The following is the established format for referencing this article:
Stenhouse, E. H., P. Bellamy, I. Vaughan, W. Kirby, W. O. C. Symondson, and P. Orozco-terWengel. 2023. Using DNA metabarcoding to explore spatial variation in diet across European Hawfinch populations. Journal of Field Ornithology 94(1):12.ABSTRACT
The investigation of diet in avian species is essential for understanding their ecology and local adaptations, as well as long-term conservation. This can be particularly challenging because of the wide distribution and high ecological plasticity of many bird species. Here, we focused on the Hawfinch (Coccothraustes coccothraustes), which has shown variation in population trends. Across Europe, central and eastern European populations are moderately declining while western European populations are moderately increasing. Ecological drivers behind these differing trends are still unknown; one possibility is differences in diet, yet little research has been conducted into Hawfinch diet in mainland Europe or elsewhere. Dietary richness and variation are under-studied in woodland bird species, due primarily to challenges in accurately identifying plant and invertebrate taxa consumed. This study presents the first molecular dietary analysis of Hawfinch populations across two European countries. Faecal samples were collected between January and July of 2019 from Hawfinch caught at six artificial feed sites: two in Denmark and four in Germany. We successfully extracted DNA from 80 samples by amplifying plant Internal Transcribed Spacer 2 (ITS2) and invertebrate Cytochrome Oxidase Subunit 1 (COI) barcodes. A total of 35 plant and 37 invertebrate taxa were found, with plant and insect orders Fagales and Lepidoptera, respectively, the most frequently detected. Hawfinch dietary composition differed significantly between European countries, suggesting Hawfinch can make use of available food resources that are likely to differ spatially. Our study shows how DNA metabarcoding can be used to provide novel ecological information associated with under-studied bird species, thus providing essential information for future management and conservation of Hawfinch and their habitats.
RESUMEN
El estudio de la dieta en especies de aves es esencial para entender su ecología y adaptaciones locales, así como su conservación a largo plazo. Esto puede ser particularmente difícil por las amplias distribuciones y la alta plasticidad ecológica que muestran varias especies de aves. Aquí nos enfocamos en Coccothraustes coccothraustes la cuál ha mostrado variación en tendencias poblacionales. A lo largo de Europa, las poblaciones del centro y del este están disminuyendo moderadamente mientras que las poblaciones del oeste están incrementando moderadamente. Los factores ecológicos detrás de esta diferencia en las tendencias son desconocidos hasta el momento; una posibilidad es la diferencia en la dieta, pero hay poca investigación en la dieta de C. coccthraustes en Europa continental o en cualquier otro lugar. La riqueza en la dieta y su variación ha sido poco estudiada en especies de aves de bosque, principalmente debido a retos relacionados con la correcta identificación de plantas e invertebrados consumidos. Este estudio presenta el primer análisis molecular de la dieta de las poblaciones de C. coccothraustes a lo largo de dos países europeos. Muestras de materia fecal fueron colectadas entre enero y julio de 2019 de individuos de C. coccothraustes capturados en seis sitios con alimentación artificial: dos en Dinamarca y cuatro en Alemania. Extrajimos con éxito el ADN de 80 muestras mediante la amplificación de los códigos de barra Espaciador Interno Transcrito 2 (ITS2) de las plantas y Citocromo Oxidasa 1 (CO1) en invertebrados. Encontramos un total de 35 plantas y 37 invertebrados y los ordenes Fagales y Lepidóptera de plantas e invertebrados, respectivamente, fueron los más frecuentemente detectados. La composición de la dieta de C. coccothraustes fue significativamente diferente entre los países europeos. Sugiriendo que C. coccothraustes puede usar los recursos alimenticios disponibles que muy probablemente se diferencian espacialmente. Nuestro estudio muestra como los meta códigos de barras de ADN pueden ser usados para proveer nueva información ecológica asociada con especies de aves pobremente estudiadas, proporcionando información esencial para futuros manejo y conservación de C. coccothraustes y sus hábitats.
INTRODUCTION
Characterizing the dietary niche of avian species is a vital step in identifying their role within ecosystems (Hoenig et al. 2022). Diet is a central component of a bird’s ecology, and therefore provides a crucial dimension within life history, as well as determining species’ energetic investment, survival, reproduction, and subsequent fitness (Sibly et al. 2012, Barnagaud et al. 2019). Avian diet studies have helped to characterize ecological interactions of birds (Burin et al. 2016) as well as identify prey preferences as an ecological driver of the evolution across the Class Aves (Kissling et al. 2012, Barnagaud et al. 2014). Therefore, having an in-depth understanding of avian diet allows an accurate interpretation of the complex interactions that birds have within their environment (Hoenig et al. 2022).
An individual’s energy balance is influenced by biotic factors such as food availability (Czenze et al. 2018). As a result, birds are likely to experience location-dependent factors that directly influence their energetic balance (Dunbar and Brigham 2010, Stawski and Geiser 2011). Therefore, investigating how diet differs spatially is fundamental in understanding how populations are locally adapted to the species on which they feed (Romano et al. 2020). Ecological and climatic conditions directly affect the presence and availability of organisms, resulting in substantial impacts on species composition within the diet (Willig et al. 2003, Romano et al. 2020). Variation in the distribution of prey species across large spatial gradients has been shown to impact food consumption and predation strategies (Terraube and Arroyo 2011, Romano et al. 2020).
Spatial adjustments in dietary composition are likely to be of high significance to individuals for life-history characteristics such as reproduction. Therefore, the question of where and how differently populations exploit resources has practical implications for conservation management (Terraube and Arroyo 2011). Despite the importance of acquiring information regarding intraspecific dietary variation, particularly across large spatial scales, this information is currently lacking for many woodland bird species.
A major issue to address within avian ecology studies is how the same species’ diet differs between urban and semi-natural habitats (Coogan et al. 2018). Anthropogenic food resources can distort the diets of species that frequent urban areas (Coogan et al. 2018). Dietary analysis of Australian Silver Gulls (Chroicocephalus novaehollandiae) found 85% of stomach contents consisted of human discarded food resources, while approximately 38% of suburban Florida Scrub Jay diet (Aphelocoma coerulescens) consisted of anthropogenically provided peanuts (Smith and Carlile 1993, Fleischer et al. 2003). Dietary differences can have strong implications for fitness of individuals utilizing anthropogenic food resources, if their diet is imbalanced relative to the nutritional requirements needed for survival within a natural environment (Coogan et al. 2018).
The ability of certain bird species to persist within urban environments is attributed to their successful exploitation of resources within urban areas (Sol et al. 2014). This ability is an important driver of the demographic response to urbanization (Evans and Gawlik 2020). Foraging theory predicts lower value food items should be incorporated into the diet when natural food resources become scarce, as individuals become less selective (MacArthur and Pianka 1966). As a result, birds would be expected to access anthropogenic food resources when natural food is less abundant (Evans and Gawlik 2020).
One of the main difficulties when conducting dietary studies is related to limitations of traditional dietary analysis methods, such as hard-part microscopy. For example, microscopic analysis of fecal samples rarely provides the depth of taxonomic resolution required to detect species-level dietary differences (da Silva et al. 2020). Recent developments in high-throughput sequencing (HTS) allows for the genetic analysis of diets through metabarcoding by using molecular barcodes amplified from fecal DNA (Taberlet et al. 2018). Metabarcoding can result in higher taxonomic resolution within dietary studies and improved taxonomic accuracy (Ando et al. 2013, Galimberti et al. 2016, Dunn et al. 2018).
The Hawfinch (Coccothraustes coccothraustes) is widespread throughout mainland Europe and can be resident, a short-distance migrant, or summer visitor (Tomiałojc 2005). Within Europe, Hawfinch are found throughout flood plain, mature, and semi-natural forests containing beech (Fagus sylvatica), hornbeam (Carpinus betulus), lime (Tilia sp.), and oak (Quercus sp.; Bijlsma 1998, Tomiałojc 2005). Hawfinch are predominately arboreal, and visual observation studies from the UK have established that during the breeding season (typically April–June), their diet includes seeds and buds of cherry (Prunus sp.) and elm (Ulmus sp.), alongside Coleoptera, Hemiptera, Annelida, Gastropoda, and Araneae (Mountford 1957, Newton 1967).
The Pan European Common Bird Monitoring Scheme (PECBMS) has revealed that Hawfinch showed an overall “moderate increase” in both long-term (1980–2014) and short-term (2005–2014) datasets (PECBMS 2019). Within this overall trend however, regional variations in population trends were considerable. Central and eastern European populations showed “moderate declines,” whereas western European populations showed “moderate increases” (Kirby et al. 2018, PECBMS 2019). Reasons behind these differing trends are unknown.
There are no studies using DNA metabarcoding to investigate dietary range and composition of European Hawfinch diet within the literature. Because of this, the capacity of metabarcoding analysis to reveal the full taxonomic breadth of Hawfinch dietary items remains unexplored. By analyzing Hawfinch diet at a high taxonomic resolution and exploring dietary variation among populations, suitably tailored management of the wider landscape can be implemented to ensure Hawfinch populations persist within areas. This could include the protection of important dietary items, or creating increased areas of suitable habitat to enable Hawfinch persistence.
Herein, we present the first metabarcoding dietary analysis of Hawfinch populations across a range of different forest types and therefore, assumed different plant and invertebrate communities. Using fecal samples collected from Hawfinches, we sought to use metabarcoding to: (a) document the complete dietary composition of plants and invertebrates consumed by Hawfinch and (b) investigate spatial diet composition changes across Hawfinch populations in Denmark and Germany.
METHODS
This study was undertaken at six artificial feeding sites, two in Denmark and four in Germany (Fig. 1). Sunflower seeds (Helianthus sp.) were provided continuously at all feeding sites. Danish artificial feeding sites were located within urban environments in central Jutland (Bryup and Solkaer), with dominant tree species present in the broader landscape being beech, oak, and fir (Abies sp.). German sites were located within heterogeneous woodlands primarily consisting of beech, oak, and birch (Betula sp.) situated near the towns of Bad Homburg, Hilden-Haan, Velbert, and Wulfrathe-Hohe.
Fecal sample collection
Fecal samples were collected between January and July 2019. Operating under European Union for Bird Ringing (EURING) approved licenses, trained bird ringers caught Hawfinches using mist nests and fit them with a metal identification ring. We placed individual birds in a new, clean, paper bag, inside a cloth bird bag, for 10–20 minutes until they defecated. After removing the feces from the paper bag using new, sterilized, plastic toothpicks, we placed them in 2 ml microcentrifuge tubes and stored them at -20 °C until laboratory processing (Fernandes et al. 2023). Each sample was assigned a sample identification number based on the site and ring number of the Hawfinch. If repeated capture occurred during the same ringing session a fecal sample was not collected unless a sample was not obtained during the first capture. However, if the same individual was re-trapped during a separate session, a fecal sample was taken if provided and ring number and date recorded.
