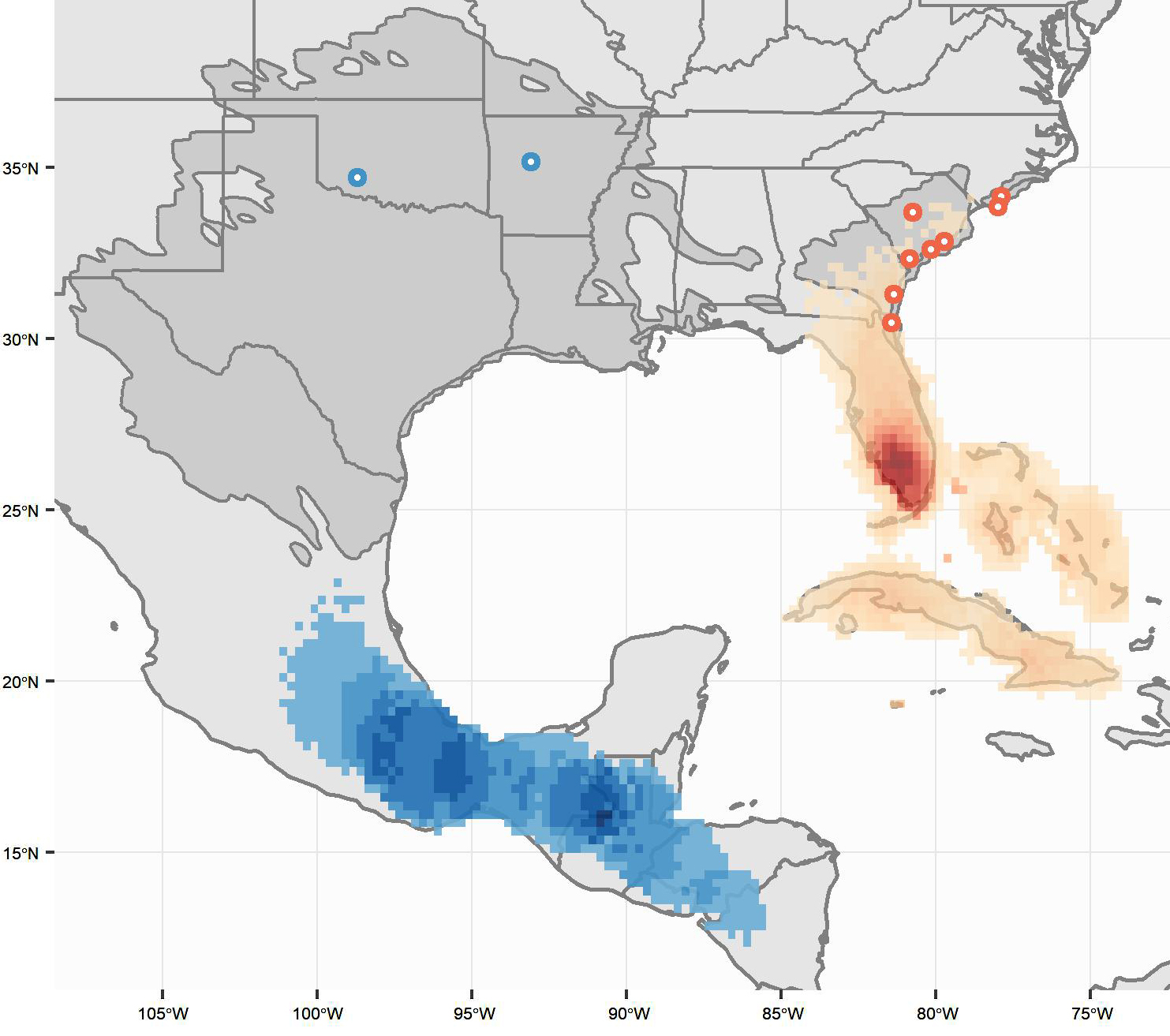
Fig. 1. Deployment locations (circles) and estimated wintering locations for eastern (red, n = 82) and interior (blue, n = 10) Painted Buntings. The raster surfaces represent the overlap of each individual’s 95% density surface. The intensity of the color ramp increases with the number of overlapping density surfaces in that area (interior maximum value = 5, eastern maximum value = 30). The breeding range is shaded dark gray.

