The following is the established format for referencing this article:
Vanausdall, R. A., D. P. Collins, W. C. Conway, K. Kraai, C. D. Mason, and J. W. Gunnels. 2022. Body condition and molt chronology of waterfowl in east central Texas. Journal of Field Ornithology 93(4):5.ABSTRACT
Waterfowl undergo changes in body condition during migratory and wintering periods that can affect survival and reproduction. Understanding how condition varies among age and sex cohorts and temporally can inform habitat management during the post- and pre-breeding period. We examined changes in body composition and molt of three waterfowl species migrating and wintering in managed moist-soil units in east central Texas and determined the influence of season, age, and sex using Analysis of Variance. We examined age, sex, and season effects on body condition indices (body mass divided by wing chord) using mixed-effects models and determined the influence of molt score on body condition using linear regression. For specimens collected from 2004–2007, we found differences in body composition among age and sex groups for Blue-winged Teal (Spatula discor), a migrant through this study area, and for Green-winged Teal (Anas crecca), and Northern Shoveler (Spatula clypeata), which are wintering species. We found a mid-winter decline in body condition for Green-winged Teal. Blue-winged Teal were in worse condition in spring than in fall, which could be due to differences in energy accumulation prior to departure to wintering grounds in fall and post-arrival in spring. We did not find an effect of season for Northern Shoveler. For all species, adults were in better condition than juveniles. Molt score was greatest in late-winter for all species combined (x̄ = 11.60, SE = 2.45). We did not find an effect of molt score on body condition for any species. Wintering waterfowl in east central Texas appear to be in good condition and did not show molt-induced stress during the migratory and wintering periods. Changes in condition among specimens and across seasons indicate that there should be consideration in the timing and quality of management in regional approaches to moist-soil management for migrating and wintering waterfowl.RESUMEN
INTRODUCTION
During winter and migration, waterfowl body mass and carcass composition vary due to a variety of internal and external variables influencing body condition, defined as an individual’s ability to meet present and future energetic demands (Labocha and Hayes 2012). Accumulation and maintenance of nutrient reserves are important to waterfowl during nonbreeding periods, as changes can directly affect survival and have cross-seasonal effects on survival and reproduction (Blem 1976, Baldassarre et al. 1986, Schmutz and Ely 1999, Moon et al. 2007, Devries et al. 2008a). Morphological changes and variation in organs may be indicative of exogenous factors influencing condition, but they can also be adaptive and allow birds to balance the energetic demands of the migratory and wintering seasons with a molting period that exposes them to predation (Hohman 1993, King and Fox 2012). Condition indices have been recognized as valuable tools for managing waterfowl and other species for which condition has been linked to various fitness components (Hepp et al. 1986, Moser and Rusch 1988, DeVault et al. 2003, Devries et al. 2008b, Labocha and Hayes 2012). Estimating body condition from easily obtained measurements in the field can allow managers to monitor changes in condition throughout the season and determine the overall health or adaptive responses of a population (Johnson et al. 1985, Moser and Rusch 1988, Labocha and Hayes 2012).
Several methods exist to estimate the condition indices of birds, particularly in waterfowl. Lipids are the main source of energy for migrating birds (Odum and Connell 1956, Jenni and Jenni-Eiermann 1998), but direct measurements of percent body fat can be time-consuming and expensive to obtain. Body mass alone is often correlated with lipid reserves (Miller 1989) and is often used as an index for body condition (Labocha and Hayes 2012, Fleskes et al. 2016). Historically, other waterfowl body condition indices have been estimated using total body mass standardized by external and/or internal morphological measurements (i.e., total body length, wing cord, flight muscle mass, etc.) to create indices that are presumably correlated with lipid or protein levels (Wishart 1979, Johnson et al. 1985, Moser and Rusch 1988). The aim of creating body condition indices is to separate the influence of body mass on condition due to structural size from aspects that reflect fat and other energy reserves (Wishart 1979, Johnson et al. 1985, Green 2001). Indices thus allow researchers and managers to evaluate variability in body condition over time that is not due to structural growth. Accounting for temporal variation is particularly important when considering that condition at certain times of the year may have a more profound impact on survival or reproduction than others (Schmutz and Ely 1999, Devries et al. 2008b).
Although estimates of condition alone are key elements in understanding the winter and migration ecology of waterfowl, singly they may provide limited insight into factors driving variation in condition besides obvious temporal variation due to different age–sex cohorts. Examination of both body composition and molt can reveal possible energetic requirements and changes in dietary type and quality during the nonbreeding period and inform management needs for waterfowl (Ankney 1979, DuBowy 1985, Moorman et al. 1992). Molt, for instance, is a costly process for birds (Payne 1972), requiring physiological demands through feather formation and growth, affecting thermoregulatory capabilities, and potentially resulting in a change in behavior (Murphy 1996). Regardless of the direct role of molt in body condition, both juveniles and adult waterfowl experience physiological demands to recover after fall migration, complete body molt, and successfully acquire a mate, all while avoiding predation, harvest, and maintaining body condition to improve over-winter survival (Heitmeyer 1988, Miller 1986, Smith and Sheeley 1993, Hohman and Crawford 1995).
Simultaneous estimates of body condition and molt chronology are limited for some species, particularly for migrating and overwintering waterfowl using moist-soil managed wetlands (hereafter, managed wetlands). Management of such wetlands is focused specifically on food production during migration and winter, where waterfowl using managed wetlands should avoid food shortages and consequent delays in molt progression while simultaneously maintaining body condition (Collins 2012). Moreover, waterfowl migrating or wintering in more southerly latitudes should avoid extended periods of severe weather which may alleviate (1) commonly observed mid-winter declines in body condition (Fredrickson and Taylor 1982), (2) pressures to extend or delay molt, and (3) potential food shortages. The objectives of this study were to quantify body condition, body composition, and feather molt progression and intensity of Blue-winged Teal (Spatula discor), Green-winged Teal (Anas crecca), and Northern Shoveler (Spatula clypeata) during winter using managed wetlands on the Richland Creek Wildlife Management Area (RCWMA) in east-central Texas.
METHODS
Study area
Our research was conducted on the RCWMA’s North Unit managed wetlands 1–4. Richland Creek Wildlife Management Area (31º13'N, 96º11'W) is located 40 km southeast of Corsicana, Texas, along U.S. Highway 287 and FM 488 between Richland-Chambers Reservoir and the Trinity River in Freestone and Navarro counties, Texas (Collins 2012). The RCWMA contains two units (North and South) encompassing 6271 ha located in the ecotone separating the Post Oak Savannah and Blackland Prairie ecological regions (TPWD 2005) and lies almost entirely within the Trinity River floodplain. The local climate is considered subtropical with mild winters and warm humid summers, with an average winter temperature of 5°C and average rainfall of 101.6 cm per year (NRCS 2002). Rainfall is typically distributed evenly throughout the year. Soils in the area are predominately of the Trinity series, which are fine, montmorillonitic, thermic, very haplaquolls, and mollisol soils (NRCS 2002).
Vegetation within the South Unit is characterized by vast bottomland hardwood forest communities dominated by eastern red cedar (Juniperus virginiana), sugarberry (Celtis laevigata), and green ash (Fraxinus pennsylvanica). Other species include honey locust (Gleditisia triacanthos), box elder (Acer negundo), black willow (Salix nigra), bur oak (Quercus macrocarpa), water oak (Q. nigra), overcup oak (Q. lyrata), willow oak (Q. phellos), and pecan (Carya illinoensis). The North Unit contains the managed wetlands, which are large non-forested areas characterized by a diverse herbaceous community. The typical water management strategy consists of slow drawdown (i.e., removal of water) starting late March – early April and lasting until mid-August. Inundation (i.e., flooding) begins in late August and lasts throughout fall and winter, until drawdown the following spring. These management actions produced common species such as barnyard grass, erect burhead (Echinodorus spp.), delta duck potato (Sagittaria spp.), square-stem spike rush (Eleocharis quadrangulata), wild millet, and water primrose (Ludwigia peploides).
