The following is the established format for referencing this article:
Leys, R. A. C., and L. A. Grieves. 2023. Sex determination of Eastern White-crowned Sparrows (Zonotrichia leucophrys leucophrys) using wing chord length. Journal of Field Ornithology 94(3):2.ABSTRACT
Determining the sex of individuals in sexually monomorphic bird species outside of the breeding season is difficult. However, many monochromatic species exhibit sex differences in morphometrics, including wing chord length. For example, recent studies found that the Puget Sound (pugetensis) and Gambel’s (gambelii) subspecies of White-crowned Sparrow (Zonotrichia leucophrys) can be sexed reliably using wing chord length. However, the Eastern subspecies (leucophrys) has not been evaluated. We measured the wing chord length of Eastern White-crowned Sparrows migrating through the Long Point Bird Observatory and specimens from the Fatal Light Awareness Program to determine if this subspecies can be reliably sexed using wing chord length. We combined wing chord length measurements with molecular sexing to test the prediction that males have a significantly longer wing chord length than females. Unfortunately, we only captured one definitive-cycle female and were thus unable to test our prediction in definitive-cycle birds. Males in first-cycle plumage had significantly longer wing chord lengths than females in first-cycle plumage and, outside of an overlapping range (74–76 mm), this subspecies can be reliably sexed using wing chord length. Our results support the use of wing chord length as a tool for bird banders and researchers to determine the sex of Eastern White-crowned Sparrows in first-cycle plumage in the hand outside of their breeding season or in the absence of a cloacal protuberance or brood patch.
RESUMEN
La determinación del sexo en individuos de especies de aves sexualmente monomórficas fuera de su temporada de reproducción es difícil. Sin embargo, muchas especies monocromáticas muestran diferencias entre sexos en morfometría, incluyendo la longitud de la cuerda alar. Por ejemplo, estudios recientes encontraron que las subespecies pugetensis y gambelii de Zonotrichia leucphrys pueden ser sexadas confiablemente usando la longitud de la cuerda alar. Sin embargo, la subespecie leucophrys no ha sido evaluada. Medimos la cuerda alar de individuos de Zonotrichia leucophrys leucophrys que migran a través del observatorio de aves de Long Point y especímenes del programa Fatal Light Awareness, para determinar si esta subespecie puede ser sexada confiablemente usando la longitud de la cuerda alar. Combinamos mediciones de la longitud de la cuerda alar con sexado molecular para poner a prueba la predicción de que la longitud de la cuerda alar en los machos es significativamente más larga que en las hembras. Desafortunadamente, solo capturamos una hembra en ciclo definitivo y por lo tanto no pudimos poner a prueba nuestra predicción en aves en ciclo definitivo. Los machos en el plumaje del primer ciclo tuvieron una longitud de la cuerda alar significativamente más larga que las hembras en plumaje de primer ciclo y, fuera del rango de sobreposición (74-76 mm), esta especie puede ser sexada confiablemente utilizando la longitud de la cuerda alar. Nuestros resultados dan soporte al uso de la longitud en la cuerda alar como una herramienta para los anilladores de aves e investigadores para determinar el sexo de Zonotrichia leucophrys leucophrys en plumaje de primer ciclo, en la mano, fuera de su temporada reproductiva o en la ausencia de protuberancia cloacal o parche de incubación.
INTRODUCTION
Sex is an important factor to consider in biological studies. In species with sexually dimorphic plumage (e.g., dichromatism), sex is easily determined by an individual’s plumage, either throughout the annual cycle or when in alternate (breeding) plumage. However, in sexually monomorphic species (species with no morphological sex differences), which represent > 50% of extant bird species (Vučićević et al. 2016), it is difficult or even impossible to determine the sex of individuals, even when birds are in the hand, without taking genetic samples. This situation is a challenge that many ornithologists face when conducting ecological studies that rely on accurately determining the sex of individuals to evaluate sex-specific differences in populations or other features of interest. For example, sex differences may be important considerations for studies of molt (Morton 1992a), physiology (Moore et al. 1982, Morton 1992b), protandry (Seewagen et al. 2013, Schmaljohann et al. 2016), sex ratios (King et al. 1965), social behaviors (Jeans 2022), and stress (Romero et al. 1997, Landys et al. 2004). Many species with monochromatic plumage exhibit sex differences in morphometrics, including wing chord length, but these data are not available for all species (Pyle 1997).
The most common method of sex determination in monochromatic bird species is by examining the individual for the presence of sex characteristics during the breeding season, typically either a cloacal protuberance (male) or a brood patch (female; Pyle 1997). However, outside of the breeding season, individuals do not exhibit these characteristics, and therefore, researchers cannot reliably determine their sex during the pre-migratory, migratory, and overwintering periods. When breeding characteristics are not present, the most common method of sex determination is using polymerase chain reaction (PCR) to amplify two conserved chromo-helicase-DNA-binding (CHD) genes that allow for DNA-based sex determination (Griffiths et al. 1996, 1998, Vučićević et al. 2016).
These procedures require taking biological samples, typically blood or feathers, from each individual being studied. Such procedures are minimally invasive but increase bird handling time and require researchers to undergo additional training and permitting. Furthermore, PCR is a somewhat time-consuming and expensive method of sex determination and, most importantly, does not allow researchers to sex individuals immediately in the field. The ability to determine the sex of individuals rapidly in the field is important for many research questions and study designs. Thus, although PCR is an excellent tool for sexing birds, it is not feasible for all researchers or study systems. This situation is an ongoing issue for many bird species because researchers may be unable to balance the sexes during sampling or otherwise examine sex-specific effects when studying wild, monochromatic species, especially in remote areas or in other situations where laboratory facilities are unavailable and DNA-based sex determination methods are not feasible. Therefore, developing tools that allow researchers to determine sex rapidly and accurately in the field is important from both research and accessibility standpoints.
