The following is the established format for referencing this article:
Buck, E. J., J. D. Sullivan, C. S. Teitelbaum, D. Brinker, P. C. McGowan, and D. J. Prosser. 2022. An evaluation of transmitter effects on adult and juvenile Common Terns using leg-loop harness attachments. Journal of Field Ornithology 93(4):3.ABSTRACT
Marking birds with transmitters allows for the collection of data that are critical for fully understanding avian life history, but researchers must also be confident that performing such studies is as safe as possible for transmittered individuals. While much could be learned from tracking juveniles across dependency periods and first migration, doing so would require a harness-based attachment method which has not been evaluated on any species of juvenile tern. Therefore, we monitored the reproductive success and behavior of adult Common Terns (Sterna hirundo) and the growth and behavior of juvenile Common Terns after attaching transmitters to adults and juveniles with leg-loop harnesses made of elastic cord. We found that transmittered adults had similar reproductive success to untransmittered controls (hatching success for nests of transmittered adults = 0.553; nests of control adults = 0.665). Transmittered adults also expressed minimal behavioral differences from untransmittered controls when the groups were compared via paired treatment-control nest observations along with observations away from the nest. Transmittered juveniles had similar fledging success and growth rates to untransmittered control juveniles (fledging success for transmittered juveniles = 0.766; control juveniles = 0.817). Transmittered juveniles exhibited slight differences in behavior from controls, with increased rates of preening, although these differences did not appear to be detrimental. Finally, monitoring efforts during the breeding season following transmitter deployment found no difference in the return rate, nesting attempt rate, or hatching success rate based on treatment (P > 0.05). However, despite evidence of an individual retaining its transmitter into fall migration, no individuals retained their transmitters when resighted the following breeding season. While our results show that leg-loop harnesses made of elastic cord present a potential option for transmitter attachment to both adult and juvenile Common Terns, additional testing could provide further insight into potential long-term impacts.RESUMEN
INTRODUCTION
Documenting avian movement using tracking devices has vastly improved our knowledge of many aspects of avian life history (Robinson et al. 2010, Kays et al. 2015). For instance, adult satellite tracking of Caspian Terns (Hydroprogne caspia) and Gull-billed Terns (Gelochelidon nilotica) through multiple migration cycles has revealed information about migration routes and wintering area usage (Lyons et al. 2018, Goodenough and Patton 2020). However, most work on terns (Sternidae) has focused on adults, with the few studies on juveniles implemented via shorter-term device attachment lasting no more than a few months (Keedwell 2003, Whittier and Leslie Jr 2005, Ackerman et al. 2009, Herzog et al. 2020). Due to technological limitations, most previous tracking studies on juvenile terns have focused on a short time span, typically using battery powered transmitters and ending before or soon after fledging (Keedwell 2003, Whittier and Leslie Jr 2005, Ackerman et al. 2009, Herzog et al. 2020). These studies have allowed the collection of valuable data on local movements and survival of young terns before they depart on their first migration.
Many tern species disperse soon after fledging, leaving their movements during this early life stage and the first migration cycle largely unknown, with many individuals not returning to their breeding grounds until their third summer (Greenwood and Harvey 1982, Braasch et al. 2009). The amount of growth between hatch and fledging (Lecroy and Collins 1972), and high mortality rates that occur during the first year of life, pose additional challenges to tracking young terns (Spendelow et al. 2002, Keedwell 2003). However, with technological advancements allowing reduced masses of transmitters, such as solar-powered transmitters for use with the Motus network (Taylor et al. 2017), longer-term attachment on juvenile terns has become feasible. Unfortunately, the suture or adhesive-based attachments used in previous studies on juvenile terns are not compatible with solar charging and remain attached for a particularly limited time period. Therefore, a harness-based attachment may be a viable option when attempting to achieve longer-term device retention on juvenile terns, with potential for multi-year device retention.
While there is a wealth of knowledge that can be gained by tracking birds, especially at life stages representing gaps in our understanding, there are inherent risks associated with placing tracking devices on birds. Tracking devices can reduce survival rates, reproductive success, and drive other negative changes in behavior, inherently compromising the usefulness of the movement data collected (Geen et al. 2019). These negative effects vary in frequency by avian guild, species, and by methodological components such as device mass proportional to body mass and attachment style (Bodey et al. 2018, Geen et al. 2019). Due to the potential experimental biases and other issues associated with deploying a substantially-sized device on a wild bird, it is important to critically evaluate new methods for affixing tracking devices to each avian guild, species, and age class. Although several methods have been successfully used to affix tracking devices to adult terns, including sutures (Bluso-Demers et al. 2010, Loring et al. 2017, Carloni 2018), leg attachments (Nisbet et al. 2011), and harnesses (Lyons et al. 2018), only harness attachment allows for long-term device retention while leaving a solar panel exposed. While large terns have been successfully tracked using harness-based transmitter attachments (Lyons et al. 2018, Goodenough and Patton 2020), early studies in smaller terns of the genus Sterna have exhibited mixed responses to harness-attached transmitters, from no effect to bill entanglement or nest abandonment (Bracey et al. 2016, Paton et al. 2021). This variability in harness effects within tern species reinforces the need to perform thorough evaluations on the effects of tracking devices when conducting a tern biologging study.
Given the limitations of available transmitters and the retention periods of various attachment methods, harness-based attachment for solar-powered transmitters is likely needed for longer-term tracking of juvenile terns. However, the ability to move forward with large scale deployment with such an approach is limited as no previous work has tested this method for Common Terns (Sterna hirundo). In preparation for tagging juvenile Common Terns with harness-mounted transmitters, we conducted a captive bird pilot study using a surrogate species, Japanese Quail (Coturnix japonica; Buck et al. 2021), which indicated that leg-loop harnesses constructed from elastic cord (i.e., Stretch Magic) produced the least skin abrasion and had no effect on juvenile growth. Therefore, we designed this study to expand our work and test the impacts of solar-powered radio-transmitters attached via elastic cord leg-loop harnesses on the reproduction and behavior of adult and juvenile Common Terns. Specifically, we sought to understand reproductive effects by assessing changes in the hatching success and fledging success of nests of transmittered adults implementing paired controls and comparing to the success of the entire colony as well as assessing the fledging success of transmittered juveniles. Similarly, we aimed to understand impacts of marking on behavior of transmittered Common Tern adults and juveniles using a series of behavioral observations with paired treatment-control comparisons.