Scientifically collected and hunter-harvested specimens
Specimens of Blue-winged Teal, Green-winged Teal, and Northern Shoveler were collected on managed wetlands using a 12-gauge shotgun and steel shot from 1 September – 30 March 2004 – 2005, and 2005 – 2006. Specimens were collected using decoys during days RCWMA was closed to hunting by the general public and are hereafter referred to as scientifically collected ducks. All scientifically collected focal species were collected under the appropriate permits (U.S. Fish and Wildlife Scientific Collection Permit MB093036-0 and Texas Parks and Wildlife Department Scientific Collecting Permit SPR-0704-399, both issued to D. Collins). For all birds, age and sex were recorded following Carney (1992) and the following morphological features were measured: body mass (g), bill length (cm), culmen length (cm), maximum bill width (cm), keel length (cm), tarsus length (cm), and total body length (cm). Specimens were put on ice, transported back to the lab, and frozen for future dissection. In addition to selectively collecting (i.e., scientifically collected) the focal species, we also obtained hunter-harvested specimens from September to January for the same time-period, along with an additional hunting season in 2007, through check stations on days RCWMA was open to hunting. Both scientifically collected and hunter-harvested ducks were shot over decoys.
Body composition
Prior to dissection, scientifically collected birds were thawed and weighed (±0.01 g) to obtain total body mass. Feathers were removed, and the bill, tarsi, skin, and all internal organs (i.e., heart, gastrointestinal tract, liver, lungs, and gizzard) were removed and weighed (±0.01 g). Mesentery fat was removed from the viscera and returned to the carcass (Morton et al. 1990), while all digestive contents were removed from the esophagus, proventriculus, gizzard, intestine, and caeca (Hohman et al. 1992). If material was present in the digestive tract it was washed into a container and stored (Morton et al. 1990). Digestive contents were weighed (±0.01 g) and subtracted, with feather mass, from total body mass to obtain feather-free and ingesta-free carcass mass (i.e., corrected body mass). Omental fat and visceral fat were removed and weighed (±0.01 g; Woodall 1978), and the entire length of the gastrointestinal tract was measured (±5 mm) and weighed (±0.01 g; Austin and Fredrickson 1987). Flight muscles (i.e., pectoralis, supracoracoideus, and coracobrachialis) on the left side were removed from the sternum (Owen and Cook 1977, Morton et al. 1990). Leg muscles were also removed from the left side. External fat was removed and returned to the carcass from the gizzard and flight muscles. Wet mass of the gizzard, heart, liver, and flight and leg muscles were weighed (±0.01 g; Austin and Fredrickson 1987). We obtained a subset of morphological measurements on hunter-harvested specimens, including fresh body mass, wing chord, tarsal length, and total body length. Measurements on hunter-harvested specimens were collected within two hours after harvest.
Body condition
For body condition, we used fresh body mass for the whole carcass, as this value was available from both scientifically collected and hunter-harvested ducks. We calculated a commonly used body condition index (BCI) by dividing body mass by wing chord for our index of body condition (Owen and Cook 1977, Haukos et al. 2001). The mass-wing index has been shown to be highly correlated with lipid content for several waterfowl species (Woodall 1978, Whyte and Bolen 1984, Gauthier and Bédard 1985) and is often used by waterfowl managers to examine changes in body condition over time.
Feather molt chronology and intensity
As feather removal occurred, we inspected 17 feather tracts (i.e., crown, face, rump, tail, belly, etc.) to score feather molt intensity following Heitmeyer (1988). For each tract, the number of sheathed feathers was counted and used to calculate total molt score for each specimen. Molt score was calculated by summing the total number of feathers found erupting (i.e., containing a sheath) on all tracts and then dividing by the total number of feather tracts examined, which in this case was 17 (e.g., 56 sheathed feathers found across all 17 tracts would result in a molt score of 56/17=3.29 for that individual bird). We summarized molt score by species and season.
Statistical analyses
For scientifically collected specimens, we used analysis of variance (ANOVA) with Type III sums of squares to examine differences among external and internal morphological features (i.e., body structure and organ measurements) among different age and sex classes and season. We did not include hunter-harvested specimens because not all morphological features were recorded for this group. We defined 4 seasons: fall (1 September – 15 November), early winter (16 November – 31 December), late winter (1 January – 28 February), and spring (1 March – 30 March). Seasons align with definitions in previous research in the study area (Collins et al. 2017) and with other waterfowl studies (e.g., Heitmeyer and Fredrickson 1981). Due to a limited sample size for some species and body parts during all four seasons, we examined only main effects models with age, sex, and season for all species. We included ducks collected in all seasons for Green-winged Teal and Northern Shoveler, which migrate through and winter in the study area. We only included Blue-winged Teal collected in fall and spring, as this species mostly migrates through the study area. We used the Tukey-Kramer post hoc test to determine significant (P < 0.05) differences among seasons.
To examine differences in the body condition indices, we included both scientifically collected and hunter-harvested specimens in analyses. We used mixed-effects ANOVA to determine differences in BCI for each species. We used model selection methods modified from Fleskes et al. (2016). Initially, we used a model with the fixed effects of collection type and an interaction of age and sex, a random effect of year, and a variance structure that allowed for different variances in BCI for each season (Zuur et al. 2009). We used maximum likelihood estimation and Akaike’s Information Criterion (AICc) to compare this full model with nested models (Burnham and Anderson 2002, Zuur et al. 2009). If a model without collection type did not improve model fit (i.e., ΔAICc > 2), we discarded this variable for subsequent analyses (Burnham and Anderson 2002). We then examined different combinations of sex and age. We considered models with main effects of both sex and age and an interaction of sex and age and separate models with either sex or age. We included season with the top sex–age model to determine if there was an overall season effect on BCI (i.e., ΔAICc > 2 for the season model). We examined interactions of season with the sex–age effects. We did not examine an interaction of collection type with season because hunter-harvested specimens were exclusively collected during hunting seasons occurring in fall and early winter. We included a null model to compare to all models with covariates. We reported all models and identified competitive models to be those with a ΔAICc < 2 and that were not more complex versions of the top model (Burnham and Anderson 2002, Devries et al. 2008a, Arnold 2010). We used the Tukey-Kramer post hoc test to identify significant (P < 0.05) differences in condition indices among seasons or interactions with more than two groups. We used the “nlme” package in Program R (R Core Team 2021) to build and compare models (Pinheiro et al. 2021) and the “multcomp” package to examine seasonal differences (Hothorn et al. 2008).
We used ANOVA with Type III sums of squares to examine molt scores for a subset of scientifically collected specimens across species, seasons, and year. Due to a limited sample size, we did not include effects of age or sex. Additionally, we used a linear regression to determine the effect of molt intensity on BCI for each species. We considered variables to influence the response if P < 0.05 in all cases.