DNA extraction and PCR
DNA was extracted from fecal samples using the Qiagen QIAamp DNA Stool Mini Kit (Manchester, UK), following the protocol for pathogen detection with modifications by Davies et al. (2022) designed to improve DNA yields from avian feces. The primer sets used in this study were selected based on their successful application in studies exploring avian diet (Dunn et al. 2018, Moorhouse-Gann et al. 2018, Stockdale 2018, Davies et al. 2022). The “Uniplant” primer set, amplified a 187-387-bp fragment of the Internal Transcribed Spacer 2 (ITS2) region of plant nuclear DNA (Moorhouse-Gann et al. 2018, 2020). We used a combination of mlCOIintF (Leray et al. 2013) and Nancy (Simon et al. 1992) to amplify invertebrate DNA. This was following selection and modification by Stockdale (2018), for amplification of a 306-bp fragment of the cytochrome C oxidase subunit 1 (COI) region (Davies et al. 2022).
Extracted fecal samples underwent molecular identifier tagged (MID-tag) Polymerase Chain Reaction (PCR). This process involved labeling the forward and reverse primers with MID-tagged primers, with samples having a unique pairing of forward and reverse tags for sample identification post-sequencing (Brown et al. 2014, Davies et al. 2022). Reactions were conducted in an Applied Biosystems SimpliAmp™ 96-well thermocycler, with annealing temperatures optimized through temperature gradient PCRs in the same machine. All PCR reactions totaled 25µL and contained 12.5μL of multiplex PCR mix (Qiagen, Manchester, UK), 2.5μL of 2 μM forward and reverse primer, 2.5μL of water, and 5μL template DNA. The ITS2 PCRs comprised 15 minutes at 95 °C, followed by 40 cycles of 95 °C for 30s, 58 °C for 90s, 72 °C for 90s, followed by a final extension at 72 °C for 10 min. All COI PCRs were conducted according to Davies et al. (2022).
Within each PCR 96-well plate, 12 negative (extraction and PCR) and two positive controls were included following Taberlet et al. (2018). Negative PCR controls consisted of DNase- free water. To ensure equimolar concentration of all samples, we pooled samples according to concentrations determined by the Qubit Fluorometer. We pooled negative controls based on the average volume pooled per plate for the Hawfinch samples.
Each pool was cleaned using SPRIselect beads (Beckman Coulter, Brea, USA) with a left-side size selection using a 1.2:1 ratio for ITS2 amplicons to remove fragments below 100 bp, and a 0.8:1 ratio for COI amplicons to remove fragments below 200 bp. The concentration of the pooled DNA was quantified using Qubit dsDNA High‐sensitivity Assay Kits, and quality checked via TapeStation 2200 with a D1000 ScreenTape (Agilent, Santa Clara, USA). The concentration across all pools was quantified using Qubit dsDNA High‐sensitivity Assay Kits. Library preparation for Illumina sequencing was undertaken via NEXTflex Rapid DNA-Seq kit (Bioo Scientific, Austin, USA), with unique adapters added to enable the libraries to be individually bioinformatically identified post sequencing. Pools were combined equimolarly and sequenced on a MiSeq desktop sequencer with a v2 chip.
Bioinformatics and sequencing
The bioinformatic pipeline followed Drake et al. (2021) and Davies et al. (2022). Trimming, aligning, and quality checks were conducted using FastP 0.20.0 (Chen et al. 2018). Tagged reads were demultiplexed using Mothur (Schloss et al. 2009). Unoise3 within Usearch 11 (Edgar 2016, unpublished manuscript, https://doi.org/10.1101/081257) was implemented to remove chimeras and noise, as well as to generate and cluster reads into zero radius Operational Taxonomic Units (hereafter zOTUs) with a 100% clustering threshold (Edgar 2016, unpublished manuscript). Typically, OTUs are defined as clusters of reads which differ by less than a specified sequence dissimilarity threshold, commonly defined at 3% (Callahan et al. 2017). However, newer methodologies use a “denoising” method, which distinguishes sequence variants differing by a single nucleotide (Callahan et al. 2017; Edgar 2016, unpublished manuscript). We chose a denoising method because of the increased taxonomic resolution of this method, as all biological sequences are kept, rather than “lumped” together (Callahan et al. 2017; Edgar 2016, unpublished manuscript).
Blast+ assigned taxonomic identities to zOTU sequences by comparing dietary sequences within fecal samples to reference sequences held on Genbank (Camacho et al. 2009). Unique dietary items were identified through the top hit for each zOTU based on bit-score, using MEGAN6 (Huson et al. 2016). As described in Drake et al. (2021), a minimum percentage score of 97% was deemed suitable for species or genus level classification. Genus level classification was only assigned if multiple zOTUs matched with several species of the same genus. A minimum percentage score of 95% was implemented for classification to the family level. We further removed artefacts, contamination, and implemented the application of sequence thresholds to take into consideration “tag-jumping,” caused by false combinations of used tags in sequencing outputs (Schnell et al. 2015). Sequences that were considered to be noise were also removed. Data from the ITS2 and COI libraries were aggregated to form a single taxon list for each marker, and any sequences assigned to non-target taxa were removed.
Statistical analysis
For all statistical analysis, we used the presence/absence of each taxonomic unit within a fecal sample. The use of relative read abundance (RRA) was not deemed suitable, because of, invariably, the high number of reads detected from the artificially provided sunflower seeds, which may skew ecological conclusions. Furthermore, there are inherent biases present throughout the HTS workflow, including differential DNA extraction success and PCR amplification rates between taxa detected within the diet (Lamb et al. 2019). Additionally, count-based inferences are not advised if little a priori knowledge of the communities analyzed exists (Lamb et al. 2019). Based upon this, we calculated the more conservative frequency of occurrence (FOO)—the proportion of samples in which each dietary taxon was found—to identify the most prevalent taxa within Hawfinch diet. Sunflower seed data were retained for analysis. This was considered reasonable because we wanted to explore supplementary feed use by Hawfinch across urban and semi-natural habitats. All statistical analyses were undertaken in R 4.2.1 (R Core Team 2022) unless otherwise stated.
To estimate sampling completeness of Hawfinch diet, we used calculated Hill number coverage-based sampling curves of the three most commonly used species diversity measures; species richness (Hill-richness), Shannon’s entropy (Hill-Shannon), and Simpson’s index (Hill-Simpson) for each country (Denmark and Germany). Hill-richness is the most sensitive to rare species, while Hill-Shannon can respond to both high and very low rarity values, with Hill-Simpson most sensitive to differences in low rarity values (the relative abundance of common species; Roswell et al. 2021, Tercel et al. 2022). For all diversity estimates, 95% confidence intervals were plotted (Chao and Jost 2012). These metrics were computed using the R package iNEXT (Hsieh et al. 2016).
To explore whether Hawfinch diet differed between Denmark and Germany, we fitted plant and invertebrate multivariate generalized linear models (MGLMs) using the function manyglm within the R package mvabund (Wang et al. 2012). Where an individual had been sampled multiple times, only the first capture was included to avoid pseudo-replication. For each comparison, binomial MGLMs were fitted for multivariate presence/absence data. Two models were fitted using manyglm, with the predictor variable being country (Denmark/Germany) and response being the plant or invertebrate multivariate dietary dataset. The function anova.manyglm was used to significance test each predictor variable within the model and the p.uni = adjusted argument was implemented in order to allow univariate results to be returned (Wang et al. 2012). The p-values returned in this argument were adjusted to control for multiple testing, using a Holm’s step-down resampling algorithm, allowing control over family error rates (Westfall and Young 1993). Parametric bootstrap (Monte Carlo) resampling was applied, as this is recommended for presence-absence data (Wang et al. 2012). For all models, quantile-quantile (Q-Q) diagnostic plots were checked to assess normality in multivariate data, with multivariate homoscedasticity checked by plotting Dunn-Smyth residuals against fitted linear predicted values (Wang et al. 2012, Bates et al. 2015).
Dietary differences in Hawfinch populations were visualized using non-metric multidimensional scaling analysis (nMDS) via the function metaMDS in the vegan package (Oksanen et al. 2019). The nMDS was performed with Jaccard distance in three-dimensional space (k = 3). Spider plots were produced using nMDS results via ordispider and plotted through ggplot2 (Wickham 2016) in R to visualize community differences between countries.
RESULTS
Analyses were conducted on 80 Hawfinch fecal samples (Denmark n = 44; Germany n = 36). From those 80 samples, the sequencing runs produced 1,970,111 ITS2 (plant) and 4,385,796 COI (invertebrate) sequence reads, respectively. A total of 90,847 and 119,241 sequences were detected within negative controls included within the ITS2 and COI runs, respectively. Because of likely human-error based contamination (reads detected in negative controls), tag-jumping, and poor-quality sequences, a total of 61,721 and 555,017 unique ITS2 and COI sequences, respectively, were removed (Appendices 1 and 2). After taxonomic filtering and aggregating, we identified a total of 72 dietary taxa across the 80 Hawfinch samples. From the 55 samples (Denmark n = 33; Germany n = 22) that produced plant DNA, 35 plant taxa, of which 86% could be resolved to species, and 14% to genus, were detected. The same 80 samples were tested for invertebrate DNA. From the 25 samples (Denmark n = 11; Germany n = 14) that produced invertebrate taxa, a total of 37 taxa were identified; 84% to species, 8% to genus, and 8% to family.
The calculated dietary diversity revealed that species richness (Hill-richness) provided the highest diversity estimate compared to Shannon’s entropy (Hill-Shannon), and Simpson’s index (Hill-Simpson; Fig. 2). These diversity estimates indicate Hawfinch consume numerous rarely eaten individual taxa, as opposed to focusing on a few commonly eaten, or evenly consuming dietary taxa. We estimated that our sampling effort provided a dietary coverage of 71.8% (±95% CI: 27.3%) for Danish samples and 76.6% (±95% CI: 18.1%) for German samples (Figs. 3 and 4). This indicates 28.2% and 23.4% of species in the total theoretical diet were from undetected species in Denmark and Germany.
Of the plant dietary items detected, the most frequently occurring were sunflower seed (Helianthus sp.), beech, and English oak (Quercus robur) (detected in 38.2%, 25.5%, and 20% of samples, respectively; n = 55). All herbivorous dietary items detected are listed in Table 1. Within Denmark, a total of 18 taxa were detected, with sunflower, beech, and downy birch (Betula pubescens) the most frequently detected taxa (54.5%, 36.4%, and 15.2%, respectively, n = 33). Within Germany, 29 plant taxa were detected, with hornbeam, English oak, and North American native red oak (Quercus rubra) having the highest prevalence (40.9%, 36.4%, and 22.7%, respectively, n = 22). Sequences belonging to the genus Quercus were detected in 31.8% of Hawfinch sampled from Germany, however these could not be identified to a lower taxonomic classification.
MGLM analysis revealed a significant difference in plant dietary composition between Denmark and Germany (Wald = 109.3, p = < 0.001; visualized in Fig. 5). Four taxa showed a significant GLM result: Carpinus betulus (Wald = 13.4, p = 0.003), detected in 40.9% of samples from Germany compared with 3% from Denmark. Helianthus sp. (Wald = 10.1, p = 0.016), detected in 54.5% of Danish samples compared with 13.6% from Germany. Quercus rubra (Wald = 9.9, p = 0.017), detected in 22.7% of German samples, while absent from Denmark, and Quercus sp. (Wald = 9.1, p = 0.026), detected in 31.8% of German samples, contrasted with 3% from Denmark (Appendix 3). We ran the same analysis with supplementary feed removed from the dataset, and still detected a significant difference in plant dietary composition between Denmark and Germany (Wald = 99.2, p = < 0.001).