Many bird species exhibit sex differences in wing chord length (Pyle 1997). Compared to DNA-based sexing methods, using wing chord length is a rapid, repeatable, and non-invasive method of sex determination. When standard protocols are followed, wing chord length measurements are highly repeatable, especially compared to other morphometric measurements such as tail length, tarsus length, and bill length (Gill and Vonhof 2006, Warkentin et al. 2016, Carrillo-Ortiz et al. 2021). Accordingly, wing chord length is a standard morphometric measurement taken at most bird banding stations worldwide (Busse and Meissner 2015, Morris et al. 2016, Zhu et al. 2023) and is commonly used by researchers in other contexts. Thus, using wing chord length to determine sex does not increase bird handling time or stress. Reducing handling time is particularly important in cold climates, where handling small species can result in significant body temperature reductions, both during handling and after release (Andreasson et al. 2020). Therefore, when possible, researchers should use wing chord length for sex determination over DNA-based methods, especially for sensitive species or species handled during inclement weather or otherwise challenging climatic conditions.
The White-crowned Sparrow (Zonotrichia leucophrys) is sexually monochromatic and has four currently recognized subspecies: Eastern (leucophrys), Gambel’s (gambelii), Nuttall’s (nuttalli), and Puget Sound (pugetensis; Banks 1964). The White-crowned Sparrow is one of the most commonly studied passerine species in North America (Rattenborg et al. 2004, Jones et al. 2008, Lipshutz et al. 2017), so identifying morphometric methods to reliably determine the sex of individuals in the hand would be particularly beneficial for researchers. Previous studies have shown that Puget Sound (pugetensis) and Gambel’s (gambelii) subspecies can be reliably sexed using wing chord length (Fugle and Rothstein 1985, Bell 2020). The Eastern subspecies is the dominant subspecies in eastern North America and can be easily distinguished from the Gambel’s subspecies, which occurs rarely in eastern North America, by the presence of black in the lores (Fig. 1). The literature suggests that the Eastern subspecies cannot be reliably sexed using wing chord length; however, after the recent findings by Bell (2020) and personal observations in the field (R. A. C. Leys), we decided to test whether wing chord length could be used to sex the Eastern subspecies. If wing chord length is a reliable measure of sex in Eastern White-crowned Sparrows, wing chord length can be used by researchers and bird banding stations to reliably sex individuals in the field outside of their breeding season.
We hypothesized that wing chord length varies between male and female Eastern White-crowned Sparrows. Based on wing chord measurements made by R. A. C. Leys over two years of migration monitoring at Long Point Bird Observatory (LPBO), we predicted that males would have a longer mean wing chord length than females, females would have wing chord lengths ≤ 75 mm, males would have wing chord lengths ≥ 78 mm, and individuals with wing chord lengths of 76–77 mm would not be reliably sexed by wing chord length. We predicted these values because R. A. C. Leys noticed that there appeared to be a bimodal distribution of wing chord lengths among the individuals captured at Long Point Bird Observatory, with fewer individuals having wing chord lengths in the 76–77 mm range.
METHODS
Data collection
In small passerines, using blood or feathers for DNA sex determination can be equally effective (Harvey et al. 2006), so we collected blood or feather samples from two groups of Eastern White-crowned Sparrows. The first group (N = 27) consisted of live individuals captured at LPBO’s Old Cut Field Station (42°34’58.5” N, 80°23’54.4” W) near Port Rowan, Ontario, Canada. We captured individuals using mist nets during spring migration (05–20 May 2021; 25 April–17 May 2022) as part of the station’s standard, long-term migration monitoring efforts at this location. Each bird was banded, aged, weighed to the nearest 0.1 g, and its wing chord length was measured to the nearest 1 mm. We used an unflattened wing chord length measurement, which is measured from the carpal joint to the tip of the longest primary feather (Pyle 1997, see pages 4–6 for details). We then took either a small (approximately 20 μL) blood sample collected via brachial venipuncture (N = 13 in 2021) or one of the outer rectrices (R6, N = 14 in 2022) from each bird. Blood was blotted onto high wet-strength filter paper saturated with 0.5 M Na-EDTA (pH 8.0) and allowed to air-dry. The second group (N = 11) consisted of deceased birds sampled from the Fatal Light Awareness Program collected during spring (April–May 2021) and fall (August–November 2021) in Toronto, Ontario, Canada. We recorded the age, wing chord length to the nearest 1 mm, and collection date, and removed both R6 feathers from each specimen. All morphometrics were measured by the same researcher (R. A. C. Leys).
Birds were aged using the Wolfe-Ryder-Pyle system (Wolfe et al. 2010). In Eastern White-crowned Sparrows, first-cycle individuals have brown crown stripes, whereas definitive-cycle individuals have black crown stripes (Pyle 1997). From February to June, first-cycle individuals undergo a partial prealternate molt in which they replace some to all of the brown feathers in the crown stripes with black feathers, rendering this criterion unreliable in the spring for birds with black crown stripes (Pyle 1997). However, individuals with black crown stripes in the spring can be conclusively aged as first-cycle using feather shape (pointed in first-cycle vs. round in definitive-cycle) and level of abrasion (abraded in first-cycle vs. fresh in definitive-cycle) in the primary coverts and rectrices (Pyle 1997; see pages 589–591 for details). Following these guidelines, we assigned individuals a molt category of either first-cycle or definitive-cycle (Wolfe et al. 2010). Of the 38 birds we sampled, 28 were first-cycle and 10 were definitive-cycle.