RESULTS
Body composition
We collected a total of 63 and 77 Blue-winged Teal during the 2005–2006 and 2006–2007 periods, respectively. We found several differences in morphological and structural features of Blue-winged Teal, particularly between the sexes and across seasons (Table A1.1). We found a sex effect for corrected body mass (F1,136 = 18.008, P < 0.001) and mass of the esophagus-proventriculus (F1,99 = 4.538, P = 0.036) and leg muscle (F1,100 = 4.244, P = 0.036). Males weighed more than females (P = 0.009) and had heavier esophagus-proventriculus (P = 0.016), flight muscle, (P < 0.001) and leg muscle (P < 0.001). We found an age effect for corrected body mass (F1,136 = 8.820, P = 0.004), where adults weighed more than juveniles (P = 0.010). We also found a slight age effect for omental fat mass (F1,136 = 4.153, P = 0.044), with adults having more omental fat than juveniles, but the pairwise comparison was not significant (P = 0.337). Additionally, corrected body mass (F1,136 = 13.492, P < 0.001) and mass of the liver (F1,99 = 6.255, P = 0.014), intestines (F1,99 = 5.388, P = 0.023), flight muscle, (F1,100 = 5.242, P = 0.024), leg muscle, (F1,100 = 4.339, P = 0.040), omental fat (F1,99 = 34.998, P < 0.001), mesenteric fat (F1,99 = 30.769, P < 0.001), and skin (F1,100 = 29.046, P < 0.001) varied by season. Blue-winged Teal weighed more in fall than in spring (P = 0.003) and had greater mass values for liver (P = 0.039), omental fat (P < 0.001), mesenteric fat (P < 0.001), and skin (P < 0.001) in fall than in spring. Mass of intestines, flight muscle, and leg muscle were greater in fall than in spring, but these results were not significant.
We collected 93 and 23 Green-winged Teal in the 2004–2005 and 2005–2006 periods, respectively. Green-winged Teal mostly differed in internal and external features by sex, but there were some differences in age and season (Table A1.2). Corrected body mass varied by sex (F1,110 = 10.304, P = 0.002), where males were heavier than females (P = 0.003). Esophagus-proventriculus length (F1,90 = 8.300, P = 0.005) and mass (F1,90 = 16.704, P < 0.001), along with mass of the gizzard (F1,93 = 10.368, P = 0.002), heart (F1,93 = 4.226, P = 0.043), and flight muscle (F1,93 = 6.427, P = 0.013), varied by sex. Males had longer (P = 0.005) and heavier (P < 0.001) esophagus-proventriculus values compared to females. Mass of the gizzard (P = 0.001) and flight muscle (P = 0.002) were also greater for males than females. Pairwise comparisons of heart mass did not show a significant difference between males and females (P = 0.072). We found a season effect for corrected body mass (F1,110 = 4.886, P = 0.003) and mass of the intestine (F1,92 = 5.202, P = 0.002) and leg muscle (F1,93 = 8.984, P < 0.001). Corrected body mass was greater in spring than in late winter (P = 0.002). Intestine mass was greater in fall than in late winter (P = 0.002), while leg muscle mass was greater in late winter than in fall (P = 0.001) and greater in spring than in fall (P < 0.001).
We collected 67 and 54 Northern Shoveler during the 2004–2005 and 2005–2006 periods, respectively (Appendix A1.3). We found a sex effect for corrected body mass (F1,115 = 5.428, P = 0.022) and mass of the heart (F1,77 = 9.526, P = 0.003), flight muscle (F1,77 = 9.101, P = 0.003), and leg muscle (F1,77 = 5.130, P = 0.026). Pairwise comparisons did not show a difference between males and females for corrected body mass (P = 0.238), but mass of the heart (P = 0.016), flight muscle (P = 0.003), and leg muscle (P = 0.015) were greater in males than in females. Corrected body mass (F1,115 = 5.074, P = 0.026) and mass of the gizzard (F1,77 = 4.492, P = 0.037) and intestine (F1,76 = 5.642, P = 0.020) varied by age. Adults weighed more than juveniles (P = 0.012) and had greater values for intestine mass (P = 0.027). Pairwise comparisons did not show a difference between adults and juveniles for gizzard mass (P = 0.137). Finally, we found a season effect for mass of the gizzard (F1,77 = 7.727, P < 0.001), intestines (F1,76 = 9.123, P < 0.001), omental fat (F1,76 = 3.054, P = 0.033), and mesenteric fat (F1,76 = 7.686, P < 0.001). Gizzard mass appeared to decline throughout the winter, with a greater value in fall than in late winter (P = 0.004), in early winter than in late winter (P = 0.015), and in fall than in spring (P = 0.037). Similarly, intestine mass was greater in late winter than in fall (P < 0.001), early winter than in fall (P = 0.014), and spring than in late winter (P = 0.041). However, intestine mass in spring was less than in fall (P = 0.013). We found greater values of mesentery fat in fall than in early winter (P < 0.001), late winter (P < 0.001), and spring (P = 0.002). Pairwise comparisons were not significant for omental fat across seasons.
Body condition
In addition to scientifically collected specimens, we received 231, 436, and 120 hunter-harvested Blue-winged Teal, Green-winged Teal, and Northern Shoveler, respectively, over the study period. Adult males tended to weigh more than juveniles and females, and scientifically collected specimens tended to weigh more than hunter-harvested specimens on average (Table A1.4). Adults also showed a greater BCI than juveniles on average, while BCI among males and females varied for two species (Fig. 1).
For Blue-winged Teal, the top model included main effects of collection type and season and an interaction of sex and age (Table 1). Collection type and season showed significant effects on BCI. We found that scientifically collected Blue-winged Teal were in better condition than hunter-harvest specimens (P < 0.001) and that fall specimens were in better condition than spring specimens (P < 0.001; Fig. 2). Adult males were in slightly better condition than adult females (P = 0.048) and were in better condition than juvenile females (P < 0.001), and juvenile males were in better condition than juvenile females (P = 0.001). However, the interaction of sex and age in the top model was not significant (P = 0.499).
For Green-winged Teal, the top model included main effects of collection type, season, sex, and age, while the second-best model included main effects of collection type, sex, and an interaction of season and age (Table 1). Pairwise comparisons indicated that Green-winged Teal were in better condition in early winter than in late winter (P = 0.023; Fig. 2), while specimens were in slightly better condition in spring than in late winter (P = 0.043). Males were in better condition than females (P < 0.001), while adults were in better condition than juveniles (P < 0.001). Although not significant (P = 0.071), hunter-harvested specimens were in slightly better condition than scientifically collected specimens. Under the second-best model, only early-winter adults were in better condition than early-winter juveniles (P < 0.001).
For Northern Shoveler, the top model included main effects for collection type and age (Table 1). Scientifically collected Northern Shoveler were in slightly worse condition than hunter-harvested specimens (P = 0.009). Adult Northern Shoveler were in better condition than juveniles (P = 0.003). We did not find an effect of season for this species (Fig. 2).
Feather molt chronology and intensity
We found an effect of species on molt score (F2,199 = 7.422, P < 0.001). Northern Shoveler had a greater molt score than Blue-winged Teal (P < 0.001) and Green-winged Teal (P = 0.004; Fig. 3). Overall, molt score for all species combined was greatest in late winter (xˉ = 11.60, SE = 2.45), followed by spring ( = 9.06, SE = 1.34), early winter (xˉ = 4.45, SE = 0.89), and fall (xˉ = 4.14, SE = 1.00). Within species, general trends showed that molt intensity was greatest in spring for all species, but there was considerable variability (Table A1.5). We did not find significant effects of BCI on molt score for any species.
