The following is the established format for referencing this article:
Morrissey, C., and K. Swekla. 2023. Refinements in adipose tissue biopsy collection in shorebirds: effect on pain, wound healing, and mass gain. Journal of Field Ornithology 94(1):10.ABSTRACT
Non-lethal methods to sample adipose tissues from fat depots in small birds are highly valuable as a time integrated sample matrix for ecotoxicology and ecophysiology research. However, for investigators to have confidence to use adipose tissue biopsy methods, welfare concerns remain regarding minimizing pain and ensuring there are no lasting effects on health and survival, particularly for small shorebird species actively undergoing refueling for seasonal migration. We tested refinements in adipose tissue biopsies in a captive Killdeer (Charadrius vociferus) population using either injectable or topical analgesics for pain control and monitored effects on mass, fat, and wound healing time over 21 days to critically evaluate the technique. Injectable analgesics provided rapid and superior short-term pain control compared to topical treatments, and there were no lasting effects of the analgesic or biopsy treatment on healing time, mass, or fat gain over the experiment. Average time for complete healing was 17 ± 3.5 days and all Killdeer continued to gain body mass and fat post procedure. The results suggest that adipose biopsies, with some recommended refinements, should be safe and effective and are not anticipated to cause significant impacts on fueling in migratory shorebirds.
RESUMEN
Los métodos no letales para tomar muestras de tejidos adiposos de depósitos de grasa en aves pequeñas son muy valiosos como matriz de muestras integrada en el tiempo para la investigación en ecotoxicología y ecofisiología. Sin embargo, para que los investigadores utilicen con confianza los métodos de biopsia de tejido adiposo, siguen existiendo problemas de bienestar relacionados con la minimización del dolor y la garantía de que no se produzcan efectos duraderos sobre la salud y la supervivencia, en particular en el caso de especies pequeñas de aves playeras que están activamente repostando para la migración estacional. Se probaron mejoras en las biopsias de tejido adiposo en una población cautiva de Chorlo Tildío (Charadrius vociferus) utilizando analgésicos inyectables o tópicos para controlar el dolor y se monitorearon los efectos sobre la masa, la grasa y el tiempo de cicatrización de la herida durante 21 días para evaluar críticamente la técnica. Los analgésicos inyectables proporcionaron un control rápido y superior del dolor a corto plazo en comparación con los tratamientos tópicos, y no hubo efectos duraderos del tratamiento analgésico o de la biopsia sobre el tiempo de cicatrización, la masa o el aumento de grasa a lo largo del experimento. El tiempo medio para la curación completa fue de 17 ± 3,5 días y todos los Chorlos Tildíos continuaron ganando masa corporal y grasa después del procedimiento. Los resultados sugieren que las biopsias adiposas, con algunos refinamientos recomendados, deberían ser seguras y eficaces y no se prevé que causen impactos significativos en la alimentación de las aves playeras migratorias.
INTRODUCTION
Many long-distance migratory birds face energetic challenges during flight, staging, and upon arriving on the breeding grounds. To fuel the migratory journey, birds will undergo periods of hyperphagia and rapidly accumulate fat to store as fuel. Gram for gram, fats contain 8-10 times more energy than carbohydrates and proteins and are therefore the primary fuel used during migratory flight (Jenni and Jenni-Eiermann 1998, Guglielmo 2018). For example, shorebirds are known for their long migrations and during a stopover, individuals can increase their fuel load by as much as 50-100% as subcutaneous fat deposits concentrated in areas in the furcular region or along the sides of the body (Blem 1976, Bairlein 2002). These adipose depots can offer an important tissue sample that provides insight into recent dietary micronutrients (Metzger and Bairlein 2011), fatty acid composition (Araújo et al. 2019), dietary isotopic sources and tracers (Twining et al. 2020), and recent exposure to environmental pollutants (Kenntner et al. 2003, Chu et al. 2015). However, most studies on wild birds have collected adipose samples lethally or relied more on other non-lethal sample types such as blood collections.