Fig. 2
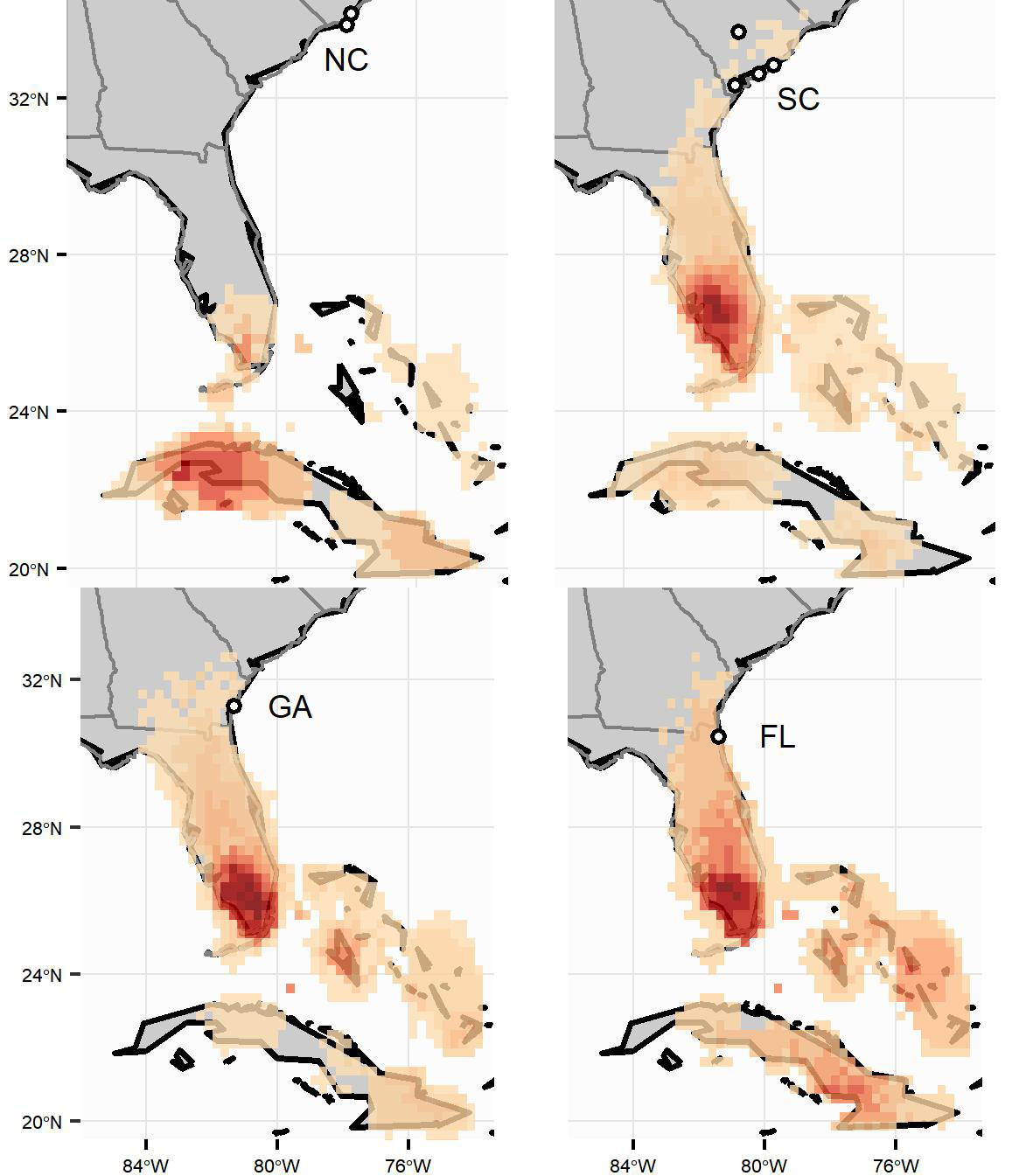
Fig. 2. Estimated wintering locations for eastern Painted Buntings breeding in North Carolina (NC) (n = 8), South Carolina (SC) (n = 32), Florida (FL) (n = 19), and Georgia (GA) (n = 23). The raster surfaces represent the overlap of each individual’s 95% density surface. The intensity of the color ramp increases with the number of overlapping density surfaces in that area (maximum values: NC = 5, SC = 19, GA = 13, FL = 8).