DISCUSSION
Blue-winged Teal, Green-winged Teal, and Northern Shoveler tended to show predictable patterns in morphological features based on age and sex and varied by body mass, body condition, and molt intensity across seasons during winter in the RCWMA. Overall, both wintering species appeared to be in better or similar condition prior to spring departure and estimates of overall body mass corroborate this finding. Blue-winged Teal were the only species that showed a significant decline between spring and fall in BCI, and molt intensity did not appear to influence body condition of migrating and wintering waterfowl. Variations in body condition over time emphasize the importance of considering a BCI that best represents the species and population and have management implications (DeVault et al. 2003, Labocha and Hayes 2012). Management programs (e.g., Wildlife Management Areas) that focus on providing habitat for migrating and wintering waterfowl may use such observed apparent changes in body condition to determine if current management strategies are adequate in supplying energy requirements throughout the year (e.g., Fleskes et al. 2016).
Some internal features differed by sex, age, and season for each species, which can often be attributed to variations in behavior and activity, diet preference and quality, endogenous processes, and nutritional demands (Austin and Fredrickson 1987, Ankney and Afton 1988, Heitmeyer 1988). For example, some digestive organs varied by sex and season for waterfowl at RCWMA. While organs and structures of the body may scale with body size, leading to inherent differences in some features among males and females, organs and muscles may also vary due to environmental conditions and diet (Kehoe and Ankney 1985, Piersma et al. 1993). Differences in the digestive organs may be due to differences in the amount or quality of foods ingested (Drobney 1984, Kehoe et al. 1988). Increased food consumption may result in an increase in intestine mass or length (Drobney 1984, DuBowy 1985, Ankney and Afton 1988), while an increased intake of high-fiber foods may result in a decrease in gizzard mass (Kehoe et al. 1988). Collins et al. (2017) did not find a significant effect of sex on the percent dry mass of plant or animal material consumed by ducks in this region in fall and winter, but differences in the amount of protein and fiber within plant and animal matter may have influenced differences in digestive organs (Anderson et al. 2000). While we did not examine a season–sex or season–age effect on internal features for waterfowl, it is possible differences in digestive organs between males and females were due to shifts in dietary preferences.
Sex and age also influenced body condition and features of the body that can indicate overall condition, such as body mass and fat mass. Structurally, males tend to be larger than females and adults tend to be larger than juveniles (Owen and Cook 1977, Euliss and Gilmer 1997, DeVault et al. 2003, Ballard et al. 2006), which we found to be mostly true for all species. Additionally, social interference can influence habitat use among individuals in a population, with more dominant individuals acquiring more suitable habitat than subordinates. Adult waterfowl tend to dominate hatch-year individuals (Morton et al. 1990, Reinecke et al. 1982), which can result in young individuals being displaced from habitat with better foraging opportunities or shelter.
We expected season to explain some of the variation in body condition for all three species but particularly for the wintering species: Green-winged Teal and Northern Shoveler. Typically, waterfowl increase in body mass and lipid reserves upon arrival to wintering areas and to early winter, show a decline in mid-winter, and increase to spring (Owen and Cook 1977, Whyte and Bolen 1984, Thompson and Baldassarre 1990, Rhodes et al. 2006, Fleskes et al. 2016). The pattern of rapid increase upon arrival to wintering grounds is likely a result of replenishing energy reserves post-breeding and preparing for the energetic demands of winter (Reinecke et al. 1982, Baldassarre et al. 1986). A lower body mass in winter may also be an adaptive strategy, as the use of energy reserves reduces (1) the need to forage, (2) exposure to potentially inclement weather, and (3) the metabolic costs of maintaining a higher weight (Reinecke et al. 1982, Baldassarre et al. 1986, Fondell et al. 2013). Indeed, east-central Texas does not typically have harsh winter weather conditions, so mid-winter declines may be more endogenous than exogenously related for Green-winged Teal. Baldassarre et al. (1986) also found a mid-winter decline for Green-winged Teal in the Southern High Plains of Texas. Although average values showed a slight decline in BCI from fall to spring, our analysis did not find a season effect for Northern Shoveler. This contrasts with findings from other studies which found a mid-winter decline in this species (Tietje and Teer 1988, Roberts and Conover 2014) or a decrease from fall to winter for body mass (Fleskes et al. 2016), but still others have found no effect of season on body condition (Euliss and Gilmer 1997). Ultimately, body condition in spring was better or similar to condition in fall for both species, indicating that Green-winged Teal and Northern Shoveler are likely acquiring adequate energy reserves during the winter in the study area.
In contrast to the other two species, there may be evidence that Blue-winged Teal in spring are in slightly worse condition than fall specimens. The BCI was predicted to be greater in fall than in spring, and both mesenteric and omental fat masses were significantly greater in fall than in spring. Blue-winged Teal winter along the coasts of Central and South America after departing stopover areas in Texas and other coastal states (Rohwer et al. 2020). It is likely that by the time they get to the RCWMA in fall, individuals may still be acquiring energy reserves in preparation for potentially long flights to the wintering grounds (Owen 1970). In contrast, those arriving in spring may have reduced body mass or condition due to longer flights from those wintering areas and may have not fully replenished their energy reserves, which may explain the difference in BCI between fall and spring in our specimens.
All waterfowl species showed typical molting patterns during winter. In general, Northern Shoveler had greater molt scores and molt intensity, primarily due to differences in overall size and greater potential feather growth. In contrast, both Teal species typically have a slower rate of feather replacement due to their small size, which makes them more susceptible to molt-induced stress than larger bodied ducks such as Northern Shoveler (Hohman 1993, Anderson et al. 2000). However, we did not find an effect of molt on body condition for any of the species examined, and all three focal species appeared to be in comparatively good condition throughout the wintering period. Several other studies have found that waterfowl likely meet the energetic demands of molt through dietary intake (Ankney 1979) or protein stores and reduced foraging activity (Portugal et al. 2007, Fondell et al. 2013). We conclude that molt-induced stress may not have been occurring for any focal species.
Our study was the first documentation of changes in body condition, molt intensity, and internal features of three species of waterfowl that heavily use east-central Texas during migration and overwinter periods. It also highlights the importance of managing habitats in the area to provide resources needed throughout the annual cycle of migrant waterfowl. The mid-winter declines of condition indices in Green-winged Teal adds to a growing body of literature documenting the potential adaptive responses of waterfowl to winter conditions. This, along with the consistent temporal patterns of body condition for Northern Shoveler, is evidence that management on the RCWMA is likely providing adequate resources for wintering waterfowl in the region. At the same time, Blue-winged Teal showed a decline in body condition between fall to spring. A reduced BCI is likely due to a difference in pre-fattening in fall prior to departure and lower energy reserves during northbound migration in spring from the wintering grounds and emphasizes the importance of the RCWMA as a potentially important refueling site for migrant waterfowl. Ensuring foraging resources and habitat are available in the spring and fall will benefit migrating waterfowl and better allow individuals to reach their breeding or wintering areas. Based on conclusions from our study and those from Collins et al. (2017), continuing to provide quality habitats during the wintering period at RCWMA and the surrounding area should allow our focal species to maintain and improve body composition and condition.
RESPONSES TO THIS ARTICLE
Responses to this article are invited. If accepted for publication, your response will be hyperlinked to the article. To submit a response, follow this link. To read responses already accepted, follow this link.ACKNOWLEDGMENTS
Financial and logistical support for this research was provided by the Texas Parks and Wildlife Department Statewide Grant T-8-1, and federal reimbursement of Texas' costs to implement the Migratory Bird Harvest Information Program, and the Arthur Temple College of Forestry and Agriculture (McIntire-Stennis) at Stephen F. Austin State University. We thank the Rumsey Research and Development Fund for additional financial assistance. Also, we thank E. Woolverton, E. Bowman, G. Rhodes, M. Symmank, J. Varnell, C. Green, S. Tyler, A. Mangiameli, H. and R. Skidmore, R. Henderson, and C. Davis for field and logistical support.