Molecular sexing
We extracted DNA from blood using a standard salt extraction protocol (Miller et al. 1988; details in Appendix 1). DNA was extracted from feathers using a Cell and Tissue DNA Isolation Kit (Norgen) following the manufacturer’s recommended protocol for lysate preparation from animal tissues. Prior to beginning this protocol, we used scissors sterilized with 70% ethanol to remove the feather barbules from the rachis, then cut the rachis into small pieces. Scissors were sterilized between each feather sample to avoid potential contamination. We used an extraction kit for feathers because we expected it to increase the DNA yield and purity compared with the in-house salt extraction protocol we typically use for blood.
After extraction, we used a nanodrop spectrophotometer (ND-1000) to measure the concentration of DNA extracted from blood and feathers. The mean concentration of DNA extracted from blood was 171 ng/µL (range = 26–296 ng/µL), with 260/280 ratios around 1.8. The mean concentration of DNA extracted from feathers was 39 ng/µL (range = 7–143.5 ng/µL), with 260/280 ratios around 1.4. One feather sample with relatively high concentration of DNA (143.5 ng/µL) had some blood on the feather shaft, which likely explains the higher DNA yield. Removing this sample from our calculation, the mean DNA concentration of feathers was 28.1 ng/µL. A 260/280 ratio of approximately 1.8 is usually considered “pure” for DNA, whereas lower ratios may indicate the presence of protein, alcohol, or other contaminants that absorb strongly at 280 nm; thus, we obtained higher quality DNA from blood than feathers, despite using a kit designed for extracting DNA from animal tissues for the feather samples. We diluted the DNA extracted from blood to 20 ng/µL; given the generally lower concentration of DNA extracted from feathers, we left those samples undiluted.
We then used the P2/P8 sexing protocol described by Griffiths et al. (1998) to determine the sex of Eastern White-crowned Sparrows, conducting PCR reactions in a total volume of 15 µL, including 20 ng total genomic DNA as template for blood or mean = 39 ng (range = 7–143.5 ng) total genomic DNA for feathers, 0.2 mM dNTPs, 2.0 mM MgCl2, 1X buffer, 0.2 µM of each primer, and 0.5 units Taq DNA polymerase (Invitrogen). For feather samples that did not amplify after our first PCR attempt, we attempted PCR up to three more times, adding 0.8–1.5 mg/mL bovine serum albumen to improve amplification. All PCR reactions included a negative control using ddH2O instead of template, and two positive controls that used 20 ng total genomic DNA from a male and female Eastern White-crowned Sparrow of known sex as determined by PCR amplification from blood (Fig. 2). Thermocycling conditions included an initial denaturation step of 94°C for 90 s; 30 cycles of 94°C for 30 s, 48°C for 45 s, and 72°C for 45 s; and a final extension step of 72°C for 5 min.
Statistical analysis
All analyses were conducted using R statistical software version 1.4.1717 (R Core Team 2021). Given the unequal sample sizes we obtained for sex within molt category (details in Results), we excluded definitive-cycle birds (N = 7) from further statistical analysis. Given our relatively small sample size for first-cycle individuals (N = 20, details in Results), we wanted to pool the data to increase statistical power. Before pooling our data, we used a Welch’s t-test in base R to evaluate whether there were significant differences in wing chord length of first-cycle individuals between live birds and specimens from the Fatal Light Awareness Program collection. Using these 20 first-cycle birds, we then ran a Welch’s t-test to test for a significant difference in wing chord length between the sexes. To control the false discovery rate associated with multiple testing, we calculated adjusted P values for each of the two t-tests with ‘p.adjust’ in base R using the Benjamini-Hochberg method (Benjamini and Hochberg 1995).
Next, we determined the wing chord length ranges in first-cycle individuals for each sex using the method outlined by Pyle (1997): we calculated the mean wing chord length for each sex, then added and subtracted two standard deviations (SD) from each mean. We used these ranges because they represent approximately 95% of the individuals in the population, which is the current acceptable standard from the Bird Banding Laboratory and the Canadian Wildlife Service (Pyle 1997). We then used long-term banding data to calculate the proportion of Eastern White-crowned Sparrows that can be reliably sexed using wing chord length. We counted the number of individuals that fell outside the overlapping range of wing chord lengths where sex determination is not reliable (based on our data set of molecularly sexed birds) and divided this number by the total number of individuals banded at LPBO between 2015 and 2022.
RESULTS
Harvey et al. (2006) reported that using blood or feathers was equally effective for DNA sex determination in small passerines. However, we found that DNA extraction from blood was more effective than feathers. We obtained usable DNA from all 13 blood samples but were only able to amplify DNA from 56% (14/25) of feather samples. After molecular sexing, we determined that we had sampled 19 males (13 first-cycle, 6 definitive-cycle) and 8 females (7 first-cycle, 1 definitive-cycle).
There was no significant difference in wing chord length between live and dead birds (t = −0.14, adjusted P = 0.891, df = 17.9). Male Eastern White-crowned Sparrows in first-cycle plumage had significantly longer wing chord lengths than first-cycle females (male: mean = 78.7 mm ± 2.2 SD, female: mean = 73.6 mm ± 1.4 SD; t = −6.4, adjusted P < 0.0001, df = 17.2; Fig. 3). First-cycle males had a wing chord length range of 74.3–83.0 mm, whereas first-cycle females had a wing chord length range of 70.8–76.4 mm. These results mean that there is an overlapping range of 74–76 mm for which sex cannot be reliably determined by wing chord length for Eastern White-crowned Sparrows in first-cycle plumage. Thus, in our full data set, 71.4% (20/28) of first-cycle birds could be reliably sexed by wing chord length. From the 2015–2022 LPBO data set, the number of individuals in first-cycle plumage that could be reliably sexed by wing chord length was 63.8% (4376/6854).