The most frequently detected invertebrate prey taxa across Denmark and Germany were winter moth (Operophtera brumata), satellite moth (Eupsilia transversa), and common quaker (Orthosia cerasi) found in 36%, 20%, and 20% of samples, respectively (n = 25). All invertebrate dietary items detected are listed in Table 2. Across Danish samples (n = 11), a total of 17 invertebrate taxa were detected, with 35.3% of prey orders identified as Lepidoptera, 23.5% Araneae, 17.6% Hymenoptera, and 5.9% Coleoptera, Diptera, Hemiptera, and Neuroptera. The most prevalent detected prey taxa being the birch sawfly (Cimbex femoratus; 27.3%), the sac spider (Clubiona brevipes; 18.2%), and the buzzing spider (Anyphaena accentuata; 9.1%). In German Hawfinch samples (n = 14) a total of 26 invertebrate taxa were detected, dominated by the order Lepidoptera (69.2%), with Hymenoptera (11.5%), Coleoptera (7.7%), Pulmonata (7.7%), and Araneae (3.9%) also present. The most prevalent dietary taxa were the winter moth (64.3%), the small quaker (Orthosia cruda; 35.7%), and the satellite moth (28.6%).
MGLM analysis revealed invertebrate prey taxa within the diet differed significantly between European countries (Wald = 78.1, p = < 0.001; visualized in Fig. 6). One taxon showed a significant GLM result: Operophtera brumata (Wald = 14.4, p = 0.003), detected within 64.3% of German samples, while absent from Hawfinch fecal samples collected from Denmark (Appendix 3).
DISCUSSION
In this study, which determines the diet of European Hawfinch populations for the first time, 35 plant and 37 invertebrate taxa were identified. Our findings suggest Hawfinch are generalist omnivores that consume a wide variety of plant and invertebrate taxa. Furthermore, our diversity estimates imply Hawfinch consume many taxa infrequently, as opposed to consuming taxa evenly. This study attained a higher taxonomic resolution when compared to personal observation studies of Hawfinch diet (Mountford 1957), resolving over 80% of plant and invertebrate taxa to species-level.
Diet differed between Hawfinch populations in Denmark and Germany. This may reflect differences in plant taxa availability within Hawfinch feeding ranges, as a high proportion of Denmark has undergone deforestation, with only 11% forest land cover remaining (Madsen et al. 2005, Stanturf et al. 2018). Modern day forestry practices within Denmark are still heavily reliant on non-native species; with the exception of Scots pine (Pinus sylvestris) all conifer species are non-native (Stanturf et al. 2018). Restored forest landscapes have primarily been built on degraded land, and consist of the highly productive mixed stands of Norway spruce (Picea abies) and Douglas fir (Pseudotsuga menziesii; Stanturf et al. 2018). Norway spruce was detected in 21% of Hawfinch sampled in Denmark, suggesting that Hawfinch can make use of this as a food resource. Widely available food resources may be available within the more heterogeneous woodland environment where the artificial feed sites within Germany were located. Within Europe, Germany is one of the most densely wooded countries, with approximately one third of the landmass forested (Polley et al. 2016). Approximately 73% of German forests consist of mixed stands, however the proportions of tree species differ with variation in natural features and site conditions, as well as historic developments (Polley et al. 2016).
Landscape features may be important drivers of food availability because they have been shown to influence dietary composition and spatial variation in insectivorous bat diet (Tournayre et al. 2021). Artificial feed sites in Denmark were located within urban landscapes, while German feed sites were in heterogeneous woodland. Such human impact on landscapes can have a direct effect on quantity and quality of resources (Chace and Walsh 2006, O’Hanlon et al. 2020), influencing consumer diet, and ultimately survival and reproductive performances of the consumers (White 2008, O’Hanlon et al. 2020). Insect species abundance and richness are heavily influenced by landscape characteristics such as plant species richness or heterogeneity of the landscape (Schuldt et al. 2019). For example, the lepidopteran Operophtera brumata is known to be associated with broadleaved woodlands (Wesołowski and Rowiński 2006) and its presence differed significantly between the diets of Hawfinch in Denmark and Germany. The presence of invertebrate genera such as Operophtera in Hawfinch diet primarily in one sampling country (85% occurrence in fecal samples from German Hawfinch populations) partly supports the spatial variation in dietary composition between countries. Urban environments may seem favorable for birds during winter because of abundant artificial food and scarce natural resources in the wider countryside, however during the breeding season, they may lack sufficient high-quality resources such as carotenoids and amino acids available from caterpillars and spiders (Demeyrier et al. 2017, Jarrett et al. 2020). Whereas specialist foragers may be negatively affected by landscape changes impacting food availability (Millon and Bretagnolle 2008), generalist foragers can buffer these changes by switching to alternative food resources (Schoener 1971, Pyke et al. 1977, O’Hanlon et al. 2020).
Despite sunflower seeds being used across all artificial feeding sites, the higher detection of sunflower in Hawfinch samples from Denmark may be due to the more urbanized locations of the Danish artificial feed sites. Urbanization has impacted natural habitats through altering vegetation composition, resulting in a shift in species community dynamics (Narango et al. 2018, Jarrett et al. 2020). Supplementary feeding is common practice within urban areas, with over half of households in northern Europe providing supplementary feed (Galbraith et al. 2015, Hanmer et al. 2017, Shutt et al. 2021). The supply of continuous and abundant food resources favor euryphagic (broader diet) and granivorous species (Palacio 2020). This could allow Hawfinch to exploit hyperabundant food resources within urban environments, as seen in other generalist species such as the Blue Tit (Cyanistes caeruleus; Shutt et al. 2021). In contrast, forest areas had little supplementary feeding in the landscape surrounding the sampling sites.
It is important to monitor the availability of anthropogenic food resources because this can lead to changes in diet, body condition (Auman et al. 2008), productivity, (Plummer et al. 2013), and population size (Duhem et al. 2008). Some of these impacts can be beneficial, such as reduced energy expenditure, increased body condition, and increased breeding performance (Auman et al. 2008, Flack et al. 2016). However, if the diet shifts toward food resources of poorer quality, this can cause nutritional stress (Will et al. 2015), reduce both adult and fledgling body mass, (Österblom et al. 2006), and may be linked to population declines (Kitaysky et al. 2006). Supplementary food is predominantly used by birds to enhance survival when natural food supplies are reduced (Lawson et al. 2018). However, there is a risk that birds may become reliant on artificial food sources, as well as increasing disease transmission risk (Murray et al. 2016, Lawson et al. 2018, Støstad et al. 2019). Evidence suggests that high congregation densities of birds over a prolonged period of time, coupled with poor hygiene, can result in pathogen contamination of feeding stations (Hanmer et al. 2017, Lawson et al. 2018). Additionally, Støstad et al. (2019) found evidence that a high intake of sunflower seed can negatively impact sperm quality of Greenfinches (Chloris chloris) and Hawfinches because sunflower seeds contain high levels of linoleic acid, which in high levels can damage the cell membrane of sperm cells. It is therefore vital to manage the volume and type of supplemental feed provided. For this study, artificial feeding sites were unavoidable for sample collection. Encouraging Hawfinch to feed in flocks on the ground was the only viable method to enable mist net capture.
An important aspect to consider within any DNA metabarcoding study is detection biases, which can impact the results and subsequent ecological interpretation of metabarcoding studies (Forsman et al. 2022). The invertebrate primer pair used in this study were originally used to characterize the diet of Blackbirds (Turdus merula) and Song Thrush (Turdus philomelos), and subsequently were designed to amplify a broad range of invertebrate taxa, including Lepidoptera (Stockdale 2018). Therefore, the absence of green oak moth (Tortrix viridana; previously described as a frequently consumed taxa in Mountford 1957) in the diet is likely related to availability of the green oak moth within Hawfinch foraging environment, rather than a result of a false negative. It is important to note however, that no primer pair can provide a completely unbiased and comprehensive account of species’ diet because of highly degraded DNA failing to amplify in PCR reactions, primer biases, and differences in mitochondrial copy number per cell (reviewed in Clare 2014). A one-locus-several-primer approach should be used more readily within DNA metabarcoding studies in order to maximize taxonomic coverage and minimize false negatives (Corse et al. 2019).
It is important to note that these conclusions are based upon a small number of individual field sites within Denmark (n = 2) and Germany (n = 4), such that inferences about large-scale variation in diet are somewhat speculative. To increase the spatial coverage shown within this study, future work should incorporate an increased number of field sites across each sampling country, so that Hawfinch populations are better represented across them. Because most Hawfinch fecal samples were collected during the winter and spring months, our results only represent a snapshot of Hawfinch dietary habits. The lower number of samples testing positive for invertebrate DNA within the diet may be a result of the time of sampling. A significant number of fecal samples were collected between January and March (n = 19) when seasonal invertebrate activity is lower (Driessen et al. 2013).
This study provides the first detailed insight into the diet of mainland European Hawfinch populations, and further strengthens the use of high-throughput sequencing in utilizing metabarcoding to infer omnivorous dietary information (Robeson et al. 2018, da Silva et al. 2020, Tercel et al. 2022). This study also shows that identifying the drivers of dietary differentiation within and between populations is important in our understanding of how species adjust to fluctuating environmental conditions.
RESPONSES TO THIS ARTICLE
Responses to this article are invited. If accepted for publication, your response will be hyperlinked to the article. To submit a response, follow this link. To read responses already accepted, follow this link.
AUTHOR CONTRIBUTIONS
POTW, WOS, WBK, IPV, and PEB conceived the idea and designed the formulated hypothesis; EHS collected all data, undertook all laboratory work, and analyzed the data; EHS wrote the manuscript and all authors contributed to the editing.
ACKNOWLEDGMENTS
We thank Lars Rasmussen, Jens Muff Hansen, Rolf Hennes, and Reinhard Vohwinkel for assistance with Hawfinch faecal sample collection from Denmark and Germany, respectively. We also thank Angela Marchbank and Trudy Workman of the Cardiff University Genomics Hub for assistance with high-throughput sequencing. We thank Sarah Davies for training and advice in bioinformatics. We thank the anonymous reviewers for their insightful and helpful comments.
EHS was supported by a NERC GW4+ Doctoral Training Partnership studentship from the Natural Environment Research Council (grant NE/L002434/1) and is thankful for the support and additional funding from CASE partner the Royal Society for the Protection of Birds (grant 210-x-1656). The authors thank The Genetics Society for providing additional funding through a Hereditary Fieldwork Grant.
For the purpose of open access, the author has applied a CC BY public copyright licence (where permitted by UKRI, “Open Government Licence” or “CC BY-ND public copyright licence” may be stated instead) to any Author Accepted Manuscript version arising.