METHODS
Colony monitoring
This study took place on the Paul S. Sarbanes Ecosystem Restoration Project at Poplar Island (hereafter Poplar Island; 38.762° N, -76.384° W), located in the Maryland, USA, portion of the Chesapeake Bay. Poplar Island uses clean dredged material from the shipping channels leading to the port of Baltimore, Maryland, to rebuild and restore remote island habitat (Erwin et al. 2007). These restoration efforts led to Poplar Island annually serving as breeding habitat for Common Terns (Sullivan et al. 2020). In fact, as of 2018, Poplar Island represents the largest breeding site for Common Terns in Maryland. While Common Terns breed in multiple discrete colonies across Poplar Island, this study focused on the two largest colonies: The first colony, Cell 2C (hereafter 2C colony), supported an estimated 240 pairs of Common Terns in 2021 and is a long but narrow colony characterized by a sandy substrate with partial vegetative coverage that increased throughout the 2021 growing season (Prosser 2022). The term “Cell” refers to a specific sub-section of the restoration site where dredged material has been placed for either current or future wetland or upland development. The second colony, the small “habitat island” constructed to provide nesting habitat for waterbirds within Cell 1B (hereafter 1B colony), supported an estimated 154 pairs of Common Terns in 2021 and is characterized by an oyster shell substrate with vegetative cover (primarily forbs and grasses) that emerged rapidly as the breeding season progressed.
As part of standard ongoing colony monitoring, we marked and monitored all nests within the Common Tern colonies throughout the breeding season (Sullivan et al. 2020). Colony monitoring consisted of researchers walking through the colony, two to three times weekly, in a line abreast formation identifying and marking new nests with wooden stakes, recording the number of eggs and their condition by nest, and capturing juveniles for banding. All detected juveniles old enough for amniotic fluid to have dried were transported to a nearby banding station (just outside the colony) where they were fitted with stainless steel U.S. Geological Survey Bird Banding Lab (BBL) and plastic field readable (PFR) bands, aged to the nearest day (Wails et al. 2014), and weighed to the nearest 0.1 gram before they were returned to the place of capture. When we encountered previously banded juveniles during colony surveys, they were recaptured by hand to confirm identity, weighed to the nearest gram using an electronic scale, and released. Additionally, to inform fledging success estimates for juveniles and to determine adult return rates, we conducted resighting surveys for adult and juvenile terns one to four times weekly using spotting scopes both within the colonies and at nearby staging areas on Poplar Island.
We used data collected during colony monitoring to calculate two metrics of reproductive success: hatching success and fledging success. To determine hatching success, we considered nests with clear signs of at least one chick hatching to be successful whereas we considered nests with signs of depredation (e.g., bill-pierced eggshells and heron tracks), flooding (e.g., buried eggs), or nests that became empty prior to 19 days or after 31 days of clutch initiation to be failed. We assigned an unknown fate to nests that became empty between 19 and 31 days after clutch initiation and we estimated their success using a high-low approach. For the low estimate, we considered unknown nests as failed, whereas, for the high estimate, we considered them as hatched. For an additional measure of hatching success, we also determined the number of juveniles banded per nest. To determine fledging success, we considered juveniles resighted or recaptured after 22 days of age or observed flying to be fledged. We calculated fledging success by dividing the total number of banded chicks by the number confirmed to have fledged. While fledging success has some inherent uncertainty, we have no reason to believe this uncertainty would vary between experimental groups in this study.
Bird capture
We captured adult terns on the nest using walk-in traps and captured juveniles by hand during routine colony checks. For adult trapping, we paired nests: each transmittered adult’s nest (hereafter treatment) was paired with an untransmittered control’s nest (hereafter control). We selected nest pairs at similar developmental stages, choosing nests based on the following criteria: within five days of hatch date (determined using nest initiation date and status at time of initial discovery), visibility for behavioral observations from a temporary blind, and within 20 m of each other. Once we identified a pair of nests for trapping, we replaced the eggs in both nests with plaster-filled dummy eggs and set walk-in traps. We stored live eggs in an incubator set to 38° C throughout the trapping process. Observers stationed in pop-up blinds monitored traps to facilitate rapid tern retrieval upon capture. Observers also remained in the blind during bird processing to monitor captured birds for concerning behavior if they returned to their nest during the observation period. We typically replaced eggs in nests following release of the captured bird and once at least one parent was attending the nest. We chose juveniles to be fitted with transmitters (hereafter treatment) or to serve as untransmittered controls (hereafter control) from juveniles recaptured during colony checks and meeting the following criteria: at least 14 days old, > 75 g (transmitter <1.6% of body mass), and not from the nest of an adult in the transmitter effects trial. For reference, the average chick in this study weighed 86 g at 14 days old. Upon capture, we placed both adults and juveniles in individual paper bags and transported them to the nearby banding station for processing.
Processing
We weighed all birds to the nearest tenth of a gram using a digital scale, took measurements (unflattened wing chord, diagonal tarsus, culmen, and total head length, following Pyle 2008), and if unbanded, we fitted each bird with a BBL and PFR band. We swabbed birds orally for DNA sex determination and fit one bird from each treatment/control pair with a radio transmitter. We used two types of radio transmitters: Lotek NTS-1 Solar Nanotags (Lotek Wireless, Newmarket, Ontario, Canada; hereafter center-weighted transmitter), and Cellular Tracking Technologies Solar LifeTags (Rio Grande, New Jersey, USA; hereafter rear-weighted transmitter). Center-weighted transmitters were approximately 17.4 mm long, 14 mm wide, and 5.8 mm tall with an antenna protruding an additional 175 mm. There were two sets of small tubes on the transmitter that served as attachment points, adding ~2 mm in length to both ends of the transmitter body. Rear-weighted transmitters were approximately 20.9 mm in length, 7.4 mm in width, and 2.6 mm in height, with the antenna protruding an additional 174 mm. However, rear-weighted transmitters sat on a 20 mm vinyl strip that served as the attachment point, resulting in the transmitter hanging lower on the body of the bird (Fig. 1). We attached both types of radio transmitters after the methods of Buck et al. (2021) using a leg-loop harness made of 1 mm elastic cord (Stretch Magic, Soft Flex Company, Sonoma, California, USA), secured with a 2 mm sterling silver crimp bead and with the addition of an overhand knot fixed with ethyl cyanoacrylate glue (Krazy Glue®, Columbus, Ohio, USA) immediately above the crimp (Fig. 1). The entire transmitter/harness assembly weighed 1.2 g for rear-weighted transmitters and 2.0 g for center-weighted transmitters. We deployed transmitters on 18 adult and 21 juvenile Common Terns. At the time of transmitter deployment, transmitters averaged 1.8% of adult body mass for center-weighted transmitters (range = 1.6–1.9%), 1.0% of adult body mass for rear-weighted transmitters (0.9–1.1%), and 1.2% of juvenile body mass (1.0 - 1.4%). Juveniles averaged 19 days old at time of marking (14–30 d). Of the transmittered birds, 14 adults and four juveniles were in the 2C colony. However, Great Blue Heron (Ardea herodias) predation became an issue in the 2C colony midway through the nesting season, so we deployed the remaining four adult transmitters and 17 juvenile transmitters in the 1B colony (Table 1).