Non-lethal tissue sampling is advantageous for animal welfare reasons and allows concurrent measurements of diet, contaminant exposure, physiological responses, and behavior in free-living birds. Although blood or feather sampling methods are the most common tissues to non-lethally measure internal physiology or contaminants, samples of subcutaneous adipose tissue are highly valuable, but less frequently used in studies of living birds likely due to concerns about negative effects of a biopsy that may compromise individual health and survival. Methods to collect adipose biopsies in small passerines, shorebirds, and larger seabirds have been published previously (Owen et al. 2010, Rocha et al. 2016). Data from those experiments suggest that effects of small adipose tissue collections from birds with visible fat stores are negligible and short term. However, importantly, small migratory shorebirds that are actively fueling likely experience greater constraints and it is important to critically evaluate the most efficient and ethical techniques to refine non-lethal tissue collection procedures.
This study was conducted on a captive northern population of migratory Killdeer (Charadrius vociferus) as a model to assess the optimal method for adipose tissue sampling of actively fueling shorebirds in preparation for migration. In particular, our objective was to test refinements in the adipose tissue biopsy method by assessing which analgesia (injectable or topical cream) would be most effective for pain control, reduce stress, and foster healing. We monitored effects of the adipose biopsy on wound healing progression, and mass and fat gain over 21 days to assess any negative effects that may occur in shorebirds during a simulated refueling event.
METHODS
A total of 24 adult Killdeer (genetically sexed as 15 male, 8 female, 1 unknown) that were 2-3 years old and hand reared since hatch at the Facility for Applied Avian Research at the University of Saskatchewan, Saskatoon, Saskatchewan. From early fall, Killdeer were housed in two indoor open aviaries with water sources and natural enrichment under controlled autumn temperature (18 °C) and lighting conditions (10L: 14D). Birds were fed free choice Nutro Natural Choice kitten dry kibble, turkey starter meal (26% protein) and Purina 3 mm fish chow pellets, and supplemented once daily with live mealworms (Malone and Proctor 1966). Birds were uniquely banded, which permitted identification of individuals throughout the study.
In November 2020, Killdeer were captured in groups of six at a time by hand net from their indoor home aviary and placed in a ventilated holding box. Each bird was randomly assigned to one of four treatments as follows: (1) injectable analgesic (e.g., bupivacaine 2 mg/kg) followed by skin incision and fat biopsy (n = 6); (2) topical analgesic EMLA cream (lidocaine/prilocaine 2 mg/kg) followed by skin incision and fat biopsy (n = 6); (3) injectable analgesic only (injectable control, no biopsy) (n = 6); or (4) topical analgesic only (topical control, no biopsy) (n = 6). All control birds experienced the same frequency and duration of capture and handling throughout the study.
Each bird was weighed (± 0.1 g) and scored on the amount of furcular fat from 0 (no visible fat) to 5 (bulging fat over the pectorals) while restrained in the hand by an experienced handler. All birds had at least a fat score of two with visible fat in the interclavical region. The skin and feathers in the furcular (interclavical) region were parted and cleaned with diluted chlorhexidine solution (2% vv). The second experimenter applied either the topical analgesic consisting of 20 mg of 2% EMLA cream applied over an area of 1 cm² or administered the injectable subcutaneous bupivacaine analgesic (0.03-0.04 ml/bird based on 70-100 g range in body mass) into the furcular depression. The time of analgesic application was recorded, and birds were then replaced in the holding box for approximately 8-12 minutes to allow for numbing of the biopsy site. Note that injectable analgesics only require a 1-2-minute wait time, whereas topical applications require longer times of 10 minutes. In the interest of maintaining consistency in holding time across treatments, we kept the wait time the same for all birds. Following the numbing period, a small 5 mm incision was made in the furcular region to collect a subcutaneous adipose tissue sample using a scalpel and forceps following the method described by Rocha et al. (2016). Each fat sample was placed in a microcentrifuge tube and weighed (mean mass 32.5 mg ± 14 (SD); range 15-65 mg). The biopsy site was pinched closed and sealed with Vetbond 3M skin glue and the incision site was photographed. After five minutes of post procedure recovery time in the box, birds were returned to their room and visually observed after one hour.