DATA AVAILABILITY
The data/code that support the findings of this study are openly available in Dryad at https://datadryad.org/stash/share/b2ALe0lHUnF0pvqcR72MXSwDnS91CCPERb21mcW3ppc, DOI https://doi.org/10.5061/dryad.44j0zpchw
LITERATURE CITED
Ando, H., S. Setsuko, K. Horikoshi, H. Suzuki, S. Umehara, M. Inoue-Murayama, and Y. Isagi. 2013. Diet analysis by next-generation sequencing indicates the frequent consumption of introduced plants by the critically endangered Red-headed Wood Pigeon (Columba janthina nitens) in oceanic island habitats. Ecology and Evolution 3:4057-4069. https://doi.org/10.1002/ece3.773
Auman, H. J., C. E. Meathrel, and A. Richardson. 2008. Supersize Me: does anthropogenic food change the body condition of Silver Gulls? A comparison between urbanized and remote, non-urbanized areas. Waterbirds 31:122-126. https://doi.org/10.1675/1524-4695(2008)31[122:SMDAFC]2.0.CO;2
Barnagaud, J.-Y., W. Daniel Kissling, B. Sandel, W. L. Eiserhardt, Ç. H. Şekercioğlu, B. J. Enquist, C. Tsirogiannis, and J.-C. Svenning. 2014. Ecological traits influence the phylogenetic structure of bird species co-occurrences worldwide. Ecology Letters 17:811-820. https://doi.org/10.1111/ele.12285
Barnagaud, J.-Y., N. Mazet, F. Munoz, M. Grenié, P. Denelle, M. Sobral, W. D. Kissling, Ç. H. Şekercioğlu, and C. Violle. 2019. Functional biogeography of dietary strategies in birds. Global Ecology and Biogeography 28:1004-1017. https://doi.org/10.1111/geb.12910
Bates, D., M. Mächler, B. Bolker, and S. Walker. 2015. Fitting linear mixed-effects models using lme4. Journal of Statistical Software 67:1-48. https://doi.org/10.18637/jss.v067.i01
Bijlsma, R. G. 1998. Breeding biology and population trend of Hawfinches Coccothraustes coccothraustes in Flevoland. Limosa 71:137-148.
Brown, D. S., R. Burger, N. Cole, D. Vencatasamy, E. L. Clare, A. Montazam, and W. O. C. Symondson. 2014. Dietary competition between the alien Asian Musk Shrew (Suncus murinus) and a re-introduced population of Telfair’s Skink (Leiolopisma telfairii). Molecular Ecology 23:3695-3705. https://doi.org/10.1111/mec.12445
Burin, G., W. D. Kissling, P. R. Guimarães, Ç. H. Şekercioğlu, and T. B. Quental. 2016. Omnivory in birds is a macroevolutionary sink. Nature Communications 7:11250. https://doi.org/10.1038/ncomms11250
Callahan, B. J., P. J. McMurdie, and S. P. Holmes. 2017. Exact sequence variants should replace operational taxonomic units in marker-gene data analysis. ISME Journal 11:2639-2643. https://doi.org/10.1038/ismej.2017.119
Camacho, C., G. Coulouris, V. Avagyan, N. Ma, J. Papadopoulos, K. Bealer, and T. L. Madden. 2009. BLAST+: architecture and applications. BMC Bioinformatics 10:421. https://doi.org/10.1186/1471-2105-10-421
Chace, J. F., and J. J. Walsh. 2006. Urban effects on native avifauna: a review. Landscape and Urban Planning 74:46-69. https://doi.org/10.1016/j.landurbplan.2004.08.007
Chao, A., and L. Jost. 2012. Coverage-based rarefaction and extrapolation: standardizing samples by completeness rather than size. Ecology 93:2533-2547. https://doi.org/10.1890/11-1952.1
Chen, S., Y. Zhou, Y. Chen, and J. Gu. 2018. Fastp: an ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 34:884-890. https://doi.org/10.1093/bioinformatics/bty560
Clare, E. L. 2014. Molecular detection of trophic interactions: emerging trends, distinct advantages, significant considerations and conservation applications. Evolutionary Applications 7:1144-1157. https://doi.org/10.1111/eva.12225
Coogan, S. C. P., D. Raubenheimer, S. P. Zantis, and G. E. Machovsky-Capuska. 2018. Multidimensional nutritional ecology and urban birds. Ecosphere 9:e02177. https://doi.org/10.1002/ecs2.2177
Corse, E., C. Tougard, G. Archambaud-Suard, J.-F. Agnèse, F. D. Messu Mandeng, C. F. Bilong Bilong, D. Duneau, L. Zinger, R. Chappaz, C. C. Y. Xu, E. Meglécz, and V. Dubut. 2019. One-locus-several-primers: a strategy to improve the taxonomic and haplotypic coverage in diet metabarcoding studies. Ecology and Evolution 9:4603-4620. https://doi.org/10.1002/ece3.5063
Czenze, Z. J., J. L. Tucker, E. L. Clare, J. E. Littlefair, D. Hemprich-Bennett, H. F. M. Oliveira, R. M. Brigham, A. J. R. Hickey, and S. Parsons. 2018. Spatiotemporal and demographic variation in the diet of New Zealand lesser short-tailed bats (Mystacina tuberculata). Ecology and Evolution 8:7599-7610. https://doi.org/10.1002/ece3.4268
da Silva, L. P., V. A. Mata, P. B. Lopes, R. J. Lopes, and P. Beja. 2020. High-resolution multi-marker DNA metabarcoding reveals sexual dietary differentiation in a bird with minor dimorphism. Ecology and Evolution 10:10364-10373. https://doi.org/10.1002/ece3.6687
Davies, S. R., I. P. Vaughan, R. J. Thomas, L. E. Drake, A. Marchbank, and W. O. C. Symondson. 2022. Seasonal and ontological variation in diet and age-related differences in prey choice, by an insectivorous songbird. Ecology and Evolution 12:e9180. https://doi.org/10.1002/ece3.9180
Demeyrier, V., A. Charmantier, M. M. Lambrechts, and A. Grégoire. 2017. Disentangling drivers of reproductive performance in urban Great Tits: a food supplementation experiment. Journal of Experimental Biology 220:4195-4203. https://doi.org/10.1242/jeb.161067
Drake, L. E., J. P. Cuff, R. E. Young, A. Marchbank, E. A. Chadwick, and W. O. C. Symondson. 2021. An assessment of minimum sequence copy thresholds for identifying and reducing the prevalence of artefacts in dietary metabarcoding data. Methods in Ecology and Evolution 13:694-710. https://doi.org/10.1111/2041-210X.13780
Driessen, M. M., J. B. Kirkpatrick, and P. B. Mcquillan. 2013. Shifts in composition of monthly invertebrate assemblages in moorland differed between lowland and montane locations but not fire-ages. Environmental Entomology 42:58-73. https://doi.org/10.1603/EN12322
Duhem, C., P. Roche, E. Vidal, and T. Tatoni. 2008. Effects of anthropogenic food resources on Yellow-legged Gull colony size on Mediterranean islands. Population Ecology 50:91-100. https://doi.org/10.1007/s10144-007-0059-z
Dunbar, M. B., and R. M. Brigham. 2010. Thermoregulatory variation among populations of bats along a latitudinal gradient. Journal of Comparative Physiology B 180:885-893. https://doi.org/10.1007/s00360-010-0457-y
Dunn, J. C., J. E. Stockdale, R. J. Moorhouse-Gann, A. McCubbin, H. Hipperson, A. J. Morris, P. V. Grice, and W. O. C. Symondson. 2018. The decline of the Turtle Dove: dietary associations with body condition and competition with other columbids analysed using high-throughput sequencing. Molecular Ecology 27:3386-3407. https://doi.org/10.1111/mec.14766
Evans, B. A., and D. E. Gawlik. 2020. Urban food subsidies reduce natural food limitations and reproductive costs for a wetland bird. Scientific Reports 10:14021. https://doi.org/10.1038/s41598-020-70934-x
Fernandes, S., V. A. Mata, and L. P. da Silva. 2023. Feeding ecology of a highly aerial bird during its long breeding season. Avian Research 14:100073. https://doi.org/10.1016/j.avrs.2022.100073
Flack, A., W. Fiedler, J. Blas, I. Pokrovsky, M. Kaatz, M. Mitropolsky, K. Aghababyan, I. Fakriadis, E. Makrigianni, L. Jerzak, H. Azafzaf, C. Feltrup-Azafzaf, S. Rotics, T. M. Mokotjomela, R. Nathan, and M. Wikelski. 2016. Costs of migratory decisions: a comparison across eight white stork populations. Science Advances 2:e1500931. https://doi.org/10.1126/sciadv.1500931
Fleischer, A. L., R. Bowman, and G. E. Woolfenden. 2003. Variation in foraging behavior, diet, and time of breeding of Florida Scrub-Jays in suburban and wildland habitats. Condor 105:515-527. https://doi.org/10.1093/condor/105.3.515
Forsman, A. M., B. D. Hoenig, S. A. Gaspar, J. D. Fischer, J. Siegrist, and K. Fraser. 2022. Evaluating the impacts of metabarcoding primer selection on DNA characterization of diet in an aerial insectivore, the Purple Martin. Ornithology 139:ukab075. https://doi.org/10.1093/ornithology/ukab075
Galbraith, J. A., J. R. Beggs, D. N. Jones, and M. C. Stanley. 2015. Supplementary feeding restructures urban bird communities. Proceedings of the National Academy of Sciences 112:2648-2657. https://doi.org/10.1073/pnas.1501489112
Galimberti, A., S. Spinelli, A. Bruno, V. Mezzasalma, F. De Mattia, P. Cortis, and M. Labra. 2016. Evaluating the efficacy of restoration plantings through DNA barcoding of frugivorous bird diets. Conservation Biology 30:763-773. https://doi.org/10.1111/cobi.12687
Hanmer, H. J., R. L. Thomas, and M. D. E. Fellowes. 2017. Provision of supplementary food for wild birds may increase the risk of local nest predation. Ibis 159:158-167. https://doi.org/10.1111/ibi.12432
Hoenig, B. D., A. M. Snider, A. M. Forsman, K. A. Hobson, S. C. Latta, E. T. Miller, M. J. Polito, L. L. Powell, S. L. Rogers, T. W. Sherry, D. P. L. Toews, A. J. Welch, S. S. Taylor, and B. A. Porter. 2022. Current methods and future directions in avian diet analysis. Ornithology 139:ukab077. https://doi.org/10.1093/ornithology/ukab077
Hsieh, T. C., K. H. Ma, and A. Chao. 2016. iNEXT: an R package for rarefaction and extrapolation of species diversity (Hill numbers). Methods in Ecology and Evolution 7:1451-1456. https://doi.org/10.1111/2041-210X.12613
Huson, D. H., S. Beier, I. Flade, A. Górska, M. El-Hadidi, S. Mitra, H. J. Ruscheweyh, and R. Tappu. 2016. MEGAN Community Edition - interactive exploration and analysis of large-scale microbiome sequencing data. PLoS Computational Biology 12(6):e100495. https://doi.org/10.1371/journal.pcbi.1004957
Jarrett, C., L. L. Powell, H. McDevitt, B. Helm, and A. J. Welch. 2020. Bitter fruits of hard labour: diet metabarcoding and telemetry reveal that urban songbirds travel further for lower-quality food. Oecologia 193:377-388. https://doi.org/10.1007/s00442-020-04678-w
Kirby, W. B., A. J. Stanbury, J. Lewis, D. L. Smith, A. V. Cross, P. V. Grice, and P. E. Bellamy. 2018. Nest survival, causes of failure and productivity of British Hawfinches Coccothraustes coccothraustes. Bird Study 65:279-289. https://doi.org/10.1080/00063657.2018.1506424
Kissling, W. D., C. H. Sekercioglu, and W. Jetz. 2012. Bird dietary guild richness across latitudes, environments and biogeographic regions. Global Ecology and Biogeography 21:328-340. https://doi.org/10.1111/j.1466-8238.2011.00679.x
Kitaysky, A. S., E. V Kitaiskaia, J. F. Piatt, and J. C. Wingfield. 2006. A mechanistic link between chick diet and decline in seabirds? Proceedings of the Royal Society B: Biological Sciences 273:445-450. https://doi.org/10.1098/rspb.2005.3351
Lamb, P. D., E. Hunter, J. K. Pinnegar, S. Creer, R. G. Davies, and M. I. Taylor. 2019. How quantitative is metabarcoding: a meta-analytical approach. Molecular Ecology 28:420-430. https://doi.org/10.1111/mec.14920
Lawson, B., R. A. Robinson, M. P. Toms, K. Risely, S. MacDonald, and A. A. Cunningham. 2018. Health hazards to wild birds and risk factors associated with anthropogenic food provisioning. Philosophical Transactions of the Royal Society B: Biological Sciences 373:e20170091. https://doi.org/10.1098/rstb.2017.0091
Leray, M., J. Y. Yang, C. P. Meyer, S. C. Mills, N. Agudelo, V. Ranwez, J. T. Boehm, and R. J. Machida. 2013. A new versatile primer set targeting a short fragment of the mitochondrial COI region for metabarcoding metazoan diversity: application for characterizing coral reef fish gut contents. Frontiers in Zoology 10:34. https://doi.org/10.1186/1742-9994-10-34
MacArthur, R. H., and E. R. Pianka. 1966. On optimal use of a patchy environment. American Naturalist 100:603-609. https://doi.org/10.1086/282454
Madsen, P., F. A. Jensen, and S. Fodgaard. 2005. Afforestation in Denmark. Pages 211-224 in J. A. Stanturf, editor. Restoration of boreal and temperate forests. CRC Press, Boca Raton, Florida, USA.