DISCUSSION
We show that males of the Eastern subspecies of the White-crowned Sparrow in first-cycle plumage have significantly longer wing chord lengths than females. While our sample size was small (20 first-cycle birds), our results do suggest that the Eastern subspecies can be reliably sexed using wing chord length outside an overlapping range of 74–76 mm in first-cycle individuals. Individuals within the overlapping range cannot be reliably sexed by wing chord length and should be denoted as having an unknown sex. Finding an overlapping range in wing chord length for which sex determination is not reliable is common in passerines, but when the range of overlap is small enough, this measurement can still be used to reliably sex most individuals in a population (Pyle 1997). According to our calculations for first-cycle birds, the 2 mm overlapping range in wing chord length for males and females would allow nearly two-thirds of birds in first-cycle plumage to be reliably sexed in the hand. Therefore, we recommend that banders and other researchers use wing chord length to determine the sex of individuals in first-cycle plumage, and only use PCR when individuals fall within the overlapping range. When conducting DNA-based sexing, we recommend using blood samples because DNA extraction from blood was 100% effective, whereas DNA extraction from feathers was only 56% effective.
Other methods have been developed to sex monochromatic bird species, such as performing discriminant function analysis of multiple morphometric measurements (including tarsus length, tibia length, bill length, and tail length; Desrochers 1990, Analla et al. 2022). These measurements, combined with wing chord length, could be used to reduce the overlapping range (i.e., birds that cannot be sexed) in monochromatic first-cycle Eastern White-crowned Sparrows; however, these data are time-consuming to collect and may not be practical for all researchers. Nevertheless, future studies that incorporate additional morphometric measurements could evaluate the utility of discriminant function analysis for sex determination of Eastern White-crowned Sparrows regardless of molt cycle. Future research is also needed to test whether other sexually monochromatic bird species can be reliably sexed by wing chord length. Based on personal observations (R. A. C. Leys), candidate species to test are the American Tree Sparrow (Spizelloides arborea) and the Brown Creeper (Certhia americana).
Our findings are important for banding stations and researchers. Banding stations play a critical role in population trend monitoring for migratory species (Hussell and Ralph 2005, Miller-Rushing et al. 2008). They are also crucial for researchers because they provide access to birds for research projects and information that is vital for the conservation of migratory bird species, the latter of which is highlighted by the fact that the 2022 State of the World’s Birds report included data that were made possible by banding stations (BirdLife International 2022). The tool that we have created for sexing Eastern White-crowned Sparrows in first-cycle plumage will allow banding stations in eastern North America to improve their data by recording the sex of first-cycle individuals in this subspecies. Providing a reliable sex determination range for first-cycle birds is particularly important because the proportion of first-cycle birds captured at banding stations can be as high as 87% in autumn and 66.4% in spring (Mills 2016). Further, collecting wing chord length data is part of the standard banding protocol for most banding stations, so the tool we have provided can be seamlessly implemented into each banding station’s operations without adding additional steps to the process or causing the birds additional stress. For other researchers, our study provides a simple tool for sexing first-cycle Eastern White-crowned Sparrows in the hand. Wing chord length measurements are quick, inexpensive, non-invasive, and repeatable, and they allow researchers to select individuals for their study based on sex and to test for differential responses between the sexes when considering aspects of this subspecies’ biology.
Our tool is particularly important in the pre-migratory, migratory, and overwintering periods when breeding characteristics are not present. One limitation of our study is that, due to our small sample size, we were only able to statistically determine that first-cycle birds can be reliably sexed by wing chord length. Future research should focus on evaluating the use of wing chord length as a tool for sexing definitive-cycle Eastern White-crowned Sparrows. We encourage researchers working on Eastern White-crowned Sparrows during the breeding season to compare the wing chord lengths of individuals with breeding characteristics (i.e., individuals of known sex) with the wing chord length ranges we present here. We expect that doing so will further validate this method and increase researcher confidence in the wing chord length ranges we have identified as reliable for sex determination in first-cycle Eastern White-crowned Sparrows. Relatedly, if there are researchers with existing DNA libraries for Eastern White-crowned Sparrows, we encourage them to validate our method using DNA-based sex determination.
Our findings open avenues for future research on this subspecies; for example, researchers may be able to use sex, based on wing chord length, to test for evidence of protandry in first-cycle Eastern White-crowned Sparrows or to evaluate temporal changes in the population sex ratio of first-cycle birds. Protandry is the phenomenon of males arriving earlier to breeding locations than females (Coppack and Pulido 2009). Climate change is increasing the degree of protandry in many species, including Barn Swallows (Hirundo rustica; Møller 2004) and Willow Warblers (Phylloscopus trochilus; Hedlund et al. 2022), but so far seems to have no effect on other passerine species (Rainio et al. 2007). Protandry has risks, i.e., in years with adverse conditions at breeding sites, early arriving males experience increased mortality (Møller 1994), but also has benefits, i.e., early arrival leads to increased fitness in years with favorable conditions (Kokko et al. 2006). Our study provides a critical tool for testing whether first-cycle Eastern White-crowned Sparrows exhibit protandry, and if the degree of protandry has shifted due to climate change. Long-term banding stations such as LPBO have decades of historical migration banding data, including information on wing chord length and capture date, which can be used to determine whether protandry exists and how the degree of protandry may be shifting due to climate change.