We assessed the procedure duration (hold time) and pain reaction. Pain, although subjective, was determined by the two investigators as any sign of vocalization, flinching, or struggling during any part of the experimental procedure (observed during skin incision or gluing). We further monitored body mass, fat score, and incision healing on days 0, 1, 3, 7, 10, 14, 17, and 21 post procedure until all birds were fully healed. Scoring of the incision healing was photographed and conducted separately by 2 individuals at the same time of day using a matrix of 5 categories of wound length, wound edges, bleeding or infection, inflammation or redness, and granulation/proliferation (score: 5 to 20; see Appendices 1, 2). Control birds received the same handling as biopsy birds throughout the study. No individuals showed obvious signs of post-procedure pain or lethargy, infection, or poor performance requiring intervention. All procedures were previously approved by the University of Saskatchewan’s University Animal Care Committee under a Certificate of Approval number 20120021.
Data analysis included comparing counts of the pain response during any part of the procedure (response/no response) by analgesic treatment using a chi-square contingency test. Wound healing time was analyzed using a Kaplan-Meier survival curve (time to event analysis) to determine how long it took each treatment group to obtain a wound healing score of 20 (fully healed) as well comparing how long (days) to reach full healing by treatment (t test). We used a repeated measures MANOVA to test for differences in individual wound healing score using fixed factors of treatment (injectable or topical) and time (day post procedure). Responses in body mass and fat scores were similarly assessed over time and treatment using a repeated measures MANOVA. All statistical analyses were conducted in JMP v.16 (SAS Institute).
RESULTS
Pain Response
On average, Killdeer were held in boxes for 11 ± 3.8 mins after application of the analgesic treatment to ensure the area was numb prior to the procedure. During the biopsy, there was a significant difference in pain response between the topical and injectable treatments (χ² = 5.82, p = 0.02, df = 1, n = 12). Differences suggest the injectable analgesic provided greater pain relief because there was a smaller proportion of birds that had a reaction to either the incision or skin glue procedures with the injectable bupivacaine (17%, 1/6 birds) compared to the topical EMLA cream (83%, 5/6 birds; Table 1). Notably, we more commonly observed a reaction to the skin glue (42%, 5/12 birds) than the surgical incision (25%, 3/12 birds) across treatment groups, which suggests the skin glue may be more painful if contacting deeper tissues. The biopsy procedure took on average 4 minutes (range 2-8 mins). The longer time for one individual was caused by multiple gluing attempts needed to adhere the wound edges.
Wound healing
Wound healing times ranged from 14 to 21 days (mean 17 days) with the median time to healing of 15.5 days for birds receiving the injectable analgesic and 17.5 days for topical analgesic (Fig. 1A; Appendix 2). There was no significant difference between the two analgesic treatments on time to reach complete healing (t-ratio = 0.69, df = 10, p = 0.51). Wound healing scores on day 1 after the biopsy procedure ranged from 11 to 16 out of a maximum score of 20 (EMLA mean score = 14, Injectable mean score = 13). Repeated measures MANOVA indicated there was a strong effect of time with progressive healing scores (F6,5 = 143.1, p < 0.0001) but no effect of treatment (F1,10 = 0.004, p = 0.95) or the interaction between time and treatment (F6,5 = 2.4, p = 0.18) such that both analgesic groups followed a similar trajectory of wound healing (Fig. 1B).
Body mass and fat
Killdeer all started the experiment with visible fat and, on average, gained 3.4 g over the 21 days (mean ± SD = 0.16 ± 0.11 g d-1). We observed random differences in the starting body mass between treatment groups (intercept; Fig. 2). Many individuals dropped mass within 24 hours after the experimental day 0 but all birds subsequently gained mass over time (F6,15 = 13.52, p < 0.0001) which peaked at 14 days post biopsy (Fig. 2). Experimental treatment had no effect on body mass over time (time*treatment interaction: F6,15 = 0.34, p = 0.99). Analgesic and biopsy treatments also had no effect on the rate of mass gain (F3,20 = 0.22, p = 0.88). Similar to the mass measurement, fat scoring on days 0, 14, and 21 indicated all birds gained fat at similar rates with no time by treatment interaction (F6,15 = 0.56, p = 0.76).