DATA AVAILABILITY
The data and code that support the findings of this study are openly available at figshare using the link: (https://figshare.com/s/863febc7085719542b6e) Ethical approval for this research was granted by Stephen F. Austin State University.
LITERATURE CITED
Anderson, J. T., L. M. Smith, and D. A. Haukos. 2000. Food selection and feather molt by non-breeding American Green-winged Teal in Texas. Journal of Wildlife Management 64:222-230. https://doi.org/10.2307/3802994
Ankney, C. D. 1979. Does the wing molt cause nutritional stress in lesser Snow Geese? The Auk 96:68-72.
Ankney, C. D., and A. D. Afton. 1988. Bioenergetics of breeding Northern Shovelers: diet, nutrient reserves, clutch size, and incubation. The Condor 90:459-472. https://doi.org/10.2307/1368574
Arnold, T. W. 2010. Uninformative parameters and model selection using Akaike's Information Criterion. Journal of Wildlife Management 74:1175-1178. https://doi.org/10.1111/j.1937-2817.2010.tb01236.x
Austin, J. E., and L. H. Fredrickson. 1987. Body and organ mass and body composition of postbreeding female lesser scaup. The Auk 104:694-699. https://doi.org/10.1093/auk/104.4.694
Baldassarre, G. A., R. J. Whyte, and E. G. Bolen. 1986. Body weight and carcass composition of nonbreeding Green-winged Teal on the Southern High Plains of Texas. The Journal of Wildlife Management 50:420-426. https://doi.org/10.2307/3801098
Ballard, B. M., J. E. Thompson, and M. J. Petrie. 2006. Carcass composition and digestive-tract dynamics of northern pintails wintering along the lower Texas coast. Journal of Wildlife Management 70:1316-1324. https://doi.org/10.2193/0022-541X(2006)70[1316:CCADDO]2.0.CO;2
Blem, C. R. 1976. Patterns of lipid storage and utilization in birds. American Zoologist 16:671-684. https://doi.org/10.1093/icb/16.4.671
Burnham, K. P., and D. R. Anderson. 2002. Model Selection and Multimodel Inference: A Practical Information Theoretic Approach Second edition. Springer-Verlag, New York, New York, USA.
Carney, S. M. 1992. Species, age and sex identification of ducks using wing plumage. U.S. Deptartment of the Interior, U.S. Fish and Wildlife Service, Washington, DC, USA.
Collins, D. P. 2012. Moist-soil managed wetlands and their associated vegetative, aquatic invertebrate, and waterfowl communities in east-central Texas. Dissertation for Ph.D., Stephen F. Austin State University, Nacogdoches, Texas, USA.
Collins, D. P., W. C. Conway, C. D. Mason, and J. W. Gunnels. 2017. Winter diet of Blue-winged Teal Anas discors, Green-winged Teal Anas carolinensis, and Northern Shoveler Anas clypeata in east-central Texas. Wildfowl 67:87-99.
DeVault, T. L., J. Rhodes, O. E. and L. M. Smith. 2003. Condition indices for wintering American wigeon. Wildlife Society Bulletin 31:1132-1137.
Devries, J. H., L. M. Armstrong, R. J. MacFarlane, L. Moats, and P. T. Thoroughgood. 2008b. Waterfowl nesting in fall-seeded and spring-seeded cropland in Saskatchewan. Journal of Wildlife Management 72:1790-1797. https://doi.org/10.2193/2007-513
Devries, J. H., R. W. Brook, D. W. Howerter, and M. G. Anderson. 2008a. Effects of spring body condition and age on reproduction in mallards (Anas platyrhynchos). The Auk 125:618-628. https://doi.org/10.1525/auk.2008.07055
Drobney, R. D. 1984. Effect of diet on visceral morphology of breeding wood ducks. Auk 101:93-98. https://doi.org/10.1093/auk/101.1.93
DuBowy, P. J. 1985. Seasonal organ dynamics in post-breeding male Blue-winged Teal and Northern Shovelers. Comparative Biochemistry and Physiology 82A:899-906. https://doi.org/10.1016/0300-9629(85)90503-1
Euliss, N. D., Jr., R. L. Jarvis, and D. S. Gilmer. 1997. Relationship between waterfowl nutrition and condition on agricultural drainwater ponds in the Tulare Basin, California: waterfowl body condition. Wetlands 17:106-115. https://doi.org/10.1007/BF03160722
Fleskes, J. P., J. L. Yee, G. S. Yarris, and D. L. Loughman. 2016. Increased body mass of ducks wintering in California's Central Valley. The Journal of Wildlife Management 80:679-690. https://doi.org/10.1002/jwmg.1053
Fondell, T. F., P. L. Flint, J. A. Schmutz, J. L. Schamber, and C. A. Nicolai. 2013. Variation in body mass dynamics among sites in Black Brant (Branta bernicla nigricans) supports adaptivity of mass loss during moult. Ibis 155:593-604. https://doi.org/10.1111/ibi.12056
Fredrickson, L. H., and T. S. Taylor. 1982. Management of seasonally flooded impoundments for wildlife. United States Fish and Wildlife Service, Resource Publication 148.
Gauthier, G., and J. Bédard. 1985. Fat reserves and condition indices in greater snow geese. Canadian Journal of Zoology 63:331-333. https://doi.org/10.1139/z85-050
Green, A. J. 2001. Mass/length residuals: measure of body condition or generators of spurious results? Ecology 82:1473-1483. https://doi.org/10.1890/0012-9658(2001)082[1473:MLRMOB]2.0.CO;2
Haukos, D. A., J. E. Neaville, and J. E. Myers. 2001. Body condition of waterfowl harvested on the Upper Gulf Coast of Texas, 1986-2000. Texas Tech University, Lubbock, Texas.
Heitmeyer, M. E. 1988. Body composition of female mallards in winter in relation to annual cycle events. The Condor 90:669-680. https://doi.org/10.2307/1368357
Heitmeyer, M. E., and L. H. Fredrickson. 1981. Do wetland conditions in the Mississippi Delta hardwoods influence mallard recruitment? Pages 46-57 in Transactions of the Forty-sixth North American Wildlife and Natural Resources Conference.