Population sex ratios can be described in two main ways: focusing on offspring or adult sex ratios. Offspring sex ratio, the ratio of male to female offspring produced by breeding adults, has been the subject of many studies (e.g., Svensson and Nilsson 1996, Whittingham and Dunn 2000, Bonier et al. 2007). Adult sex ratio, however, has received less attention and remains poorly studied (Donald 2007). Contrary to mammals, wild bird populations tend to be male biased, with males outnumbering females by 33% on average (Donald 2007). This information is consistent with our finding, as only 29.6% (8/27) of the individuals in our study were female. Male bias in bird populations is even more pronounced in globally threatened species (Donald 2007), which has profound implications for the management of threatened species and illustrates the importance of quantifying sex ratios in bird populations. The tool we provide here will allow researchers to calculate the sex ratio of first-cycle Eastern White-crowned Sparrows.
RESPONSES TO THIS ARTICLE
Responses to this article are invited. If accepted for publication, your response will be hyperlinked to the article. To submit a response, follow this link. To read responses already accepted, follow this link.
AUTHOR CONTRIBUTIONS
R. A. C. L. designed the study in consultation with L. A. G. Both R. A. C. L. and L. A. G. collected and analyzed the data. R. A. C. L. wrote the manuscript with input from L. A. G. Both coauthors approved the final manuscript.
ACKNOWLEDGMENTS
We thank two anonymous reviewers and the editor, Mark Hauber, for their comments, which improved this manuscript. R. A. C. L. was supported by the Natural Sciences and Engineering Research Council of Canada. Further funding for this project was provided by the West Elgin Nature Trust, the Society of Canadian Ornithologists, and the Robert Cooper Audubon Society. Data for this project were collected at the Long Point Bird Observatory, Port Rowan, Ontario and from the Fatal Light Awareness Program (FLAP) collection, Toronto, Ontario. We thank both organizations for allowing us access to collect data. Special thanks to Liam McGuire, Mark Peck, Mark Conboy, Paloma Plant, Stuart Mackenzie, and the many volunteers for assistance with data collection. Quinlan Mann assisted with feather DNA extraction. This research was conducted on the traditional lands of the Mississaugas of the Credit First Nation and the Haudenosaunee, Attiwonderonk, and Mississauga peoples.
DATA AVAILABILITY
The data/code that support the findings of this study are openly available in Open Science Framework at https://osf.io/rhaex/. Ethical approval for this research study was granted by the Canadian Wildlife Service and Environment and Climate Change Canada: permits 10169 CM, SC-OR02022-00089, and 10169E,F. All animal procedures were approved by the McMaster University Animal Research Ethics Board (AUP 18-05-20) and the University of Waterloo Animal Care Committee (43009).
LITERATURE CITED
Analla, M., P. Fernández-Rodríguez, N. Martínez-Medina, and C. Azorit. 2022. Sexing Eurasian Eagle Owls by external body and skeletal measurements. Journal of Field Ornithology 93(4):1. https://doi.org/10.5751/JFO-00175-930401
Andreasson, F., A. Nord, and J.-Å. Nilsson. 2020. Body temperature responses of Great Tits Parus major to handling in the cold. Ibis 162(3):836-844. https://doi.org/10.1111/ibi.12737
Banks, R. C. 1964. Geographic variation in the White-crowned Sparrow, Zonotrichia leucophrys. University of California Press, Berkeley, California, USA.
Bell, D. M. 2020. Determination of sex using wing chord in southern Vancouver Island populations of Bewick’s Wren and “Puget Sound” White-crowned Sparrow. North American Bird Bander 45(3):112-118. https://sora.unm.edu/node/153379
Benjamini, Y., and Y. Hochberg. 1995. Controlling the false discovery rate: a practical and powerful approach to multiple testing. Journal of the Royal Statistical Society B 57(1):289-300. https://www.jstor.org/stable/2346101
BirdLife International. 2022. State of the world’s birds 2022: insights and solutions for the biodiversity crisis. BirdLife International, Cambridge, UK. https://www.birdlife.org/papers-reports/state-of-the-worlds-birds-2022/
Bonier, F., P. R. Martin, and J. C. Wingfield. 2007. Maternal corticosteroids influence primary offspring sex ratio in a free-ranging passerine bird. Behavioral Ecology 18(6):1045-1050. https://doi.org/10.1093/beheco/arm075
Busse, P., and W. Meissner. 2015. Bird ringing station manual. De Gruyter Open Poland, Warsaw, Poland. https://doi.org/10.2478/9788376560533
Carrillo-Ortiz, J., S. Guallar, J. Martínez-Vargas, and J. Quesada. 2021. A revision of bird skin preparation aimed at improving the scientific value of ornithological collections. Avian Biology Research 14(2):48-54. https://doi.org/10.1177/1758155920987151
Coppack, T., and F. Pulido. 2009. Proximate control and adaptive potential of protandrous migration in birds. Integrative and Comparative Biology 49(5):493-506. https://doi.org/10.1093/icb/icp029
Desrochers, A. 1990. Sex determination of Black-capped Chickadees with a discriminant analysis. Journal of Field Ornithology 61(1):79-84. https://www.jstor.org/stable/4513504
Donald, P. F. 2007. Adult sex ratios in wild bird populations. Ibis 149(4):671-692. https://doi.org/10.1111/j.1474-919X.2007.00724.x
Fugle, G. N., and S. I. Rothstein. 1985. Age- and sex-related variation in size and crown plumage brightness in wintering White-Crowned Sparrows. Journal of Field Ornithology 56(4):356-368. https://www.jstor.org/stable/4513049
Gill, S. A., and M. J. Vonhof. 2006. Sexing monochromatic birds in the field: cryptic sexual size dimorphism in Buff-breasted Wrens (Thryothorus leucotis). Ornitologia Neotropical 17:409-418.