Following transmitter attachment, we placed treatment birds in a clear plastic bin (approx. 1 x 0.7 x 0.5 m) for approximately two to five min. prior to release to monitor for abnormal behavior such as bow-legged walking or intense stress. We processed control birds before treatment birds and replaced them in a paper bag until the treatment bird was fully processed. This way we restrained treatment and control birds for a similar amount of time and they could be simultaneously released. Upon release, we visually tracked adult terns for either three minutes or until out of sight to check for any immediate behavioral impacts or differences between treatment and control birds. If a released bird landed within sight of the release location, we visually monitored it using a nearby untransmittered bird as a control. We processed juveniles in the same manner as adults, except treatment birds were all fitted with rear-weighted transmitters. As with adults, we handled control juveniles in the same manner and for a similar amount of time as treatment juveniles. We released juveniles near the point of capture in their natal colony. We also monitored juveniles for adverse behavioral reactions immediately following release by video recording each bird until it disappeared or for a maximum of one minute. The number of individuals included in the study by age, colony, and transmitter type is provided in Table 1. It should be noted that data on measurements collected from each individual, as well as processing time, are available in the USGS ScienceBase: https://doi.org/10.5066/P9IF16T5.
Behavioral observations
For three to four days starting the day after transmitter deployment, we simultaneously observed treatment/control nest pairs in the 2C colony from a pop-up ground blind for at least 30 minutes per day. Observers used binoculars and spotting scopes to record behaviors of the two captured birds and their mates, with point-in-time observations recorded for each nest every 60 seconds, with 30 seconds separating an observation of the treatment and control nest. We characterized behaviors using shorthand codes (Table A1.1), where the primary code denoted location (e.g., incubating, by nest, no bird) and the secondary code denoted specific behavior as applicable (e.g., preening, panting). We also noted any unusual observations outside predetermined codes. For adult treatment/control paired nests, we monitored nests once per week after the initial three-day post-transmitter deployment observation period until nests were complete (hatched or failed). We were unable to conduct detailed behavioral observations in the 1B colony due to vegetation obstructing the view of birds when on the ground in the colony.
In addition to behavioral nest observations, we visually monitored adults and juveniles away from the nest to compare the behavior of transmittered and untransmittered birds. We surveyed major loafing areas on Poplar Island at least once per week to resight banded birds and to search for transmittered birds. During these surveys, equal effort was made to identify all banded birds in an area. Although much of our resighting effort was conducted without the aid of receivers and we did not use handheld receivers to triangulate or home on birds for resighting/behavioral observations, we used receivers to record which transmittered birds were present in an area (with low spatial precision), which may have biased our search. When we encountered a transmittered bird, we took an observation for up to 10 minutes using point time observations on a 1-min interval in the same manner as the nest observations and using the same behavioral codes. When possible, we opportunistically chose a control of equivalent age class to the transmittered tern from among nearby banded terns and observed it simultaneously with the transmittered bird. Like nest observations, we took observations of controls on a 30-second offset from transmittered birds. In some cases, logistical constraints necessitated that we take observations of untransmittered controls immediately after the observation of the transmittered bird ended. When we recaptured a transmittered juvenile, we also evaluated it for any transmitter-related issues: we visually inspected the region under the thigh most prone to abrasion from the leg-loop material, checked the tension by gently tugging on the transmitter, and evaluated the bird’s overall condition. We did not recapture transmittered juveniles other than when encountered during routine colony surveys and never recaptured transmittered adults.
Molecular sex determination
We determined the sex of treatment and control terns via molecular sexing using cheek swabs collected during bird processing. Swabs were stored on ice until return to the laboratory (within 12 hours) where they were stored at -20° until extraction and PCR testing following the methods outlined in Brubaker et al. (2011).
Subsequent detection and adult success
We regularly downloaded transmitter detection data from the Motus network using the R package ‘motus’ (Birds Canada 2022). To improve data reliability, we only considered detection events registering at least three consecutive signals from rear-weighted transmitters and at least five consecutive signals from center-weighted transmitters (Birds Canada 2022). After data cleaning, we investigated detections starting from the time birds left the breeding grounds in 2021, the year of transmitter deployment. Additionally, as part of ongoing colony monitoring, we attempted to identify previously banded individuals on Poplar Island during the 2022 breeding season. As previously described, resighting was conducted with a spotting scope throughout all colonies on site. In 2022, this effort included identifying which banded individuals were at each nest. We used these data to determine the return rate of treatment and control adults, as well as hatching success for birds with confirmed nest locations. Each transmittered or previously transmittered bird seen was carefully examined using binoculars or spotting scope for any indication of transmitter retention. Although we would have preferred to recapture individuals to assess transmitter retention and body condition, concerns of potentially circulating highly pathogenic avian influenza precluded capture efforts in 2022.
Statistical analysis
Breeding success
We examined the effects of capture and transmitter attachment on hatching success and fledging success using the base functionality for generalized linear models (GLMs) in R 4.1.2 (R Core Team 2021). We modeled hatching success (binary variable, any chicks hatched) using a binomial generalized linear model with a logit link. Predictor variables were treatment (categorical variable, three levels: reference [no capture], control [capture and banding only], treatment [capture + banding + transmitter]), colony (binary variable, 1B or 2C), and sex of captured bird (categorical variable, male, female, or unknown). Because sexes were unknown for most birds, including for all birds in the reference group, we built two separate models: one using data from sexed birds that included sex as a predictor variable and another that included all data but did not include sex as a predictor. We tested for differences among groups using Tukey post hoc tests in the emmeans package (Lenth 2021). We found no evidence that sex was related to hatching success, so we present models with the full data in Results. We also modeled the number of chicks banded per nest (i.e., our second metric of hatching success) using a generalized linear model with Poisson error structure and log link function, followed by Tukey post hoc tests. This model included the same predictor variables as the hatching success model. In both models, for reference birds (i.e., not captured), we only included nests that survived until at least seven days before estimated hatch date, which was the median number of days before estimated hatch at which treatment and control adults were captured. We included all treatment and control nests regardless of days until hatch.