Hepp, G. R., R. J. Blohm, R. E. Reynolds, J. E. Hines, and J. D. Nichols. 1986. Physiological condition of autumn-banded mallards and its relationship to hunting vulnerability. The Journal of Wildlife Management 50:177-183. https://doi.org/10.2307/3801893
Hohman, W. L. 1993. Body composition dynamics of ruddy ducks during wing moult. Canadian Journal of Zoology 71:2224-2228. https://doi.org/10.1139/z93-312
Hohman, W. L., C. D. Ankney, and D. L. Roster. 1992. Body condition, food-habits, and molt status of late-wintering ruddy ducks in California. Southeastern Naturalist 37:268-273. https://doi.org/10.2307/3671868
Hohman, W. L., and R. D. Crawford. 1995. Molt in the annual cycle of Ring-necked Ducks. Condor 97:473-483. https://doi.org/10.2307/1369033
Hothorn, T., F. Bretz, and P. Westall. 2008. Simultaneous inference in general parametric models. Biometrical Journal 50:346-363. https://doi.org/10.1002/bimj.200810425
Jakob, E. M., S. D. Marshall, and G. W. Uetz. 1996. Estimating fitness: a comparison of body condition indices. Oikos 77:61-67. https://doi.org/10.2307/3545585
Jenni, L., and S. Jenni-Eiermann. 1998. Fuel supply and metabolic constraints in migrating birds. Journal of Avian Biology 29:521-528. https://doi.org/10.2307/3677171
Johnson, D. H., G. L. Krapu, K. J. Reinecke, and D. G. Jorde. 1985. An evaluation of condition indices for birds. Journal of Wildlife Management 49:569-575. https://doi.org/10.2307/3801673
Kehoe, F. P., and C.D. Ankney. 1985. Variation in digestive organ size among five species of diving ducks (Athya spp.). Canadian Journal of Zoology 63:2339-2342. https://doi.org/10.1139/z85-346
Kehoe, F. P., C. D. Ankney, and R. T. Alisauskas. 1988. Effects of dietary fiber and diet diversity on digestive organs of captive Mallards (Anas platyrhynchos). Canadian Journal of Zoology 66:1597-1602. https://doi.org/10.1139/z88-233
King, R. and A. D. Fox. 2012. Moulting mass dynamics of female Gadwall Anas strepera and male Wigeon A. penelope at Abbertson Reservoir, southeast England. Birdy Study 59:252-254. https://doi.org/10.1080/00063657.2012.655706
Labocha, M. K., and J. P. Hayes. 2012. Morphometric indices of body condition in birds: a review. Journal of Ornithology 153:1-22. https://doi.org/10.1007/s10336-011-0706-1
Miller, M. R. 1986. Molt chronology of northern pintails in California. Journal of Wildlife Management 50:57-64. https://doi.org/10.2307/3801488
Miller, M. R. 1989. Estimating carcass fat and protein in northern pintails during the nonbreeding season. The Journal of Wildlife Management 53:123-129. https://doi.org/10.2307/3801318
Moon, J. A., D. A. Haukos, and L. M. Smith. 2007. Declining body condition of northern pintails wintering in the Playa Lakes Region. Journal of Wildlife Management 71:218-221. https://doi.org/10.2193/2005-596
Moorman, T. E., G. A. Baldassarre, and D. M. Richard. 1992. Carcass mass, composition and gut morphology dynamics of mottled ducks in fall and winter in Louisiana. The Condor 94:407-417. https://doi.org/10.2307/1369213
Morton, J. M., R. L. Kirkpatrick, and M. R. Vaughan. 1990. Changes in body composition in American black ducks wintering at Chincoteague, Virginia. The Condor 92:598-605. https://doi.org/10.2307/1368679
Moser, T. J., and D. H. Rusch. 1988. Indices of structural size and condition of Canada geese. Journal of Wildlife Management 52:202-208. https://doi.org/10.2307/3801223
Murphy, M. E. 1996. Energetics and nutrition of molt. Pages 158-198 in C. Carey, editor. Avian Energetics and Nutritional Ecology. Chapman and Hall, New York, NY, USA. https://doi.org/10.1007/978-1-4613-0425-8_6
Natural Resource Conservation Service (NRCS). 2002. Soil survey of Freestone County, Texas. United States Department of Agriculture.
Odum, E. P., and C. E. Connell. 1956. Lipid levels in migrating birds. Science 123:892-894. https://doi.org/10.1126/science.123.3203.892.b
Owen, M., and W. A. Cook. 1977. Variations in body weight, wing length and condition of Mallard Anas platyrhynchos platyrhynchos and their relationship to environmental changes. Journal of Zoology 183:377-395. https://doi.org/10.1111/j.1469-7998.1977.tb04194.x
Owen, R. B., Jr. 1970. The bioenergetics of capture blue-winged teal under controlled and outdoor conditions. Condor 72:153-163. https://doi.org/10.2307/1366625
Payne, R. B. 1972. Mechanisms and control of molt. Pages 103-155 in D. S. Farner and J. R. King, editors. Avian Biology. Academic Press, New York. https://doi.org/10.1016/B978-0-12-249402-4.50012-7
Piersma, T., A. Koolhaas, and A. Dekinga. 1993. Interactions between stomach structure and diet choice in shorebirds. Auk 110:552-564. https://doi.org/10.2307/4088419
Pinheiro, J., D. Bates, S. DebRoy, D. Sarkar, and R. C. T. 2021. R package version 3.1-152.
Portugal, S. J., J. A. Green, and P. J. Butler. 2007. Annual changes in body mass and resting metabolism in captive barnacle geese (Branta leucopsis): the importance of wing moult. Journal of Experimental Biology 210:1391-1397. https://doi.org/10.1242/jeb.004598
R Core Team. 2021. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria.
Reinecke, K. J., T. L. Stone, and R. B. Owen, Jr. 1982. Seasonal carcass composition and energy balance of female black ducks in Maine. Condor 84:420-426. https://doi.org/10.2307/1367447
Rhodes, O. E., T. L. DeVault, and L. M. Smith. 2006. Seasonal variation in carcass composition of American Wigeon wintering in the Southern High Plains. Journal of Field Ornithology 77:220-228. https://doi.org/10.1111/j.1557-9263.2006.00044.x
Roberts, A. J., and M. R. Conover. 2014. Diet and body mass of ducks in the presence of commercial harvest of brine shrimp cysts in the Great Salt Lake, Utah. The Journal of Wildlife Management 78:1197-1205. https://doi.org/10.1002/jwmg.759
Rohwer, F. C., W. P. Johnson, and E. R. Loos. 2020. Blue-winged Teal (Spatula discors), version 1.0 in A. F. Poole and F. B. Gill, editors. Birds of the World. Cornell Lab of Ornithology, Ithaca, New York, USA. https://doi.org/10.2173/bow.buwtea.01
Smith, L. M., and D.G. Sheeley. 1993. Factors affecting condition of northern pintails wintering in the Southern High Plains. Journal of Wildlife Mangement 57:226-231. https://doi.org/10.2307/3809001
Schmutz, J. A., and C. R. Ely. 1999. Survival of greater white-fronted geese: effects of year, season, sex and body condition. Journal of Wildlife Management 63:1239-1249. https://doi.org/10.2307/3802841
Thompson, J. D., and G. A. Baldassarre. 1990. Carcass composition of nonbreeding Blue-winged Teal and Northern Pintails in Yucatan, Mexico. Condor 92:1057-1065. https://doi.org/10.2307/1368742
Texas Parks and Wildlife Department (TPWD). 2005. Texas Comprehensive Wildlife Conservation Strategy 2005-2010. Texas Parks and Wildlife Department, Austin, Texas, USA.
Tietje, W. D., and J. G. Teer. 1988. Winter body condition of Northern Shovelers on freshwater and saline habitats. Pages 353-376 in M. W. Weller, editor. Waterfowl in Winter. University of Minnesota Press, Minneapolis, Minnesotr, USA.