Millon, A., and V. Bretagnolle. 2008. Predator population dynamics under a cyclic prey regime: numerical responses, demographic parameters and growth rates. Oikos 117:1500-1510. https://doi.org/10.1111/j.0030-1299.2008.16458.x
Moorhouse-Gann, R. J., J. C. Dunn, N. de Vere, M. Goder, N. Cole, H. Hipperson, and W. O. C. Symondson. 2018. New universal ITS2 primers for high-resolution herbivory analyses using DNA metabarcoding in both tropical and temperate zones. Scientific Reports 8:8542. https://doi.org/10.1038/s41598-018-26648-2
Moorhouse-Gann, R. J., E. F. Kean, G. Parry, S. Valladares, and E. A. Chadwick. 2020. Dietary complexity and hidden costs of prey switching in a generalist top predator. Ecology and Evolution 10:6395-6408. https://doi.org/10.1002/ece3.6375
Mountford, G. 1957. The Hawfinch. Collins, London, UK.
Murray, M. H., D. J. Becker, R. J. Hall, and S. M. Hernandez. 2016. Wildlife health and supplemental feeding: a review and management recommendations. Biological Conservation 204:163-174. https://doi.org/10.1016/j.biocon.2016.10.034
Narango, D. L., D. W. Tallamy, and P. P. Marra. 2018. Nonnative plants reduce population growth of an insectivorous bird. Proceedings of the National Academy of Sciences 115:11549-11554. https://doi.org/10.1073/pnas.1809259115
Newton, I. 1967. The adaptive radiation and feeding ecology of some British finches. Ibis 109:33-96. https://doi.org/10.1111/j.1474-919X.1967.tb00005.x
O’Hanlon, N. J., S. Alonso, J. A. O. Miller, R. A. R. McGill, and R. G. Nager. 2020. Landscape-mediated variation in diet is associated with egg size and maculation in a generalist forager. Ibis 162:687-700. https://doi.org/10.1111/ibi.12739
Oksanen, J., F. G. Blanchet, M. Friendly, R. Kindt, P. Legendre, D. McGlinn, P. R. Minchin, R. B. O’Hara, G. L. Simpson, P. Solymos, M. H. H. Stevens, E. Szoecs, and H. Wagner. 2019. vegan: Community ecology package. R package version 2.5-6.
Österblom, H., M. Casini, O. Olsson, and A. Bignert. 2006. Fish, seabirds and trophic cascades in the Baltic Sea. Marine Ecology Progress Series 323:233-238. https://doi.org/10.3354/meps323233
Palacio, F. X. 2020. Urban exploiters have broader dietary niches than urban avoiders. Ibis 162:42-49. https://doi.org/10.1111/ibi.12732
Pan-European Common Bird Monitoring Scheme (PECBMS). 2019. Population trends of common European breeding birds 2016. Česká Společnost Ornitologická, Prague, Czech Republic.
Plummer, K., S. Bearhop, D. I. Leech, D. E. Chamberlain, and J. D. Blount. 2013. Winter food provisioning reduces future breeding performance in a wild bird. Scientific Reports 3:2002. https://doi.org/10.1038/srep02002
Polley, H., P. Hennig, F. Kroiher, A. Marks, T. Riedel, U. Schmidt, F. Schwitzgebel, and T. Stauber. 2016. The forests in Germany. Federal Ministry of Food and Agriculture, Berlin, Germany.
Pyke, G. H., H. R. Pulliam, and E. L. Charnov. 1977. Optimal foraging: a selective review of theory and tests. Quarterly Review of Biology 52:137-154. https://doi.org/10.1086/409852
QGIS Development Team. 2021. QGIS Geographic Information System. Open Source Geospatial Foundation Project. http://qgis.osgeo.org
R Core Team. 2020. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria.
Robeson, M. S., K. Khanipov, G. Golovko, S. M. Wisely, M. D. White, M. Bodenchuck, T. J. Smyser, Y. Fofanov, N. Fierer, and A. J. Piaggio. 2018. Assessing the utility of metabarcoding for diet analyses of the omnivorous wild pig (Sus scrofa). Ecology and Evolution 8:185-196. https://doi.org/10.1002/ece3.3638
Romano, A., R. Séchaud, and A. Roulin. 2020. Global biogeographical patterns in the diet of a cosmopolitan avian predator. Journal of Biogeography 47:1467-1481. https://doi.org/10.1111/jbi.13829
Roswell, M., J. Dushoff, and R. Winfree. 2021. A conceptual guide to measuring species diversity. Oikos 130:321-338. https://doi.org/10.1111/oik.07202
Schloss, P. D., S. L. Westcott, T. Ryabin, J. R. Hall, M. Hartmann, E. B. Hollister, R. A. Lesniewski, B. B. Oakley, D. H. Parks, C. J. Robinson, J. W. Sahl, B. Stres, G. G. Thallinger, D. J. Van Horn, and C. F. Weber. 2009. Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Applied and Environmental Microbiology 75:7537-7541. https://doi.org/10.1128/AEM.01541-09
Schnell, I. B., K. Bohmann, and M. T. P. Gilbert. 2015. Tag jumps illuminated - reducing sequence-to-sample misidentifications in metabarcoding studies. Molecular Ecology Resources 15:1289-1303. https://doi.org/10.1111/1755-0998.12402
Schoener, T. W. 1971. Theory of feeding strategies. Annual Review of Ecology and Systematics 2:369-404. https://doi.org/10.1146/annurev.es.02.110171.002101
Schuldt, A., A. Ebeling, M. Kunz, M. Staab, C. Guimarães-Steinicke, D. Bachmann, N. Buchmann, W. Durka, A. Fichtner, F. Fornoff, W. Härdtle, L. R. Hertzog, A.-M. Klein, C. Roscher, J. Schaller, G. von Oheimb, A. Weigelt, W. Weisser, C. Wirth, J. Zhang, H. Bruelheide, and N. Eisenhauer. 2019. Multiple plant diversity components drive consumer communities across ecosystems. Nature Communications 10:1460. https://doi.org/10.1038/s41467-019-09448-8
Shutt, J. D., U. H. Trivedi, and J. A. Nicholls. 2021. Fecal metabarcoding reveals pervasive long-distance impacts of garden bird feeding. Proceedings of the Royal Society B: Biological Sciences 288:20210480. https://doi.org/10.1098/rspb.2021.0480
Sibly, R. M., C. C. Witt, N. A. Wright, C. Venditti, W. Jetz, and J. H. Brown. 2012. Energetics, lifestyle, and reproduction in birds. Proceedings of the National Academy of Sciences 109:10937-10941. https://doi.org/10.1073/pnas.1206512109
Simon, L., M. Lalonde, and T. D. Bruns. 1992. Specific amplification of 18S fungal ribosomal genes from vesicular-arbuscular endomycorrhizal fungi colonizing roots. Applied and Environmental Microbiology 58:291-295. https://doi.org/10.1128/aem.58.1.291-295.1992
Smith, G. C., and N. Carlile. 1993. Food and feeding ecology of breeding Silver Gulls (Larus novaehollandiae) in urban Australia. Colonial Waterbirds 16:9-16. https://doi.org/10.2307/1521551
Sol, D., C. González-Lagos, D. Moreira, J. Maspons, and O. Lapiedra. 2014. Urbanisation tolerance and the loss of avian diversity. Ecology Letters 17:942-950. https://doi.org/10.1111/ele.12297
Stanturf, J. A., P. Madsen, K. Sagheb-Talebi, and O. K. Hansen. 2018. Transformational restoration: novel ecosystems in Denmark. Plant Biosystems - An International Journal Dealing with all Aspects of Plant Biology 152:536-546. https://doi.org/10.1080/11263504.2018.1435586
Stawski, C., and F. Geiser. 2011. Do season and distribution affect thermal energetics of a hibernating bat endemic to the tropics and subtropics? American Journal of Physiology-Regulatory, Integrative and Comparative Physiology 301:R542-R547. https://doi.org/10.1152/ajpregu.00792.2010
Stockdale, J. E. 2018. Using high-throughput sequencing to track habitat use by thrushes exploiting heterogeneous farmland landscapes. Dissertation. Cardiff University, Cardiff, UK. https://orca.cardiff.ac.uk/id/eprint/122957/
Støstad, H. N., M. Rowe, A. Johnsen, O. Tomášek, T. Albrecht, and J. T. Lifjeld. 2019. Sperm head abnormalities are associated with excessive omega‐6 fatty acids in two finch species feeding on sunflower seeds. Journl of Avian Biology 50:e02056. https://doi.org/10.1111/jav.02056
Taberlet, P., A. Bonin, L. Zinger, and E. Coissac. 2018. Environmental DNA: for biodiversity research and monitoring. Oxford University Press, Oxford, UK. https://doi.org/10.1093/oso/9780198767220.001.0001
Tercel, M. P. T. G., R. J. Moorhouse-Gann, J. P. Cuff, L. E. Drake, N. C. Cole, M. Goder, R. Mootoocurpen, and W. O. C. Symondson. 2022. DNA metabarcoding reveals introduced species predominate in the diet of a threatened endemic omnivore, Telfair’s skink (Leiolopisma telfairii). Ecology and Evolution 12:e8484. https://doi.org/10.1002/ece3.8484
Terraube, J., and B. Arroyo. 2011. Factors influencing diet variation in a generalist predator across its range distribution. Biodiversity and Conservation 20:2111-2131. https://doi.org/10.1007/s10531-011-0077-1
Tomiałojc, L. 2005. Distribution, breeding density and nest sites of Hawfinches Coccothraustes coccothraustes in the primeval forest of Białowieża National Park. Acta Ornithologica 40:127-138. https://doi.org/10.3161/068.040.0208
Tournayre, O., M. Leuchtmann, M. Galan, M. Trillat, S. Piry, D. Pinaud, O. Filippi-Codaccioni, D. Pontier, and N. Charbonnel. 2021. eDNA metabarcoding reveals a core and secondary diets of the greater horseshoe bat with strong spatio-temporal plasticity. Environmental DNA 3:277-296. https://doi.org/10.1002/edn3.167
Wang, Y., U. Naumann, S. T. Wright, and D. Warton. 2012. mvabund - an R package for model-based analysis of multivariate abundance data. Methods in Ecology and Evolution 3:471-474. https://doi.org/10.1111/j.2041-210X.2012.00190.x
Wesołowski, T., and P. Rowiński. 2006. Tree defoliation by winter moth Operophtera brumata L. during an outbreak affected by structure of forest landscape. Forest Ecology and Management 221:299-305. https://doi.org/10.1016/j.foreco.2005.10.023
Westfall, P., and S. Young. 1993. Resampling‐based multiple testing. John Wiley & Sons, New York, New York, USA.