To compare fledging success of offspring among adult categories, we excluded nests with transmittered juveniles from the reference group; no known offspring of birds in the adult trial were treatment or control juveniles. Fledging success was very low in the 2C colony, which contained most of our treatment and control birds; this combination of low fledging success and low sample sizes (n = 14 treatment birds included in this analysis) meant that we observed a fledging rate of zero in our treatment group and only one fledged bird in our control group, which precluded use of GLMs for this analysis. Instead, we used nonparametric bootstrapping to understand whether fledging success differed across bird groups within the same colony. We performed 1000 bootstrap replicates in which we randomized fledging success within each colony (i.e., maintained relationships between colony ID and mean fledging success but not between treatment group and fledging success). We then calculated pairwise differences in mean fledging success between bird groups for each simulation and compared the distribution of these pairwise differences to those in the observed data. We considered differences between treatment groups to be statistically significant if the difference in the observed data was greater than the 97.5% quantile or lower than the 2.5% quantile in the bootstrapped data.
Finally, to quantify the success of transmittered juveniles, we compared fledging success of treatment, control, and reference juveniles using a binomial GLM and Tukey post hoc tests (Fledge~Treatment + Colony + Sex). As with the other breeding success models, we built two separate models: one using data from sexed birds that included sex as a predictor variable and another that included all data but did not include sex as a predictor. We again tested for differences among groups using Tukey post hoc tests and found no evidence that sex was related to the fledging success of juveniles, so we present models with the full data in Results. It should be noted that we only included juveniles encountered at 14 days of age in the reference category since that was the minimum age at which a juvenile was eligible for tagging with a transmitter.
Juvenile growth
We estimated growth curves for juveniles based on masses taken at banding and during recaptures of birds up to 30 days old to test for differences in growth rate among treatment, control, and reference groups using general additive mixed models with the gamm function from the ‘mcgv’ package in R and fit using maximum likelihood (Wood 2011). The growth model contained the effects of treatment and sex, with sex of all birds from the reference category identified as “unknown.” To examine changes in body mass as juveniles developed, we added a smooth effect of age (in days) with a cubic regression spline, as well as smooth interactions between age and bird category and between age and sex. To control for correlated errors as birds aged, we included an Ar1 temporal autocorrelation structure (Pinheiro and Bates 2000), as well as the random effect of bird ID nested within colony to account for repeated measures and colony effects, respectively. We assessed models using all-subsets model selection and AICc (Burnham and Anderson 2002).
Behavior
For all behavioral analyses, we included observations taken at least one day after transmitter attachment to allow for an acclimation period. We compared frequency of the primary “location” on-nest behavioral categories as recorded during the first three days after adult transmitter deployment among sex as well as the four adult tern categories: Treatment, Mate of Treatment, Control, and Mate of Control. For the comparison, we used a multinomial model in the package ‘nnet’ and Tukey post hoc test using the package lsmeans (Lenth 2016). Since excessive preening is a readily observable indication of transmitter-related discomfort (e.g., Paton et al. 2021), we also compared time spent preening among the four groups using a binomial generalized linear model and pairwise comparisons in the package emmeans. For off-nest observations of adults, we used a multinomial model and Tukey post hoc test in lsmeans to compare between treatment and control birds. We also compared the amount of time preening off nest between the treatment and control groups in the same manner as the on-nest observations. We compared transmittered juvenile behavioral observations using the same tests as the off-nest observations of adults.
Return rate and future success of adults
We examined the return rate of treatment and control adults in 2022 (the breeding season following transmitter deployment), along with the proportion of these returned birds that nested and successfully hatched chicks (high and low estimates) using generalized linear models. We modeled each binary response variable (i.e., whether a bird returned, nested, or hatched chicks) as a function of treatment group (two levels: control (capture + banding only) or treatment (capture + banding + transmitter)) using binomial generalized linear models with a logit link.
The data that support the findings of this study are openly available in USGS ScienceBase at https://doi.org/10.5066/P9IF16T5. Ethical approval for this research study was granted by the USGS Eastern Ecological Science Center IACUC (2013-05P and 2020-02P), USGS Bird Banding Lab (Banding Permit 23913), and Maryland Department of Natural Resources (Scientific Collection Permit 55439).
RESULTS
Breeding success
We deployed transmitters on 18 adult and 21 juvenile Common Terns. Of the transmittered birds, 14 adults and four juveniles were in the 2C colony. However, Great Blue Heron (Ardea herodias) predation became an issue in the 2C colony midway through the nesting season, so we deployed the remaining four adult transmitters and 17 juvenile transmitters in the 1B colony (Table 1). Post hoc analysis of hatching success indicated no substantial differences between our treatment, control, or reference groups when using either the high estimate (unknown nests considered successful; P > 0.893) or the low estimate (unknown nests considered failed; P > 0.774; Table 2). Similarly, there were no differences in the number of chicks banded per nest based on the parents’ transmitter status (P > 0.543) or colony (P > 0.297). Fledging success was not substantially different among juveniles from nests of treatment and control adults (P = 0.868), though both were lower than those from the reference colony (P = 0.010 and P = 0.018, respectively).
For transmittered juveniles, fledging success was not substantially different among treatment groups (P > 0.678). The 2C colony had lower fledging success than the 1B colony (P = 0.019). Though we present the results of post hoc comparisons in text, all results from main effects models are available in Table A2.1.
Although the fates of birds are not known after departing Poplar Island, we found three transmittered juveniles dead on Poplar Island by homing to their transmitters with a handheld receiver. The first juvenile was in the center of the 1B colony and cause of death was not determined. The other two were in a single Great Blue Heron pellet along with bands from three other reference juveniles, all from the 2C colony. One of the transmittered juveniles found in the pellet had been confirmed as fledged while the other juvenile had not. Additionally, we removed one transmitter from a juvenile in the 1B colony at 47 days of age since it was well past its estimated fledge date yet was only observed flying short distances on multiple flight attempts. The juvenile had a broken primary feather on the right wing, noticed during transmitter removal and possibly impacting its ability to fly. After transmitter removal, it was still unable to sustain flight for normal distances.