Whyte, R. J., and E. G. Bolen. 1984. Variation in winter fat depots and condition indices of mallards. Journal of Wildlife Management 48:1370-1373. https://doi.org/10.2307/3801799
Wishart, R. A. 1979. Indices of structural size and condition of American wigeon (Anas americana). Canadian Journal of Zoology 57:2369-2374. https://doi.org/10.1139/z79-308
Woodall, P. F. 1978. Omental fat: a condition index for redbilled teal. Journal of Wildlife Management 42:188-190. https://doi.org/10.2307/3800713
Zuur, A. F., E. N. Ieno, N. J. Walker, A. A. Saveliev, and G. M. Smith. 2009. Mixed Effects Models and Extensions in Ecology with R. Springer, New York, NY, USA. https://doi.org/10.1007/978-0-387-87458-6
Fig. 1
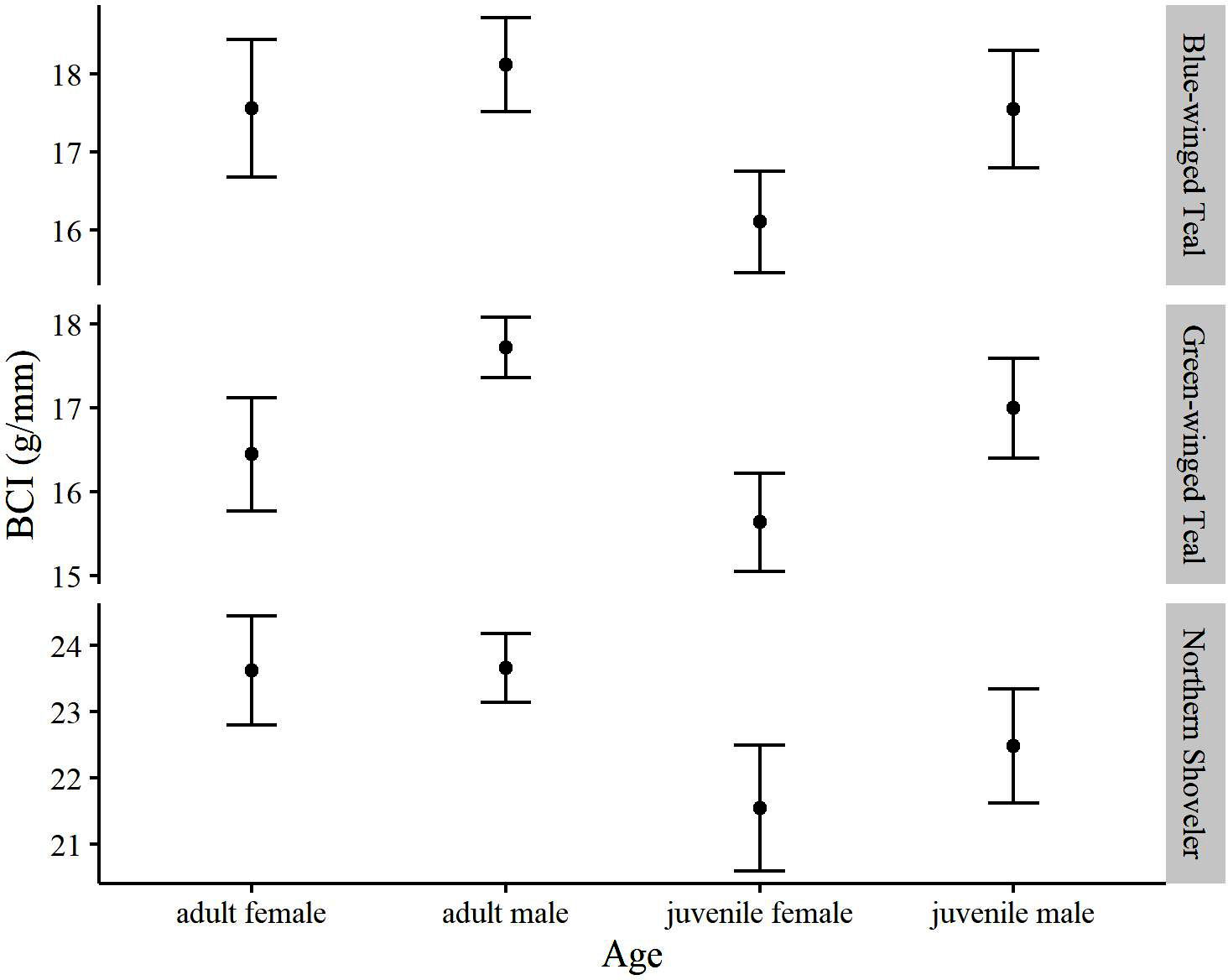
Fig. 1. Mean body condition index (body mass divided by wing chord) across age and sex for focal species collected at the Richland Creek Wildlife Management Area, Texas, in fall to spring, 2004–2007. Black bars are 95% confidence intervals.

Fig. 2
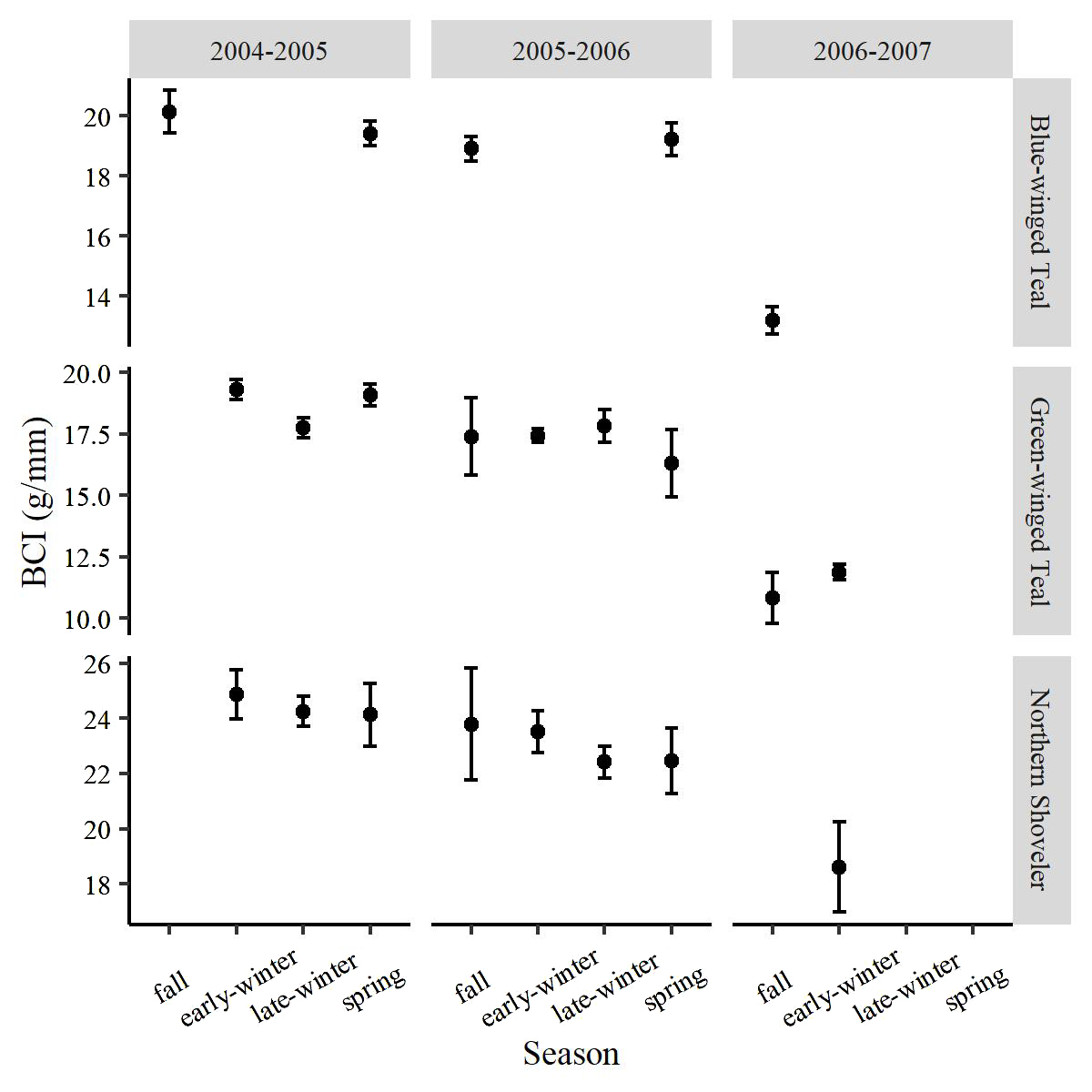
Fig. 2. Mean body condition index (body mass divided by wing chord) for focal species across age and sex, and collected at the Richland Creek Wildlife Management Area, Texas, during fall to spring, 2004–2005, 2005–2006, and 2006–2007. Seasons include fall (1 September – 15 November), early-winter (16 November – 31 December, late-winter (1 January – 28 February), and spring (1 March – 30 March). Black bars are 95% confidence intervals.