DISCUSSION
Similar to previous studies on captive bird species, we found the non-lethal adipose biopsy technique was a safe, effective, and minimally invasive procedure that has the potential to greatly add value to the fields of ornithology, ecophysiology, and ecotoxicology (Owen et al. 2010, Rocha et al. 2016). We tested the application of different analgesics to provide more effective pain control and used a more detailed wound healing scoring system to critically evaluate and refine the technique in a fueling shorebird model under controlled laboratory conditions. The Killdeer showed minimal short-term pain response to the procedure, with no lasting effects observed over the period of 21 days. Average time for complete healing in Killdeer was 2-3 weeks with progressive and notable improvements within 7-10 days. Observable gains in body mass and fat were seen after a small drop the first day post procedure. These patterns were comparable across the biopsy treatments and controls suggesting only short-term impacts of handling stress rather than an effect of biopsy or analgesics on individual fueling capability and body mass.
Wound healing time varied by individual and appears to vary across species and study. Rocha et al. (2016) reported wound healing times were more rapid for Dunlins (Calidris alpina; 15 days) and hybrid songbirds (10 days) and reports from Owen et al. (2010) were even shorter for seabirds Black-legged Kittiwakes (Rissa tridactyla), Common Murres (Uria aalge), and Northern Fulmars (Fulmarus glacialis; 6 days). Some of this may be due to differences in wound healing assessment methods or interspecific differences in healing. But more than likely, there is greater variation associated with relative size of the biopsy and efficacy of the wound closure method. For example, we collected a larger fat sample (mean 32.5 ± 3.9 mg; range 15-62 mg) than that reported by Rocha et al. (2016) for Dunlins (9.02 ± 1.18 mg; range = 4-16 mg) and hybrid songbirds (6.31 ± 0.67 mg; range = 2-11 mg). However, we also only tested birds with furcular fat scores of 2 to 4, which means birds had already started fueling. We also found that in at least one individual Killdeer, the tissue adhesive was not effective at closing the wound and this resulted in a slightly longer than average healing time. Therefore, we recommend ensuring there is visible fat in the furcular region, biopsy mass is based on the size of the study bird, minimizing incision size, taking the minimum tissue mass required for the research, and ensuring the incision is fully closed before release.
Adipose tissue is ideal to measure lipid soluble contaminants in migratory birds that are rapidly fueling during stopover in preparation for flight. Because this tissue represents integrated exposure to local contaminants and is a concentrated, lipid-rich material, even a tiny sample collected with a small incision of the furcular (interclavicle) region is anticipated to accurately reflect concentrations accumulated at the stopover. Equally, adipose tissues have successfully been used to assess diet and foraging ecology by measuring natural abundance stable isotopes, polyunsaturated fatty acids, and carotenoid levels. Despite its value, only a few researchers have employed the technique largely for non-lethal sampling in seabirds (e.g., Käkelä et al. 2005, Iverson et al. 2007, Tverin et al. 2021) and in a study with Black-tailed Godwits (Limosa limosa; Viegas et al. 2017, Araújo et al. 2019), suggesting reluctance remains in the general ornithology community for more widespread use in research. To be practical, adipose biopsies should cause minimal pain without lasting effects on health and survival, but these assumptions needed validation for small migratory birds.