White, T. C. R. 2008. The role of food, weather and climate in limiting the abundance of animals. Biological Reviews 83:227-248. https://doi.org/10.1111/j.1469-185X.2008.00041.x
Wickham, H. 2016. ggplot2: Elegant graphics for data analysis. Springer-Verlagg, Cham, Switzerland. https://doi.org/10.1007/978-3-319-24277-4
Will, A., Y. Watanuki, D. M. Kikuchi, N. Sato, M. Ito, M. Callahan, K. Wynne-Edwards, S. Hatch, K. Elliott, L. Slater, A. Takahashi, and A. Kitaysky. 2015. Feather corticosterone reveals stress associated with dietary changes in a breeding seabird. Ecology and Evolution 5:4221-4232. https://doi.org/10.1002/ece3.1694
Willig, M. R., D. M. Kaufman, and R. D. Stevens. 2003. Latitudinal gradients of biodiversity: pattern, process, scale, and synthesis. Annual Review of Ecology, Evolution, and Systematics 34:273-309. https://doi.org/10.1146/annurev.ecolsys.34.012103.144032
Fig. 1
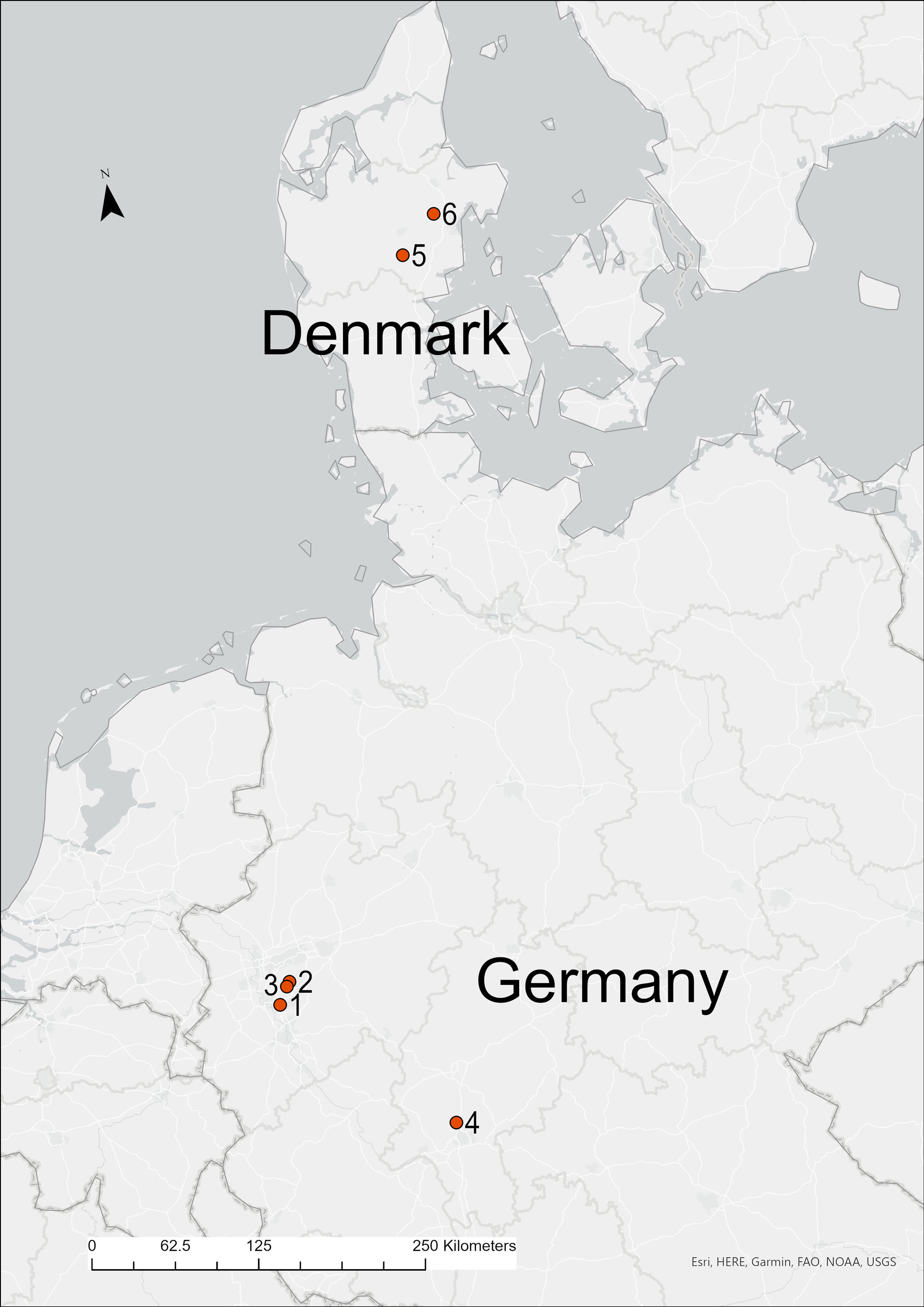
Fig. 1. Location of Danish and German fieldwork sites. Fieldwork sites are numbered 1–6. German fieldwork sites 1–4; 1: Hilden-Haan, 2: Velbert, 3: Wulfrathe-Hohe, and 4: Bad Homburg. Danish fieldwork sites 5–6; 5: Bryup and 6: Solkaer. Map was constructed using QGIS (QGIS Development Team 2021).

Fig. 2
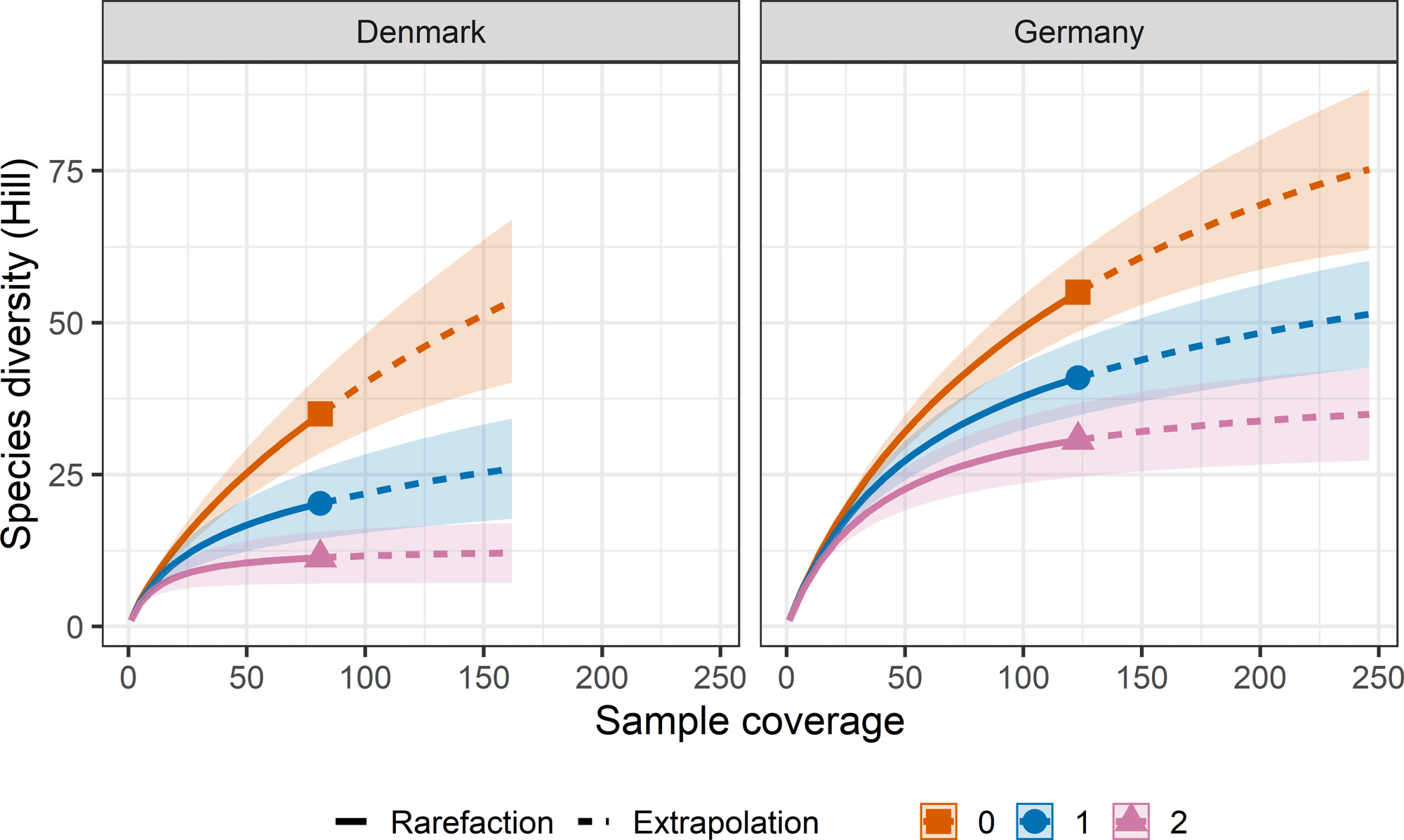
Fig. 2. Species diversity by number of dietary detections found in Hawfinch (Coccothraustes coccothraustes) fecal samples. Line colors determine the different diversity estimates: Species richness = 0, orange line with terminal square; Shannon diversity = 1, blue line with terminal circle; Simpson diversity = 2, purple line with terminal triangle. Solid lines = observed, dashed lines = extrapolated. Confidence intervals (95%) are denoted by shading around the line.