Juvenile growth
The most parsimonious model for juvenile growth curves included a smoothed effect of age but no interactions (Mass ~ s(AgeAtEncounter) + 1; Fig. A3.1). This model did not include bird category (i.e., treatment, control, reference), Sex, or Colony. The most parsimonious model that included treatment group also included a smoothed effect of age and an effect of sex (Mass ~ s(AgeAtEncounter, bs = "cr") + BirdType + Sex + 1; Fig. A3.2) and had an AICc of 6.139 lower than the top overall model. Similarly, evidence for the top model (i.e., ratio of model weights) was 21 times larger than the model that included treatment group. A full summary of candidate models is available in Table A3.1.
Behavior
We collected 1427 minutes of behavioral observations of marked adults on paired control-treatment nests. Our multinomial models of primary behavior showed no significant difference between treatment and control adults (F(3,18) = 2.378; P = 0.104; Fig. 2). Although control birds and their mates did not differ in their behavior (F(3,18) = 1.606; P = 0.223), a difference was detected between treatment birds and their mates (F(3,18) = 3.276; P = 0.045). However, post hoc comparisons made within specific behaviors between treatment birds and their mates found no significant differences; the largest difference was in incubation, where treatment birds were observed to spend less time incubating than their mates (df = 18; P = 0.135; Fig. 2). Our independent model of preening behavior showed that control birds preened less than their mates (P = 0.002); no substantial differences existed between treatment and control birds or between control birds and their mates. All results from main effects models are available in Table A2.2.
We collected behavioral observations of adult tern behavior away from the nest (treatment = 68 min, control = 34 min) and found no significant difference in the relative frequency of behavioral categories between treatment and control birds (Fig. 3) or in the amount of time spent preening.
Similarly, behavioral observations of transmittered juveniles (treatment = 316 min, control = 168 min) indicated no substantial difference in frequency of observed primary behavioral categories between treatment and control birds (Fig. 4). However, treatment juveniles spent substantially more time preening than control juveniles (P = 0.009).
Return rate and future success of adults
There were no transmitter detections following departure of treatment birds from Poplar Island, but one individual was reported to us via an external citizen science effort. This resight occurred in Aruba on 5 September 2021 (82 days after transmitter attachment) and includes a photograph clearly showing the transmitter attached to the bird (iNaturalist 2022). However, we did not observe antennae or transmitters on any of the 12 previously transmittered adults that returned to Poplar Island during the 2022 breeding season. Additionally, we did not detect any of the transmitters via the receiving tower after birds returned to the site in 2022. Cumulatively, these data suggest transmitters became detached from transmittered birds at an unknown point after departure from the breeding grounds in 2021.
Transmitter attachment and the subsequent loss of transmitter package appear to have had limited impact on return rates and breeding success of previously transmittered birds in 2022, the year after transmitter deployment. There was no significant difference in the number of adult treatment versus adult control birds that were observed on site in 2022 (12 of 18 treatment, 13 of 18 control, P = 0.718; Table 3). Similarly, there were no differences in the number of treatment versus control birds that were identified at nests (P = 0.313) or in hatching success using either low (P = 0.513) or high nest success estimates (P = 0.937). Full results from these models are available in Table A2.3.
DISCUSSION
Our results suggest that radio transmitters attached via elastic leg-loop harness have little effect on the reproductive success or behavior of adult and juvenile Common Terns within the initial months following transmitter deployment, nor on the return and subsequent success of formerly transmittered adults. For adults, these results are consistent with previous work assessing adult Common Tern response to transmitters, showing minimal effects (Klaassen et al. 1992, Morris and Burness 1992, Paton et al. 2021 but see Nisbet et al. 2011). However, the minimal impacts of transmitters observed with juveniles represent, to our knowledge, the first such successful use of a harness-based attachment method for pre-fledge Common Tern juveniles. The encouraging results of this study open the door for new research into the behaviors of this age group after leaving the breeding grounds. Still, we caution that our study does not include an evaluation of longer-term impacts, the sample size was relatively small, and there are several results that warrant careful examination and continued evaluation. While a large sample size would have allowed us to detect smaller effects, there is an inherent trade-off when testing new methods between statistical power and the need to ensure population safety should there be any adverse impacts. Similarly, while we suspect transmitters remained attached through the first migration it is unclear when detachment occurred. Still, these data present a viable path for researchers to proceed with additional trials as we work toward addressing information gaps.
An interesting, though statistically insignificant, result is that the average hatching success for nests of transmittered adults was lower than the control and reference groups when using our low-estimate approach (Table 2). Similarly, fledging success for offspring of transmittered birds was zero, although still not a statistically significant difference from the control group. This result is somewhat concerning given Paton et al. (2021) documented nest abandonment issues with both Common and Arctic Terns (Sterna paradisaea) when using leg-loop attachments with Teflon ribbon. However, we believe that this trend was at least partially driven by Great Blue Herons depredating juveniles throughout the 2C colony, where the majority of adult transmitters were deployed, causing this colony to experience poor success relative to the 1B colony as well as to its historic performance (Prosser 2020). The heron problem became serious soon after peak hatch and before many juveniles fledged and was underscored by the discovery of a heron pellet casting with transmitters from two juveniles and bands from three additional reference juveniles. We observed one nest abandonment in the 1B colony after transmitter deployment, and although its mate was seen attending the nest in following days, the eggs did not hatch. All other transmittered terns in our study were seen attending their nests in the days immediately following transmitter deployment. Still, the abandonment highlights a possible risk associated with transmitter deployment and the importance of thoroughly evaluating new methodologies for attachment on terns.