The greatest limitation in more widespread adoption of the adipose biopsy technique appears to be a lack of confidence in the method to minimize pain and distress. We can confirm two important refinements in the method that can benefit bird welfare. These are (1) adopting subcutaneous injection of bupivacaine analgesic and (2) refinements to the wound closure technique. There was a marked reduction in pain response when using injectable analgesics compared to the topical cream. Bupivicaine has a longer duration of action, up to eight hours in mammals (Lascelles and Shaw 2016) compared to EMLA cream which may only act for up to three hours in humans (Bjerring and Arendt-Nielsen 1990). In addition, injectable analgesics are faster acting, thus allowing for shorter holding times, which should minimize handling and stress. It appears that topical EMLA cream offers little benefit for pain management, it is challenging to achieve measured doses, and at higher doses can be toxic to birds if ingested; thus, we conclude topical analgesics are not recommended. When closing the wound, we noted that avian skin is very thin and the depression at the biopsy site after removing the fat made it sometimes difficult to close the wound with Vetbond tissue adhesive. Moreover, most of the pain response was during the application of skin glue, particularly if it reached deeper tissues. Vetbond is a non-toxic cyanoacrylate tissue adhesive, which like other similar products (e.g., Dermabond), is approved for veterinary use in closing minor wounds. To improve its application for sealing the biopsy site, we therefore recommend ensuring the incision is as small as possible, collecting adipose tissue from birds with at least a fat score of 2 (visible fat in the furcular region), limiting the fat tissue collection to < 50 mg for a 90 ± 10 g bird (or proportionate to the bird’s size), and gently pinching the wound closed before applying the adhesive to prevent the glue penetrating deeper epidermal layers. With limited training from a licensed veterinarian and the use of injectable analgesics, fat biopsies and wound closure procedures could be safely conducted, even under field conditions.
CONCLUSION
Although we are confident that the adipose biopsy technique had no significant effect on captive individuals, we recommend future work reports any effects on tagged wild-caught birds of different starting fat scores and species before concluding negligible pain or healing responses to the procedure. Wild birds would not likely have the same response to handling and environmental stressors as captive birds and the rate of mass gain for a wild migratory shorebird is much more pronounced (e.g., Red Knot, Calidris canutus, = +1 to 6 gd-1; Atkinson et al. 2007) than reported here for captive Killdeer. Therefore, further testing of the procedure on a small number of free-living shorebirds undergoing rapid refueling would be beneficial to determine if there is no lasting effect on departure timing, survival, and migratory success. Future work is planned to assess the utility of the fat biopsy as a tissue for measuring organic lipophilic contaminants compared to traditional methods using blood samples, which may further provide insight into the most effective sampling method for detecting contaminant exposure in birds. This method holds significant promise as a non-lethal sampling tool for avian wildlife research.
RESPONSES TO THIS ARTICLE
Responses to this article are invited. If accepted for publication, your response will be hyperlinked to the article. To submit a response, follow this link. To read responses already accepted, follow this link.
AUTHOR CONTRIBUTIONS