Fig. 3
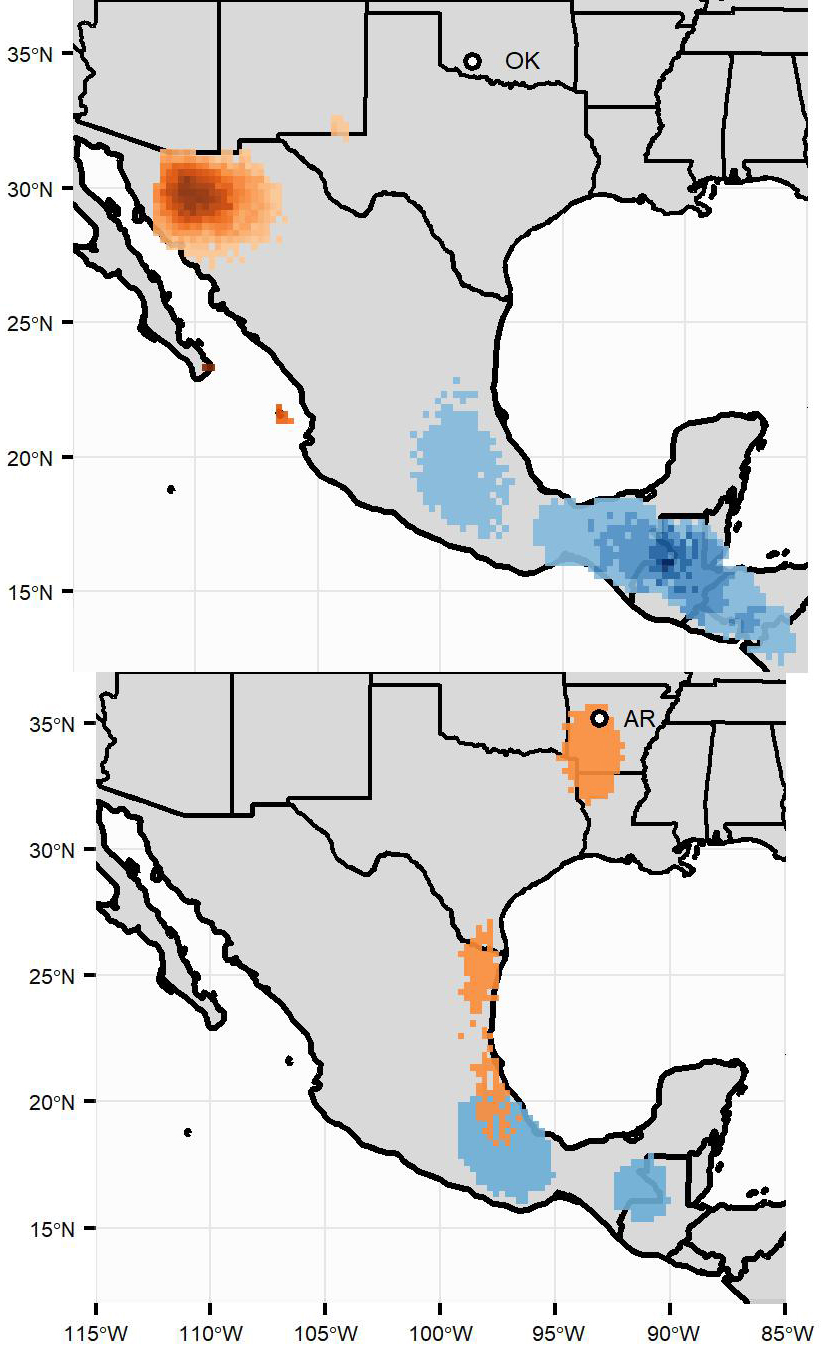
Fig. 3. Estimated locations during the post-breeding molting season (orange) and wintering season (blue) for Oklahoma (OK) (top panel, molting n = 28, winter n = 8) and Arkansas (AR) (bottom panel, n = 2) Painted Buntings. The raster surfaces represent the overlap of each individual’s 95% density surface. The intensity of the color ramp increases with the number of overlapping density surfaces in that area (OK maximum values: molting season = 24, wintering season = 4; AR maximum value = 1).

Table 1
Table 1. Geolocator deployment/recovery statistics. Geolocators were excluded from the analysis if they failed to record viable data, malfunctioned, or were included in an experimental treatment.
| Population | State | Deployed | Recovered (viable data) |
| Eastern | North Carolina | 47 | 9 (8) |
| Eastern | South Carolina | 116 | 34 (32) |
| Eastern | Georgia | 67 | 23 (23) |
| Eastern | Florida | 65 | 20 (19) |
| Interior | Oklahoma | 215 | 53 (28) |
| Interior | Arkansas | 14 | 2 (2) |
| Total | 524 | 141 (112) | |
Table 2
Table 2. Estimates of migratory connectivity (MC). Migratory connectivity can range from -1 to 1. Negative values indicate a propensity for birds from one region to spread out away from each other as they transition from one stationary period to the next, values near to 0 indicate that birds from all breeding regions mix uniformly during stationary non-breeding periods, and values near to 1 indicate that birds from individual breeding regions remain clustered together from one stationary period to the next and remain segregated from birds from other breeding regions.
| Population | MC estimate | Standard error |
| Range-wide (breeding to winter) | 0.71 | 0.10 |
| Eastern sites (breeding to winter) | -0.05 | 0.04 |
| Interior sites (breeding to winter) | 0.03 | 0.22 |
| Interior sites (breeding to molting) | 0.80 | 0.16 |