We found no evidence for an effect of transmitters on the fledging success or growth rate of transmittered juveniles, suggesting transmitters did not negatively affect growth or survival to fledging. This finding is supported by previous work with Least Tern juveniles (Sternula antillarum; Whittier and Leslie Jr 2005), which also found no detectible mass growth effect from a glue-on transmitter, although that study did not have a control population, instead using published mass data for that species. However, Herzog et al. (2020) found that Forster’s Tern juveniles with suture-attached transmitters had a lower survival rate than non-transmittered birds and exhibited suppressed rate of wing and culmen growth despite no difference in growth rate as measured by body mass. The difference in findings between our study and Herzog et al. (2020) may be driven by the age of juveniles at marking, as Herzog et al. (2020) typically marked juveniles at 3 days, while in our study juveniles were marked after most growth had taken place, at 14 days. This delay in marking may allow for important growth milestones to be reached prior to the additional stressor of transmitter attachment and should be considered for studies where early life stage movements (i.e., activity near the nest) are not of particular interest. Also of note is our finding that the only significant difference in behavior between treatment and control juveniles was that treatment juveniles spent more time preening. There were also two non-significant trends within the primary behavioral categories: treatment birds spent more time than controls loafing (resting on belly with reduced support from legs) and spent more time walking. Although these differences could be indicative of some transmitter effect, the juveniles appeared healthy when recaptured and showed no signs of injury related to the transmitters. Still, our study did not evaluate the effect of transmitters after juveniles departed the colony and should be taken in context.
Another interesting finding was that transmitters had minor impacts on the behavior of both marked adults and their mates. For instance, mates of treatment birds spent the most time of any group incubating, corresponding to treatment birds incubating the least and being away from the nest the most. A dependence on mates to incubate following transmitter attachment has also been observed in Least Terns (Morris and Burness 1992), suggesting that this behavioral change may be a direct response to the transmitter. Although reliance on the mate did not result in a statistically significant decrease in hatching success for marked adults in this study, additional research is warranted. Aside from incubation rate, treatment birds behaved normally relative to untransmittered controls when observed. For example, the amount of time treatment birds spent preening was intermediate among the four groups and was not statistically different from any other group, unlike the observations reported for backpack-style Teflon ribbon attachments (Paton et al. 2021). Additionally, when observed away from the nest, there was no difference between behavior of treatment birds and that of opportunistically chosen controls.
In summary, our results suggest that a leg-loop harness made of elastic cord is a promising potential method for attaching lightweight transmitters to adult Common Terns, with testing to date indicating limited impacts on the behavior of marked birds or their reproductive success, although all adults that returned the year following transmitter deployment had lost their transmitters. Furthermore, this same approach applied to juveniles after much of their growth has occurred does not appear to have a detrimental effect on fledging success or behavior. While some impacts are noted, we do not feel they warrant preclusion of this method for either adults or juveniles. However, this study was limited in duration and size, intending to serve as only an initial evaluation of design potential. Prior to large scale use of this method, future studies deploying transmitters on Sterna terns should continue to monitor for issues with transmitters over longer periods, especially related to nest attendance and reproductive success.
RESPONSES TO THIS ARTICLE
Responses to this article are invited. If accepted for publication, your response will be hyperlinked to the article. To submit a response, follow this link. To read responses already accepted, follow this link.ACKNOWLEDGMENTS
We would like to thank C. Callahan, M. Hack, C. Koppie, M. McHenry, I. Merkel, A. O’Donnell, J. Spendelow, and J. Taylor for assistance in the field; C. Kent for assistance with the growth curve analysis; and N. Karouna for running the molecular sex determinations. Funding for this work was provided by the U.S. Geological Survey Ecosystem Mission Area. Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government.
DATA AVAILABILITY
The data that support the findings of this study are openly available in USGS ScienceBase at https://doi.org/10.5066/P9IF16T5.
LITERATURE CITED
Ackerman, J. T., J. D. Bluso-Demers, and J. Y. Takekawa. 2009. Postfledging Forster’s tern movements, habitat selection, and colony attendance in San Francisco Bay. The Condor 111:100-110. https://doi.org/10.1525/cond.2009.080054
Birds Canada. 2022. Motus: Fetch and use data from the Motus Wildlife Tracking System.
Bluso-Demers, J. D., J. T. Ackerman, and J. Y. Takekawa. 2010. Colony attendance patterns by mated Forster’s Terns (Sterna forsteri) using an automated data-logging receiver system. Ardea 98:59-65. https://doi.org/10.5253/078.098.0108
Bodey, T. W., I. R. Cleasby, F. Bell, N. Parr, A. Schultz, S. C. Votier, and S. Bearhop. 2018. A phylogenetically controlled meta-analysis of biologging device effects on birds: Deleterious effects and a call for more standardized reporting of study data. Methods in Ecology and Evolution 9:946-955. https://doi.org/10.1111/2041-210X.12934
Braasch, A., C. Schauroth, and P. H. Becker. 2009. Post-fledging body mass as a determinant of subadult survival in Common Terns Sterna hirundo. Journal of Ornithology 150:401-407. https://doi.org/10.1007/s10336-008-0362-2
Bracey, A., G. Niemi, and F. Cuthbert. 2016. Lake Superior Common Tern Conservation Final Report.
Brubaker, J. L., N. K. Karouna-Renier, Y. Chen, K. Jenko, D. T. Sprague, and P. F. P. Henry. 2011. A noninvasive, direct real-time PCR method for sex determination in multiple avian species. Molecular Ecology Resources 11:415-417. https://doi.org/10.1111/j.1755-0998.2010.02951.x
Buck, E. J., J. D. Sullivan, C. M. Kent, J. M. Mullinax, and D. J. Prosser. 2021. A comparison of methods for the long-term harness-based attachment of radio-transmitters to juvenile Japanese quail (Coturnix japonica). Animal Biotelemetry 9(32):1-16. https://doi.org/10.1186/s40317-021-00257-9
Burnham, K. P., and D. R. Anderson. 2002. Model Selection and Multimodel Inference: a practical information-theoretic approach. Second edition. Springer-Verlag, New York, NY, USA.
Carloni, J. 2018. Analysis of long-term productivity monitoring and foraging area identification of breeding Common Terns in coastal New Hampshire. Thesis for Master of Science, University of New Hampshire, Durham, NH, USA.
Erwin, R. M., J. Miller, and J. G. Reese. 2007. Poplar Island environmental restoration project: Challenges in waterbird restoration on an island in Chesapeake Bay. Ecological Restoration 25:256-262. https://doi.org/10.3368/er.25.4.256
Geen, G. R., R. A. Robinson, and S. R. Baillie. 2019. Effects of tracking devices on individual birds - a review of the evidence. Journal of Avian Biology 50:1-13. https://doi.org/10.1111/jav.01823
Goodenough, K. S., and R. T. Patton. 2020. Satellite Telemetry Reveals Strong Fidelity to Migration Routes and Wintering Grounds for the Gull-Billed Tern (Gelochelidon nilotica). Waterbirds 42:400-410. https://doi.org/10.1675/063.042.0405
Greenwood, P. J., and P. H. Harvey. 1982. The natal and breeding dispersal of birds. Annual Review of Ecological Systems 13:1-22. https://doi.org/10.1146/annurev.es.13.110182.000245
Herzog, M. P., J. T. Ackerman, C. A. Hartman, and S. H. Peterson. 2020. Transmitter Effects on Growth and Survival of Forster’s Tern Chicks. Journal of Wildlife Management 84:891-901. https://doi.org/10.1002/jwmg.21864
iNaturalist. 2022. Common Tern SM3.