Griffiths, R., S. Daan, and C. Dijkstra. 1996. Sex identification in birds using two CHD genes. Proceedings of the Royal Society B 263(1374):1251-1256. https://doi.org/10.1098/rspb.1996.0184
Griffiths, R., M. C. Double, K. Orr, and R. J. G. Dawson. 1998. A DNA test to sex most birds. Molecular Ecology 7(8):1071-1075. https://doi.org/10.1046/j.1365-294x.1998.00389.x
Harvey, M. G., D. N. Bonter, L. M. Stenzler, and I. J. Lovette. 2006. A comparison of plucked feathers versus blood samples as DNA sources for molecular sexing. Journal of Field Ornithology 77(2):136-140. https://doi.org/10.1111/j.1557-9263.2006.00033.x
Hedlund, J., T. Fransson, C. Kullberg, J.-O. Persson, and S. Jakobsson. 2022. Increase in protandry over time in a long-distance migratory bird. Ecology and Evolution 12(7):e9037. https://doi.org/10.1002/ece3.9037
Hussell, D. J. T., and C. J. Ralph. 2005. Recommended methods for monitoring change in bird populations by counting and capture of migrants. North American Bird Bander 30(1):6-20. https://sora.unm.edu/node/93689
Jeans, L. 2022. Over-wintering grounds social behavior of White-Crowned Sparrows. Undergraduate Thesis. Claremont McKenna College, Claremont, California, USA. https://scholarship.claremont.edu/cmc_theses/3046/
Jones, S. G., V. V. Vyazovskiy, C. Cirelli, G. Tononi, and R. M. Benca. 2008. Homeostatic regulation of sleep in the White-crowned Sparrow (Zonotrichia leucophrys gambelii). BMC Neuroscience 9:47. https://doi.org/10.1186/1471-2202-9-47
King, J. R., D. S. Farner, and L. R. Mewaldt. 1965. Seasonal sex and age ratios in populations of the White-Crowned Sparrows of the race gambelii. Condor 67(6):489-504. https://doi.org/10.2307/1365613
Kokko, H., T. G. Gunnarsson, L. J. Morrell, and J. A. Gill. 2006. Why do female migratory birds arrive later than males? Journal of Animal Ecology 75(6):1293-1303. https://doi.org/10.1111/j.1365-2656.2006.01151.x
Landys, M. M., J. C. Wingfield, and M. Ramenofsky. 2004. Plasma corticosterone increases during migratory restlessness in the captive White-crowned Sparrow Zonotrichia leucophrys gambelli. Hormones and Behavior 46(5):574-581. https://doi.org/10.1016/j.yhbeh.2004.06.006
Lipshutz, S. E., I. A. Overcast, M. J. Hickerson, R. T. Brumfield, and E. P. Derryberry. 2017. Behavioural response to song and genetic divergence in two subspecies of White-crowned Sparrows (Zonotrichia leucophrys). Molecular Ecology 26(11):3011-3027. https://doi.org/10.1111/mec.14002
Miller, S. A., D. D. Dykes, and H. F. Polesky. 1988. A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Research 16(3):1215. https://doi.org/10.1093/nar/16.3.1215
Miller-Rushing, A. J., T. L. Lloyd-Evans, R. B. Primack, and P. Satzinger. 2008. Bird migration times, climate change, and changing population sizes. Global Change Biology 14(9):1959-1972. https://doi.org/10.1111/j.1365-2486.2008.01619.x
Mills, A. M. 2016. Banding data reveal bias in age-class sampling of songbirds during spring migration. Journal of Field Ornithology 87(3):323-336. https://doi.org/10.1111/jofo.12156
Møller, A. P. 1994. Phenotype-dependent arrival time and its consequences in a migratory bird. Behavioral Ecology and Sociobiology 35(2):115-122. https://doi.org/10.1007/BF00171501
Møller, A. P. 2004. Protandry, sexual selection and climate change. Global Change Biology 10(12):2028-2035. https://doi.org/10.1111/j.1365-2486.2004.00874.x
Moore, M. C., R. S. Donham, and D. S. Farner. 1982. Physiological preparation for autumnal migration in White-Crowned Sparrows. Condor 84(4):410-419. https://doi.org/10.2307/1367445
Morris, S. R., K. M. Covino, J. D. Jacobs, and P. D. Taylor. 2016. Fall migratory patterns of the Blackpoll Warbler at a continental scale. Auk 133(1):41-51. https://doi.org/10.1642/AUK-15-133.1
Morton, M. L. 1992a. Control of postnuptial molt in the Mountain White-Crowned Sparrow: a perspective from field data. Ornis Scandinavica (Scandinavian Journal of Ornithology) 23(3):322-327. https://doi.org/10.2307/3676656
Morton, M. L. 1992b. Effects of sex and birth date on premigration biology, migration schedules, return rates and natal dispersal in the Mountain White-Crowned Sparrow. Condor 94(1):117-133. https://doi.org/10.2307/1368802
Pyle, P. 1997. Identification guide to North American birds. First edition. Slate Creek Press, Bolinas, California, USA.