Fig. 3
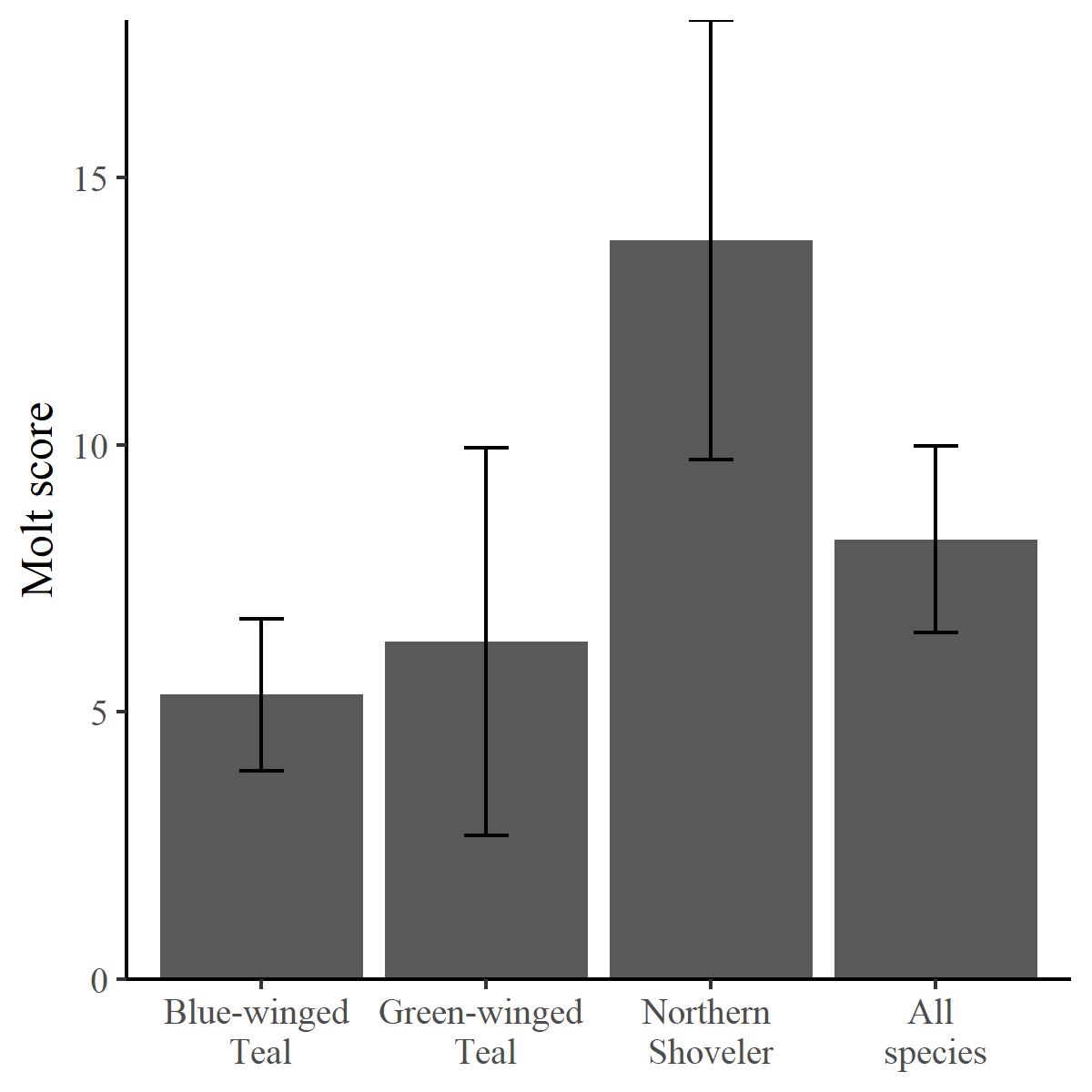
Fig. 3. Mean molt score (total number of erupting feathers divided by the number of feather tracts examined) for focal species collected at the Richland Creek Wildlife Management Area, Texas, in fall to spring, 2004–2007. Black bars are 95% confidence intervals.

Table 1
Table 1. Model selection results for mixed ANOVA of age, sex, collection type, and season on the body condition index (body mass divided by wing chord; g/mm) of three species of scientifically collected and hunter-harvested waterfowl at the Richland Creek Wildlife Management Area, Texas, in fall to spring 2004–2005, 2005–2006, and 2006–2007. Sex includes males and females, age includes adults and juveniles, and season includes fall (1 September – 15 November), early-winter (16 November – 31 December), late-winter (1 January – 28 February), and spring (1 March – 30 March). The difference in Akaike’s Information Criterion (AICc) relative to the smallest AICc is represented by ΔAICc, K is the number of parameters in the model, wi is the AICc weight, and LogeL is the log likelihood of the model.
| Model | K | ΔAICc | LogeL | wi | |
| Blue-winged Teal | |||||
| Collection type + season + sex*age | 9 | 0.000 | -796.678 | 0.886 | |
| Collection type + season*sex*age | 12 | 4.094 | -795.541 | 1.000 | |
| Collection type + sex + age | 8 | 32.742 | -814.099 | 1.000 | |
| Collection type + sex | 6 | 37.026 | -818.324 | 1.000 | |
| Collection type + age | 6 | 38.861 | -819.241 | 1.000 | |
| Collection type | 5 | 41.344 | -821.515 | 1.000 | |
| null | 4 | 50.059 | -826.900 | 1.000 | |
| Green-winged Teal | |||||
| Collection type + season + sex + age | 12 | 0.000 | -1059.006 | 0.574 | |
| Collection type + sex + season*age | 15 | 1.545 | -1056.615 | 0.84 | |
| Collection type + age + season*sex | 15 | 3.046 | -1057.366 | 0.965 | |
| Collection Type + sex + age | 9 | 5.596 | -1064.931 | 1.000 | |
| Collection type + sex | 8 | 22.480 | -1074.408 | 1.000 | |
| Collection type + age | 8 | 39.653 | -1082.994 | 1.000 | |
| null | 6 | 74.446 | -1102.448 | 1.000 | |
| Collection type | 7 | 74.673 | -1101.535 | 1.000 | |
| Northern Shoveler | |||||
| Collection type + age | 8 | 0.000 | -552.009 | 0.558 | |
| Collection type + sex + age | 9 | 2.145 | -552.000 | 0.749 | |
| Collection type + season + age | 11 | 2.379 | -549.926 | 0.919 | |
| Collection type + season*age | 14 | 4.833 | -547.792 | 0.969 | |
| Collection type | 7 | 6.680 | -556.420 | 0.988 | |
| Collection type + sex | 8 | 8.042 | -556.030 | 0.998 | |
| null | 6 | 11.760 | -560.022 | 1.000 | |