Fig. 3
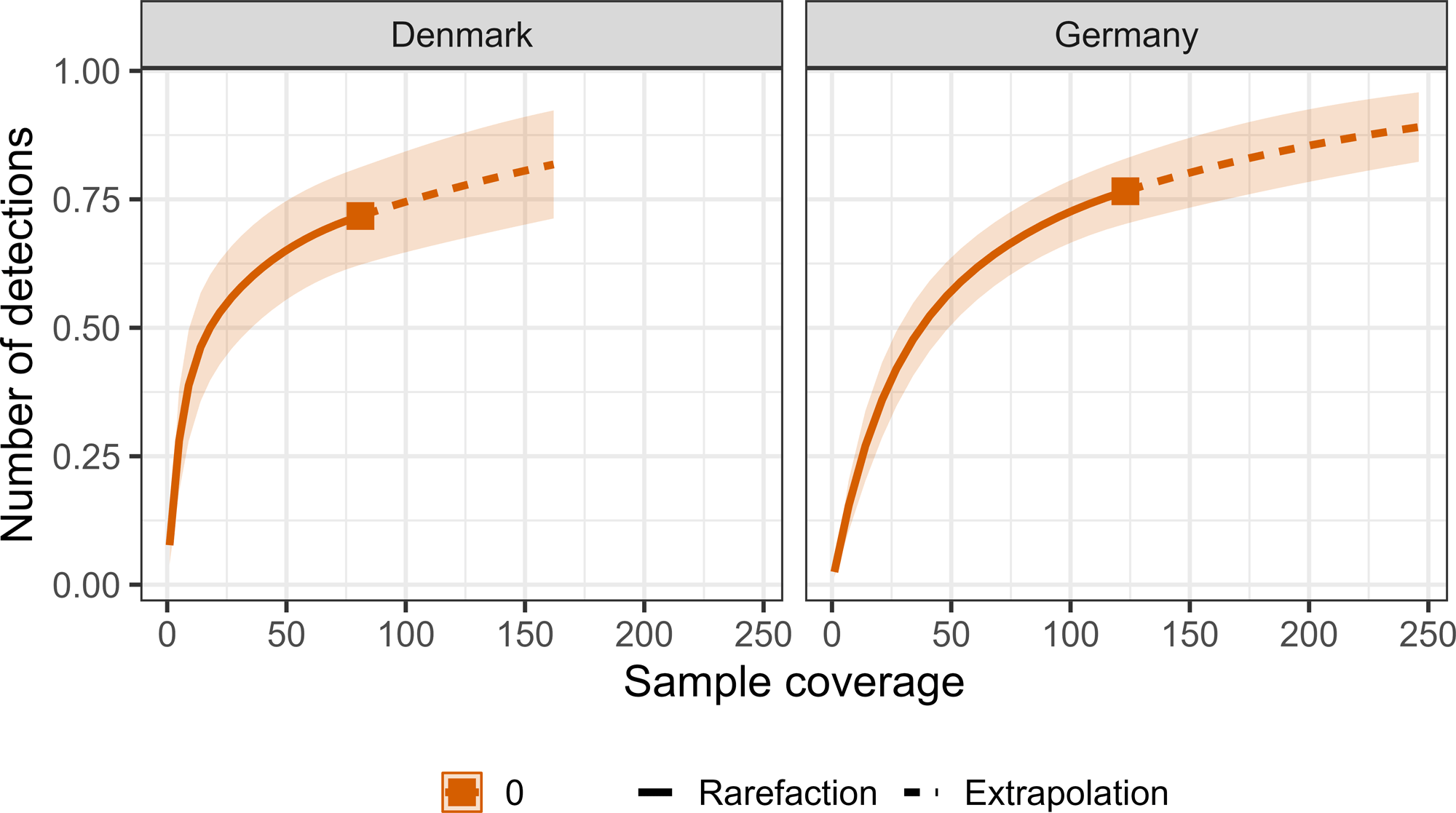
Fig. 3. The level of sample coverage by number of dietary detections in Hawfinch (Coccothraustes coccothraustes) fecal samples. Line color determine the diversity estimates: Species richness = orange line with terminal square. Solid line = observed, dashed line = extrapolated. Confidence intervals (95%) are denoted by shading around the line.

Fig. 4
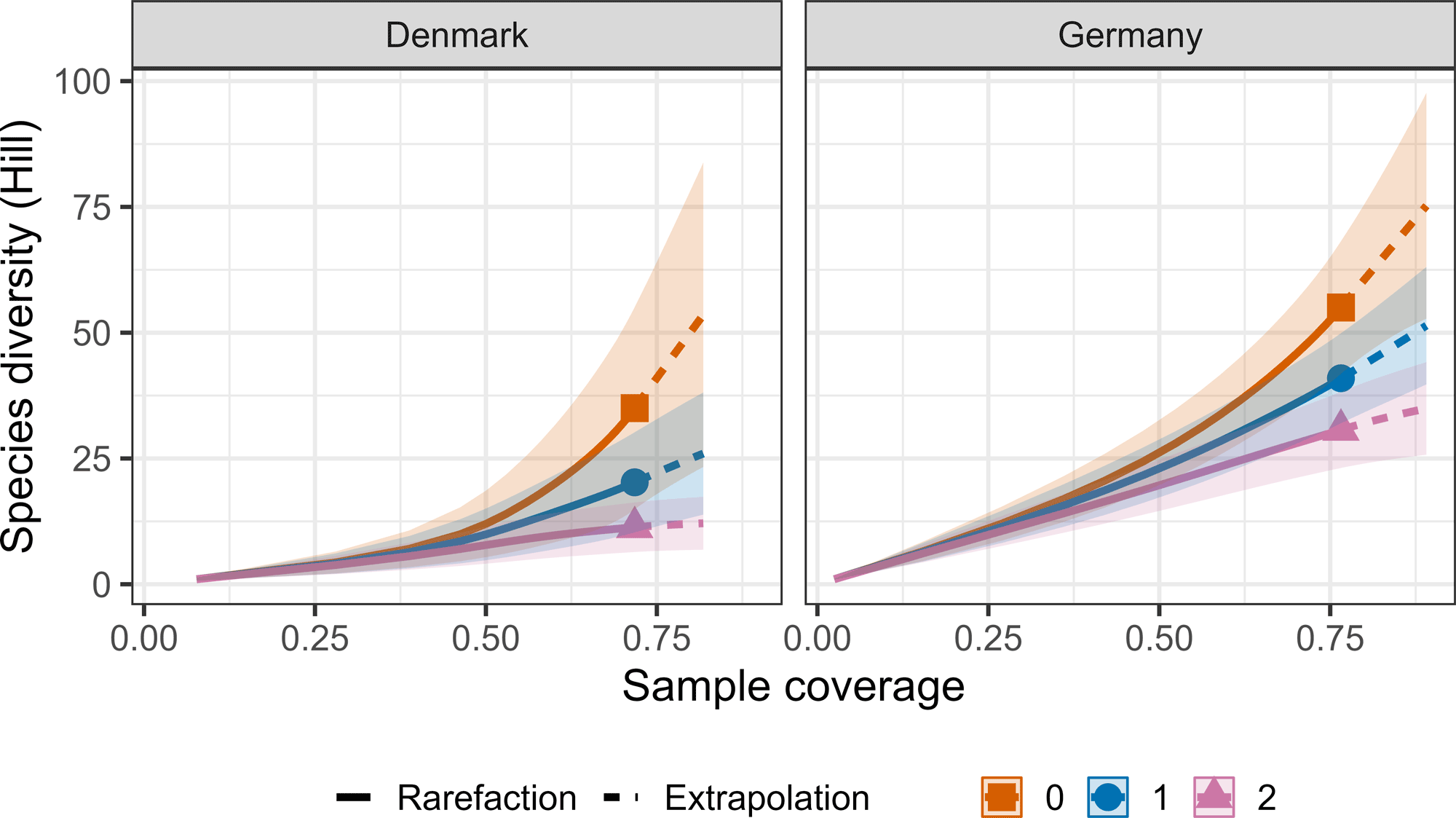
Fig. 4. The level of species diversity by sample coverage in Hawfinch (Coccothraustes coccothraustes) fecal samples. Line colors determine the differing diversity estimates: Species richness = 0, orange line with terminal square; Shannon diversity = 1, blue line with terminal circle; Simpson diversity = 2. Solid lines = observed, dashed lines = extrapolated. Confidence intervals (95%) are denoted by shading around the line.

Fig. 5
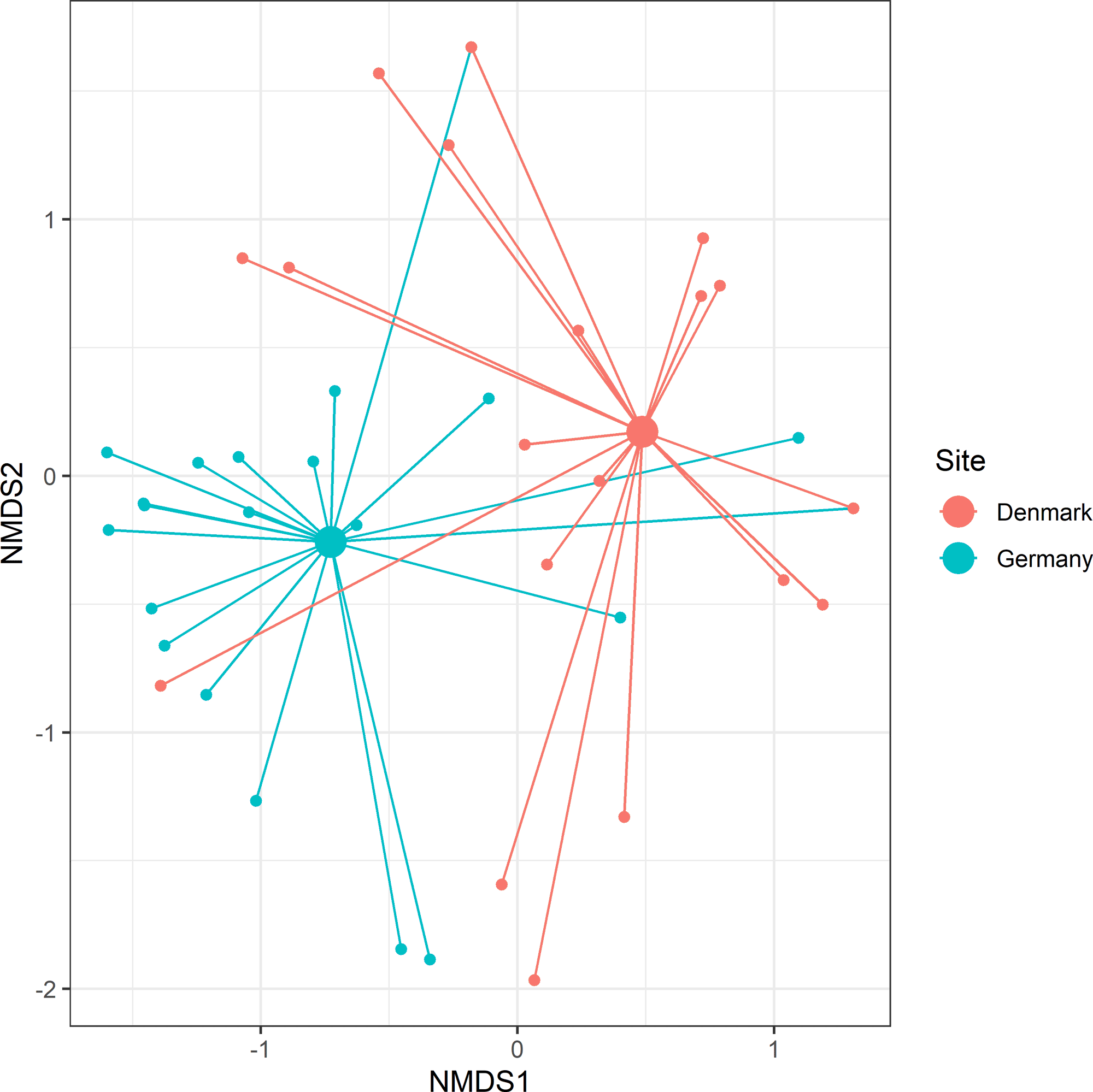
Fig. 5. Spider plot for herbivorous taxa consumed by Hawfinch (Coccothraustes coccothraustes) across European countries. Smaller nodes represent individual Hawfinch with connecting lines joining the individual to the mean centroid (larger nodes) of its region. Stress = 0.17.