Christy Morrissey and Kurtis Swekla both conceived the experiment and conducted the animal work. Christy Morrissey analyzed the data and lead the writing of the manuscript with both authors providing editorial input.
ACKNOWLEDGMENTS
We acknowledge the many students and technicians that helped to hatch, rear, and maintain this captive Killdeer population. The work was supported by funding from Natural Sciences and Engineering Research Council of Canada (NSERC) to CAM and Canada Foundation for Innovation (CFI) Infrastructure Operating grant in support of the operations of the Facility for Applied Avian Research.
DATA AVAILABILITY
The raw data files used in the study are available through an open access FRDR data repository. Morrissey, C. , Swekla, K. (2023) Data file for "Refinements in adipose tissue biopsy collection in shorebirds: effect on pain, wound healing time and mass gain". Federated Research Data Repository. https://doi.org/10.20383/102.0697
LITERATURE CITED
Araújo, P. M., I. Viegas, A. D. Rocha, A. Villegas, J. G. Jones, L. Mendonça, J. A. Ramos, J. A. Masero, and J. A. Alves. 2019. Understanding how birds rebuild fat stores during migration: insights from an experimental study. Scientific Reports 9:10065. https://doi.org/10.1038/s41598-019-46487-z
Atkinson, P. W., A. J. Baker, K. A. Bennett, N. A. Clark, J. A. Clark, K. B. Cole, A. Dekinga, A. Dey, S. Gillings, P. M. González, K. Kalasz, C. D. T. Minton, J. Newton, L. J. Niles, T. Piersma, R. A. Robinson, and H. P. Sitters. 2007. Rates of mass gain and energy deposition in Red Knot on their final spring staging site is both time- and condition-dependent. Journal of Applied Ecology 44:885-895. https://doi.org/10.1111/j.1365-2664.2007.01308.x
Bairlein, F. 2002. How to get fat: nutritional mechanisms of seasonal fat accumulation in migratory songbirds. Naturwissenschaften 89:1-10. https://doi.org/10.1007/s00114-001-0279-6
Bjerring, P., and L. Arendt-Nielsen. 1990. Depth and duration of skin analgesia to needle insertion after topical application of EMLA cream. British Journal of Anaesthesia 64(2):173-177. https://doi.org/10.1093/bja/64.2.173
Blem, C. R. 1976. Patterns of lipid storage and utilization in birds. American Zoologist 16:671-684. https://doi.org/10.1093/icb/16.4.671
Chu, S., J. Wang, G. Leong, L. A. Woodward, R. J. Letcher, and Q. X. Li. 2015. Perfluoroalkyl sulfonates and carboxylic acids in liver, muscle and adipose tissues of Black-footed Albatross (Phoebastria nigripes) from Midway Island, North Pacific Ocean. Chemosphere 138:60-66. https://doi.org/10.1016/j.chemosphere.2015.05.043
Guglielmo, C. G. 2018. Obese super athletes: fat-fueled migration in birds and bats. Journal of Experimental Biology 221:jeb165753. https://doi.org/10.1242/jeb.165753
Iverson, S. J., A. M. Springer, and A. S. Kitaysky. 2007. Seabirds as indicators of food web structure and ecosystem variability: qualitative and quantitative diet analyses using fatty acids. Marine Ecology Progress Series 352:235-244. https://doi.org/10.3354/meps07073
Jenni, L., and S. Jenni-Eiermann. 1998. Fuel supply and metabolic constraints in migrating birds. Journal of Avian Biology 29:521-528. https://doi.org/10.2307/3677171
Käkelä, R., A. Käkelä, S. Kahle, P. H. Becker, A. Kelly, and R. W. Furness. 2005. Fatty acid signatures in plasma of captive Herring Gulls as indicators of demersal or pelagic fish diet. Marine Ecology Progress Series 293:191-200. https://doi.org/10.3354/meps293191
Kenntner, N., O. Krone, G. Oehme, D. Heidecke, and F. Tataruch. 2003. Organochlorine contaminants in body tissue of free-ranging White-tailed Eagles from northern regions of Germany. Environmental Toxicology and Chemistry 22:1457-1464. https://doi.org/10.1002/etc.5620220706