R Core Team. 2021. R: a language and environment for statistical computing. Version 4.3.0. https://www.R-project.org/
Rainio, K., A. P. Tøttrup, E. Lehikoinen, and T. Coppack. 2007. Effects of climate change on the degree of protandry in migratory songbirds. Climate Research 35(1-2):107-114. https://doi.org/10.3354/cr00717
Rattenborg, N. C., B. H. Mandt, W. H. Obermeyer, P. J. Winsauer, R. Huber, M. Wikelski, and R. M. Benca. 2004. Migratory sleeplessness in the White-Crowned Sparrow (Zonotrichia leucophrys gambelii). PLoS Biology 2(7):e212. https://doi.org/10.1371/journal.pbio.0020212
Romero, L. M., M. Ramenofsky, and J. C. Wingfield. 1997. Season and migration alters the corticosterone response to capture and handling in an Arctic migrant, the White-Crowned Sparrow (Zonotrichia leucophrys gambelii). Comparative Biochemistry and Physiology Part C: Pharmacology, Toxicology and Endocrinology 116(2):171-177. https://doi.org/10.1016/S0742-8413(96)00208-3
Schmaljohann, H., C. Meier, D. Arlt, F. Bairlein, H. van Oosten, Y. E. Morbey, S. Åkesson, M. Buchmann, N. Chernetsov, R. Desaever, J. Elliott, M. Hellström, F. Liechti, A. López, J. Middleton, U. Ottosson, T. Pärt, F. Spina, and C. Eikenaar. 2016. Proximate causes of avian protandry differ between subspecies with contrasting migration challenges. Behavioral Ecology 27(1):321-331. https://doi.org/10.1093/beheco/arv160
Seewagen, C. L., C. G. Guglielmo, and Y. E. Morbey. 2013. Stopover refueling rate underlies protandry and seasonal variation in migration timing of songbirds. Behavioral Ecology 24(3):634-642. https://doi.org/10.1093/beheco/ars225
Svensson, E., and J.-Å. Nilsson. 1996. Mate quality affects offspring sex ratio in blue tits. Proceedings of the Royal Society B 263(1368):357-361. https://doi.org/10.1098/rspb.1996.0055
Vučićević, M., J. Stevanović, M. Šekler, R. Resanović, and Z. Stanimirović. 2016. Historical overview of methods for sex determination in birds. Veterinarski Glasnik 70(3-4):145-157. https://doi.org/10.2298/VETGL1604145V
Warkentin, I. G., R. H. M. Espie, D. J. Lieske, and P. C. James. 2016. Variation in selection pressure acting on body size by age and sex in a reverse sexual size dimorphic raptor. Ibis 158(3):656-669. https://doi.org/10.1111/ibi.12369
Whittingham, L. A., and P. O. Dunn. 2000. Offspring sex ratios in tree swallows: females in better condition produce more sons. Molecular Ecology 9(8):1123-1129. https://doi.org/10.1046/j.1365-294x.2000.00980.x
Wolfe, J. D., T. B. Ryder, and P. Pyle. 2010. Using molt cycles to categorize the age of tropical birds: an integrative new system. Journal of Field Ornithology 81(2):186-194. https://doi.org/10.1111/j.1557-9263.2010.00276.x
Zhu, B.-R., M. A. Verhoeven, C. J. Hassell, K. K.-S. Leung, D. Dorofeev, Q. Ma, K. Eiamampai, J. T. Coleman, U. Tserenbat, G. Purev-Ochir, D. Li, Z. Zhang, and T. Piersma. 2023. Predicting the non-breeding distributions of the two Asian subspecies of Black-tailed Godwit using morphological information. Avian Research 14:100069. https://doi.org/10.1016/j.avrs.2022.100069
Fig. 1

Fig. 1. The Eastern White-crowned Sparrow (a) is the dominant subspecies in eastern North America. It can be easily distinguished from the Gambel’s subspecies (b), which occurs rarely in eastern North America, by the presence of black in the lores, indicated by the red arrows. Photos: (a) Ryan Leys, (b) Brian Sullivan (ML 27434901).

Fig. 2
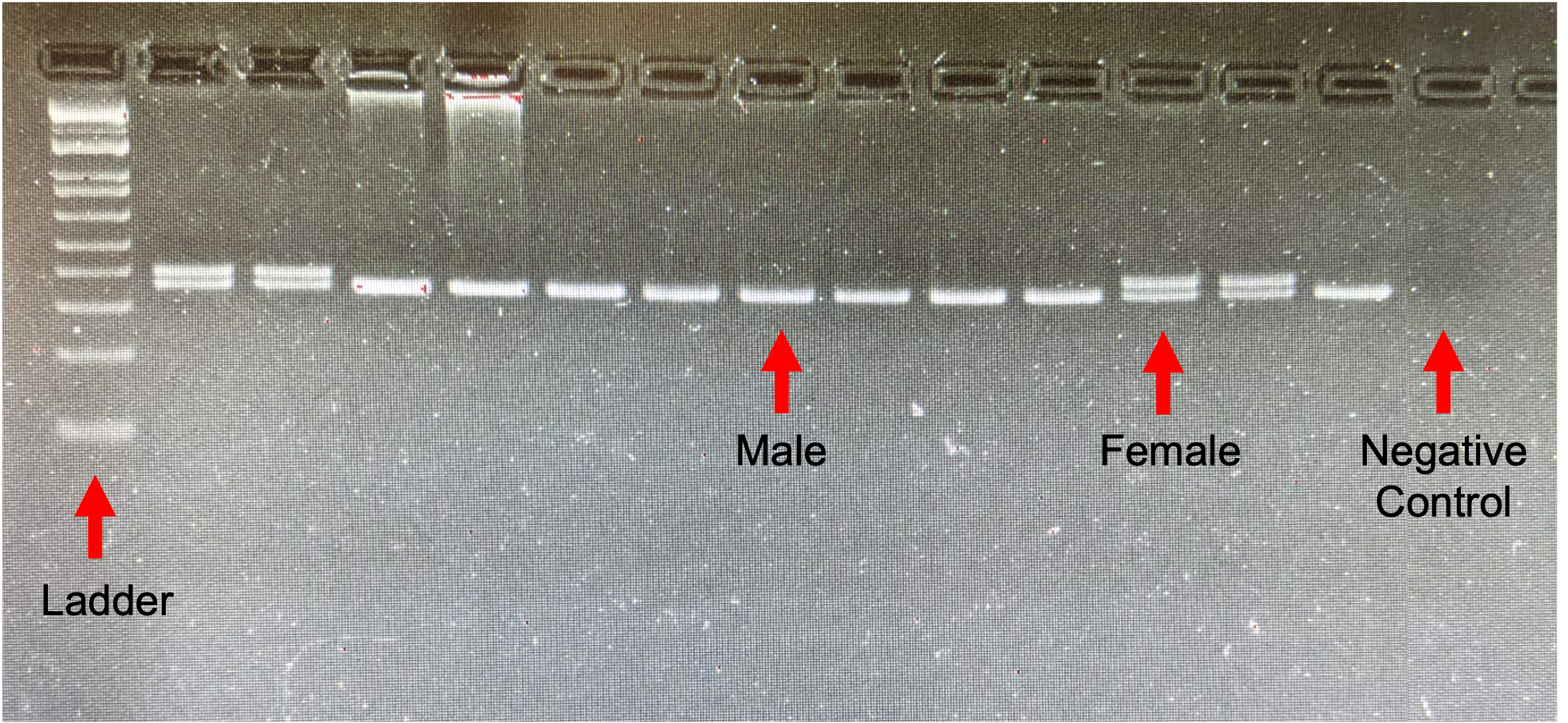
Fig. 2. P2/P8 sexing PCR results for 13 Eastern White-crowned Sparrows using DNA extracted from blood. Double bands indicate females (Z and W chromosomes), and single bands indicate males (Z chromosome), as shown by the red arrows.