Fig. 6
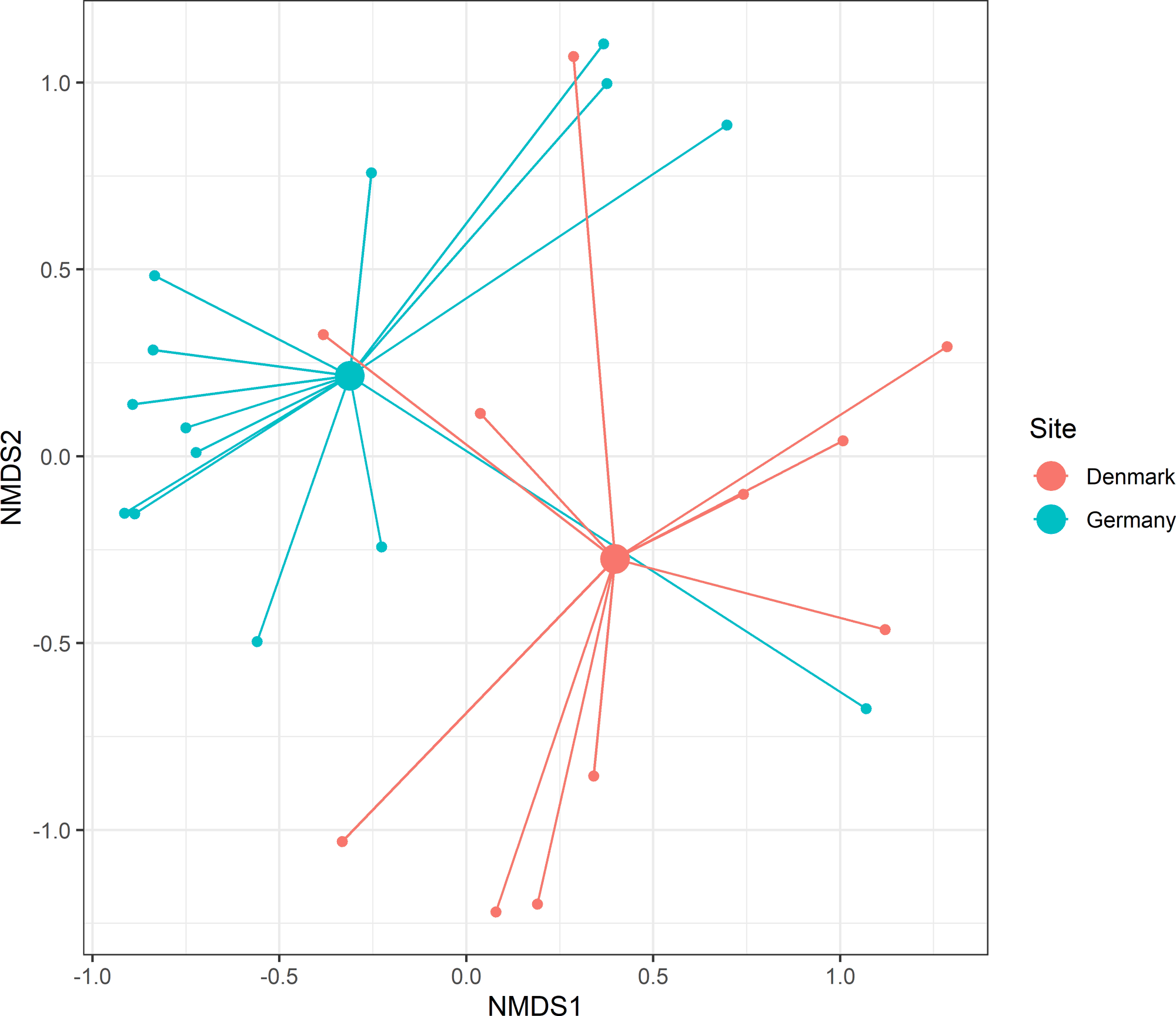
Fig. 6. Spider plot for invertebrate taxa consumed by Hawfinch (Coccothraustes coccothraustes) across European countries. Smaller nodes represent individual Hawfinch with connecting lines joining the individual to the mean centroid (larger nodes) of its region. Stress = 0.20.

Table 1
Table 1. Herbivorous dietary items detected in Hawfinch (Coccothraustes coccothraustes) sampled from Denmark and Germany (n = 55). N is the number of samples collected across all study locations combined, and within individual countries sampled.
| Percentage of samples testing positive for a dietary item | |||
| Taxon | Europe (n = 55) |
Denmark (n = 33) |
Germany (n = 22) |
| Acer pseudoplatanus | 5.5 | 6.1 | 4.5 |
| Aegopodium podagraria | 1.8 | 0 | 4.5 |
| Alnus glutinosa | 5.5 | 0 | 13.6 |
| Betula pendula | 5.5 | 6.1 | 4.5 |
| Betula pubescens | 10.9 | 15.2 | 4.5 |
| Brassica sp. | 9.1 | 12.1 | 4.5 |
| Cardamine bulbifera | 1.8 | 0 | 4.5 |
| Carpinus betulus | 18.2 | 3 | 40.9 |
| Elymus repens | 1.8 | 0 | 4.5 |
| Fagus sylvatica | 25.5 | 36.4 | 9.1 |
| Helianthus sp. | 38.2 | 54.5 | 13.6 |
| Larix decidua | 7.3 | 6.1 | 9.1 |
| Larix sp. | 1.8 | 0 | 4.5 |
| Oxalis acetosella | 1.8 | 3 | 0 |
| Picea abies | 7.3 | 6.1 | 9.1 |
| Pinus sylvestris | 1.8 | 0 | 4.5 |
| Populus nigra | 1.8 | 0 | 4.5 |
| Prunus avium | 10.9 | 9.1 | 13.6 |
| Prunus cerasifera | 1.8 | 3 | 0 |
| Prunus domestica | 1.8 | 3 | 0 |
| Prunus padus | 3.6 | 0 | 9.1 |
| Prunus salicina | 1.8 | 3 | 0 |
| Prunus serotina | 3.6 | 0 | 9.1 |
| Quercus hartwissiana | 3.6 | 0 | 9.1 |
| Quercus petraea | 3.6 | 0 | 9.1 |
| Quercus pyrenaica | 5.5 | 0 | 13.6 |
| Quercus robur | 20 | 9.1 | 36.4 |
| Quercus rubra | 9.1 | 0 | 22.7 |
| Quercus sp. | 14.5 | 3 | 31.8 |
| Salix alba | 1.8 | 0 | 4.5 |
| Salix sp. | 1.8 | 0 | 4.5 |
| Sambucus nigra | 1.8 | 3 | 0 |
| Sinapis alba | 1.8 | 0 | 4.5 |
| Tilia platyphyllos | 1.8 | 0 | 4.5 |
| Ulmus glabra | 1.8 | 3 | 0 |
Table 2
Table 2. Invertebrate dietary items detected in Hawfinch (Coccothraustes coccothraustes) sampled from Denmark and Germany (n = 25). N is the number of samples collected across all study locations combined, and within individual countries sampled.
| Percentage of samples testing positive for a dietary item | |||
| Taxon | Europe (n = 25) |
Denmark (n = 11) |
Germany (n = 14) |
| Acrobasis repandana | 4 | 0 | 7.1 |
| Agathis sp. | 4 | 0 | 7.1 |
| Agrochola macilenta | 4 | 0 | 7.1 |
| Amphipyra berbera | 12 | 0 | 21.4 |
| Amphipyra pyramidea | 12 | 0 | 21.4 |
| Anorthoa munda | 4 | 0 | 7.1 |
| Anyphaena accentuata | 4 | 9.1 | 0 |
| Archips crataeganus | 4 | 0 | 7.1 |
| Cepaea hortensis | 8 | 0 | 14.3 |
| Cimbex femoratus | 16 | 27.3 | 7.1 |
| Clubiona brevipes | 12 | 18.2 | 7.1 |
| Coleophora laricella | 16 | 9.1 | 21.4 |
| Coleophora lutipennella | 4 | 9.1 | 0 |
| Curculionoidae sp. | 4 | 9.1 | 0 |
| Erannis defoliaria | 8 | 0 | 14.3 |
| Eriocrania semipurpurella | 4 | 0 | 7.1 |
| Euchoeca nebulata | 4 | 0 | 7.1 |
| Eupsilia transversa | 20 | 9.1 | 28.6 |
| Formica pratensis | 4 | 9.1 | 0 |
| Galerucella lineola | 4 | 0 | 7.1 |
| Hedya nubiferana | 4 | 9.1 | 0 |
| Hemerobius micans | 4 | 9.1 | 0 |
| Linaeidea aenea | 4 | 0 | 7.1 |
| Nematinus steini | 4 | 0 | 7.1 |
| Noctuoidea sp. | 4 | 0 | 7.1 |
| Operophtera brumata | 36 | 0 | 64.3 |
| Orthosia cerasi | 20 | 9.1 | 28.6 |
| Orthosia cruda | 20 | 0 | 35.7 |
| Orthosia incerta | 8 | 9.1 | 7.1 |
| Pamphiliidae sp. | 4 | 9.1 | 0 |
| Phenacoccus aceris | 4 | 9.1 | 0 |
| Philodromus collinus | 4 | 9.1 | 0 |
| Ptycholoma lecheana | 8 | 0 | 14.3 |
| Smittia sp. | 4 | 9.1 | 0 |
| Succinea sp. | 4 | 0 | 7.1 |
| Tetragnatha obtusa | 4 | 9.1 | 0 |
| Tortricodes alternella | 8 | 0 | 14.3 |