Lascelles, B. D. X., and K. K. Shaw. 2016. An extended release local anesthetic: potential for future use in veterinary surgical patients? Veterinary Medical Science 2(4):229-238. https://doi.org/10.1002/vms3.43
Malone, C. R., and V. W. Proctor. 1966. Rearing Killdeers for experimental purposes. Journal of Wildlife Management 30:589-594. https://doi.org/10.2307/3798752
Metzger, B. J., and F. Bairlein. 2011. Fat stores in a migratory bird: a reservoir of carotenoid pigments for times of need? Journal of Comparative Physiology B 181:269-275. https://doi.org/10.1007/s00360-010-0511-9
Owen, E., F. Daunt, and S. Wanless. 2010. Sampling avian adipose tissue: assessing a nondestructive biopsy technique. Journal of Field Ornithology 81:92-98. https://doi.org/10.1111/j.1557-9263.2009.00265.x
Rocha, A. D., P. M. Araújo, F. R. Martinho, J. A. Ramos, and J. A. Masero. 2016. A non-lethal biopsy technique for sampling subcutaneous adipose tissue of small and medium-sized birds. Journal of Field Ornithology 87:213-221. https://doi.org/10.1111/jofo.12145
Tverin, M., J. Granroth, A. Abrahamsson, P. Tang, H. Pihlström, K. Lundström, and R. Käkelä. 2021. Adipose tissue fatty acids suggest spatial and temporal dietary differences in Great Cormorants of the Baltic Sea area. Journal of the Marine Biological Association of the United Kingdom 101:199-213. https://doi.org/10.1017/S0025315420001320
Twining, C. W., S. J. Taipale, L. Ruess, A. Bec, D. Martin-Creuzburg, and M. J. Kainz. 2020. Stable isotopes of fatty acids: current and future perspectives for advancing trophic ecology. Philosophical Transactions of the Royal Society B: Biological Sciences 375:20190641. https://doi.org/10.1098/rstb.2019.0641
Viegas, I., P. M. Araújo, A. D. Rocha, A. Villegas, J. G. Jones, J. A. Ramos, J. A. Masero, and J. A. Alves. 2017. Metabolic plasticity for subcutaneous fat accumulation in a long-distance migratory bird traced by 2H2O. Journal of Experimental Biology 220:1072-1078. https://doi.org/10.1242/jeb.150490
Fig. 1
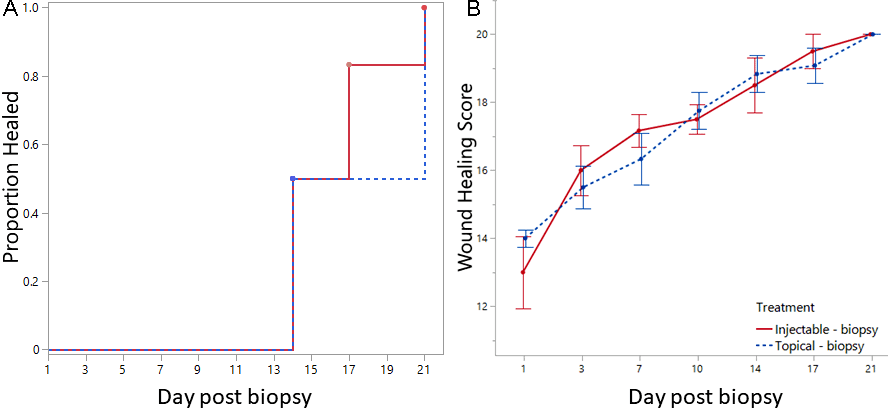
Fig. 1. (A) Time to event analysis showing the timing of when captive Killdeer (Charadrius vociferus) from each treatment (Injectable n = 6 or Topical n = 6) were fully healed after adipose biopsy sampling. All birds were fully healed by Day 21. (B) Progression of wound healing (means ± SE) over time by analgesic treatment (injectable and topical). Wound healing was scored by 2 observers using 5 categories (score = 5 to 20) where a score of 5 is an open wound and a score of 20 is fully healed (see Appendix 1 for details of wound scoring).

Fig. 2
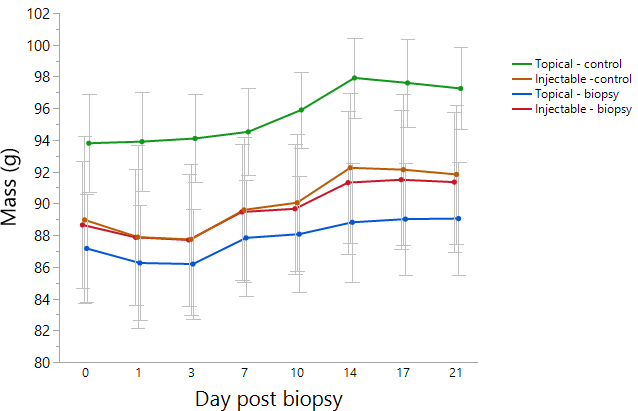
Fig. 2. Mean Killdeer (Charadrius vociferus) body mass (g) over time from 0 to 21 days post procedure. On day 0, birds were randomly assigned to 4 treatment groups receiving injectable analgesic with biopsy (n = 6), topical analgesic with biopsy (n = 6), injectable analgesic without biopsy (n = 6), or topical analgesic without biopsy (n = 6). All birds were handled identically throughout the study. Note error bars were faded for visual clarity.

Table 1
Table 1. Observable pain responses (n count, % of birds) after receiving topical lidocaine/prilocaine analgesic or injectable bupivacaine followed by biopsy treatment. Pain was observed only in response to the incision or application of the skin glue or both.
| No pain response | Response to glue | Response to incision | Response to incision and glue | |
| Topical + biopsy (n = 6) |
1 (17%) | 2 (33%) | 2 (33%) | 1 (17%) |
| Injectable + biopsy (n = 6) |
4 (67%) | 2 (33%) | 0 (0%) | 0 (0%) |
| Total (n = 12) | 5 | 4 | 2 | 1 |