Fig. 3
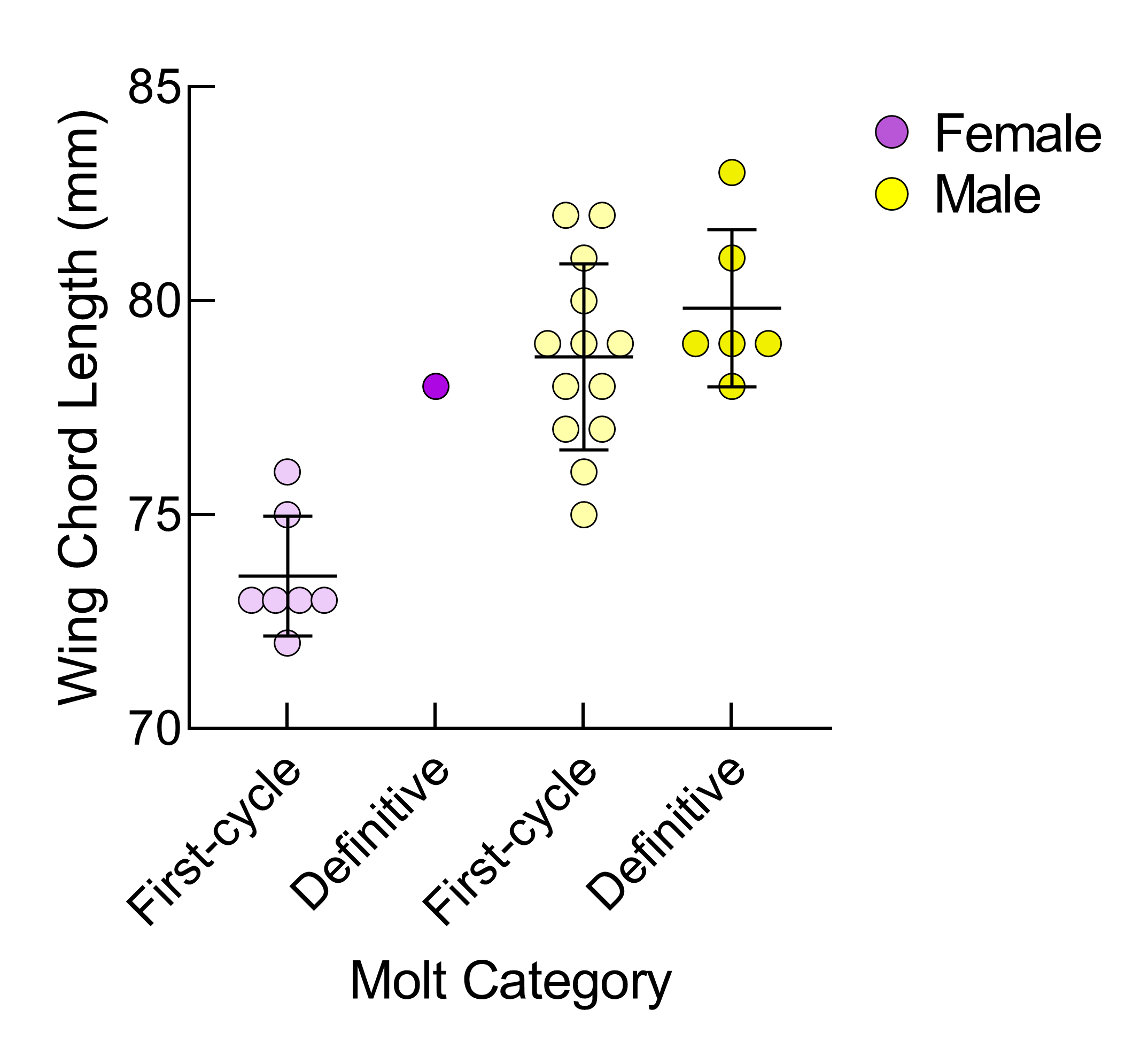
Fig. 3. Eastern White-crowned Sparrow wing chord lengths (mm) by molt category and sex. Points represent the individual measurements for all 27 birds that were molecularly sexed, and lines represent the mean ± standard deviation for each category.