Kays, R., M. C. Crofoot, W. Jetz, and M. Wikelski. 2015. Terrestrial animal tracking as an eye on life and planet. Science 348(6240):aaa2478-1. https://doi.org/10.1126/science.aaa2478
Keedwell, R. J. 2003. Does fledging equal success? Post-fledging mortality in the Black-fronted Tern. Journal of Field Ornithology 74:217-221. https://doi.org/10.1648/0273-8570-74.3.217
Klaassen, M., P. H. Becker, and M. Wagener. 1992. Transmitter Loads Do Not Affect the Daily Energy Expenditure of Nesting Common Terns (Peso del Transmisor no Afecta el Gasto de Energía Diario de Individuos de Sterna hirundo Durante el Periodo de Anidamiento). Journal of Field Ornithology 63:181-185.
Lecroy, M., and C. T. Collins. 1972. Growth and Survival of Roseate and Common Tern Chicks. The Auk 89:595-611.
Lenth, R. V. 2016. Least-squares means: The R package lsmeans. Journal of Statistical Software 69. https://doi.org/10.18637/jss.v069.i01
Lenth, R. V. 2021. emmeans: Estimated Marginal Means, aka Least-Squares Means.
Loring, P. H., R. A. Ronconi, L. J. Welch, P. D. Taylor, and M. L. Mallory. 2017. Postbreeding dispersal and staging of Common and Arctic Terns throughout the western North Atlantic. Avian Conservation & Ecology 12(2):20. https://doi.org/10.5751/ACE-01086-120220
Lyons, D. E., A. G. L. Patterson, J. Tennyson, T. J. Lawes, and D. D. Roby. 2018. The Salton Sea: critical migratory stopover habitat for Caspian Terns (Hydroprogne caspia) in the North American Pacific Flyway. Waterbirds 41:154-165. https://doi.org/10.1675/063.041.0206
Morris, R. D., and G. P. Burness. 1992. A new procedure for transmitter attachment: effects on brood attendance and chick feeding rates by male Common Terns. Condor 94:239-243. https://doi.org/10.2307/1368812
Nisbet, I. C. T., C. S. Mostello, R. R. Veit, J. W. Fox, and V. Afanasyev. 2011. Migrations and winter quarters of five Common Terns tracked using geolocators. Waterbirds 34:32-39. https://doi.org/10.1675/063.034.0104
Paton, P. W. C., P. H. Loring, G. D. Cormons, K. D. Meyer, S. Williams, and L. J. Welch. 2021. Fate of Common (Sterna hirundo) and Roseate Terns (S. dougallii) with satellite transmitters attached with backpack harnesses. Waterbirds 43:342-347. https://doi.org/10.1675/063.043.0315
Pinheiro, J. C., and D. M. Bates. 2000. Mixed-effects models in S and S-Plus. Springer, New York, NY, USA. https://doi.org/10.1007/b98882
Prosser, D. 2022. Post Phase I Dike Construction Faunal Component Surveys of the Poplar Island Environmental Restoration Project, 2021 Field Season. Laurel, MD, USA.
Prosser, D. J. 2020. Post Phase I Dike Construction Faunal Component Surveys of the Poplar Island Environmental Restoration Project, 2019 Field Season. Millersville, Maryland, USA.
Pyle, P. 2008. Identification guide to North American birds: part II Anatidae to Alcidae. Slate Creek Press, Point Reyes Station, California, USA.
R Core Team. 2021. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria.
Robinson, W. D., M. S. Bowlin, I. Bisson, J. Shamoun-Baranes, K. Thorup, R. H. Diehl, T. H. Kunz, S. Mabey, and D. W. Winkler. 2010. Integrating concepts and technologies to advance the study of bird migration. Frontiers in Ecology and the Environment 8:354-361. https://doi.org/10.1890/080179
Spendelow, J. A., J. D. Nichols, J. E. Hines, J. D. Lebreton, and R. Pradel. 2002. Modelling postfledging survival and age- specific breeding probabilities in species with delayed maturity: A case study of Roseate Terns at Falkner Island, Connecticut. Journal of Applied Statistics 29:385-405. https://doi.org/10.1080/02664760120108764
Sullivan, J. D., P. R. Marbán, J. M. Mullinax, D. F. Brinker, P. C. McGowan, C. R. Callahan, and D. J. Prosser. 2020. Assessing nest attentiveness of Common Terns via video cameras and temperature loggers. Avian Research 11:1-18. https://doi.org/10.1186/s40657-020-00208-7
Taylor, P. D., T. L. Crewe, S. A. Mackenzie, D. Lepage, Y. Aubry, Z. Crysler, G. Finney, C. M. Francis, C. G. Guglielmo, D. J. Hamilton, R. L. Holberton, P. H. Loring, G. W. Mitchell, D. R. Norris, J. Paquet, R. A. Ronconi, J. R. Smetzer, P. A. Smith, L. J. Welch, and B. K. Woodworth. 2017. The motus wildlife tracking system: A collaborative research network to enhance the understanding of wildlife movement. Avian Conservation & Ecology 12(1):8. https://doi.org/10.5751/ACE-00953-120108
Wails, C. N., S. A. Oswald, and J. M. Arnold. 2014. Are morphometrics sufficient for estimating age of pre-fledging birds in the field? A test using common terns (Sterna hirundo). PLoS ONE 9(11):e111987. https://doi.org/10.1371/journal.pone.0111987
Whittier, J. B., and D. M. Leslie Jr. 2005. Efficacy of using radio transmitters to monitor least tern chicks. Wilson Bulletin 117:85-91. https://doi.org/10.1676/04-062
Wood, S. N. 2011. Fast stable restricted maximum likelihood and marginal likelihood estimation of semiparametric generalized linear models. Journal of the Royal Statistical Society (B) 73:3-36. https://doi.org/10.1111/j.1467-9868.2010.00749.x
Fig. 1
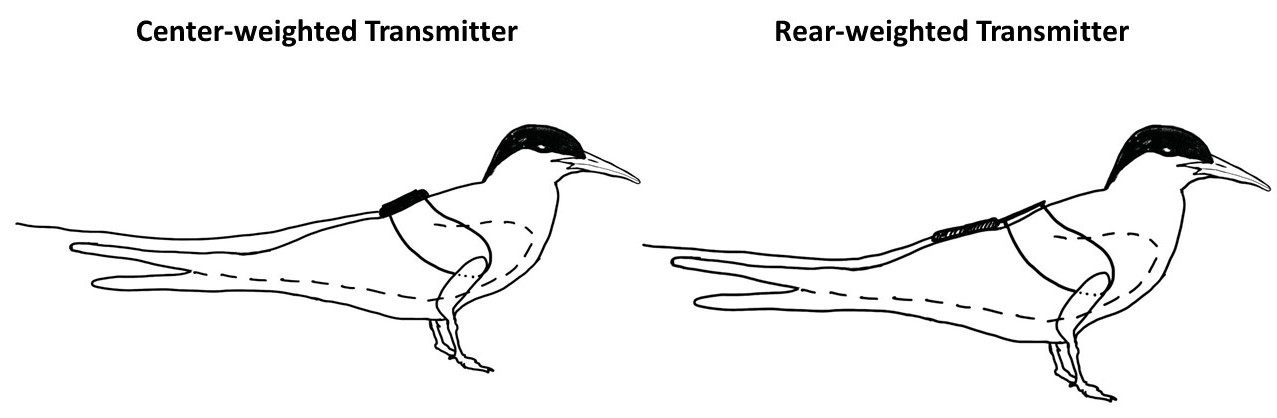
Fig. 1. Radio-transmitter types used in this study. Transmitters were attached to Common Terns (Sterna hirundo) via leg-loop harnesses made of 1 mm elastic cord following the methods of Buck et al. (2021). While images depict adults, rear-weighted transmitters were also applied to juveniles in the same manner; center-weighted transmitters were not attached to juveniles.

Fig. 2
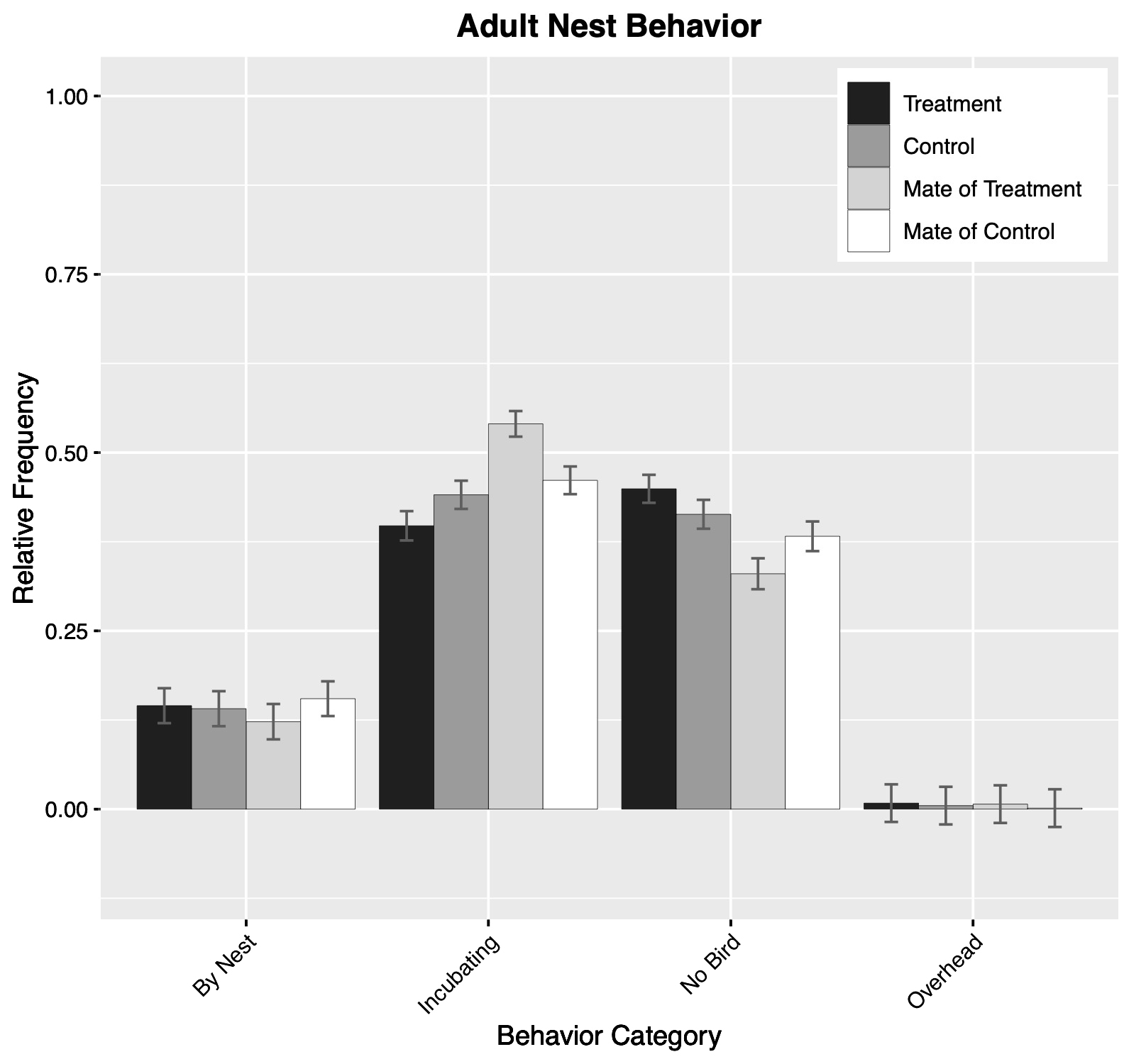
Fig. 2. Estimated frequency of behavioral categories observed at the nest for adult Common Terns (Sterna hirundo) marked with transmitters (treatment), capture-control birds (control), and the mates of each. Estimated frequencies are derived from a multinomial model that estimated behavioral category frequency as a function of treatment group and sex (not shown). Error bars show one standard error of the mean. Note that although proportions are displayed in the plots, equal amounts of time were spent observing terns from each category.

Fig. 3
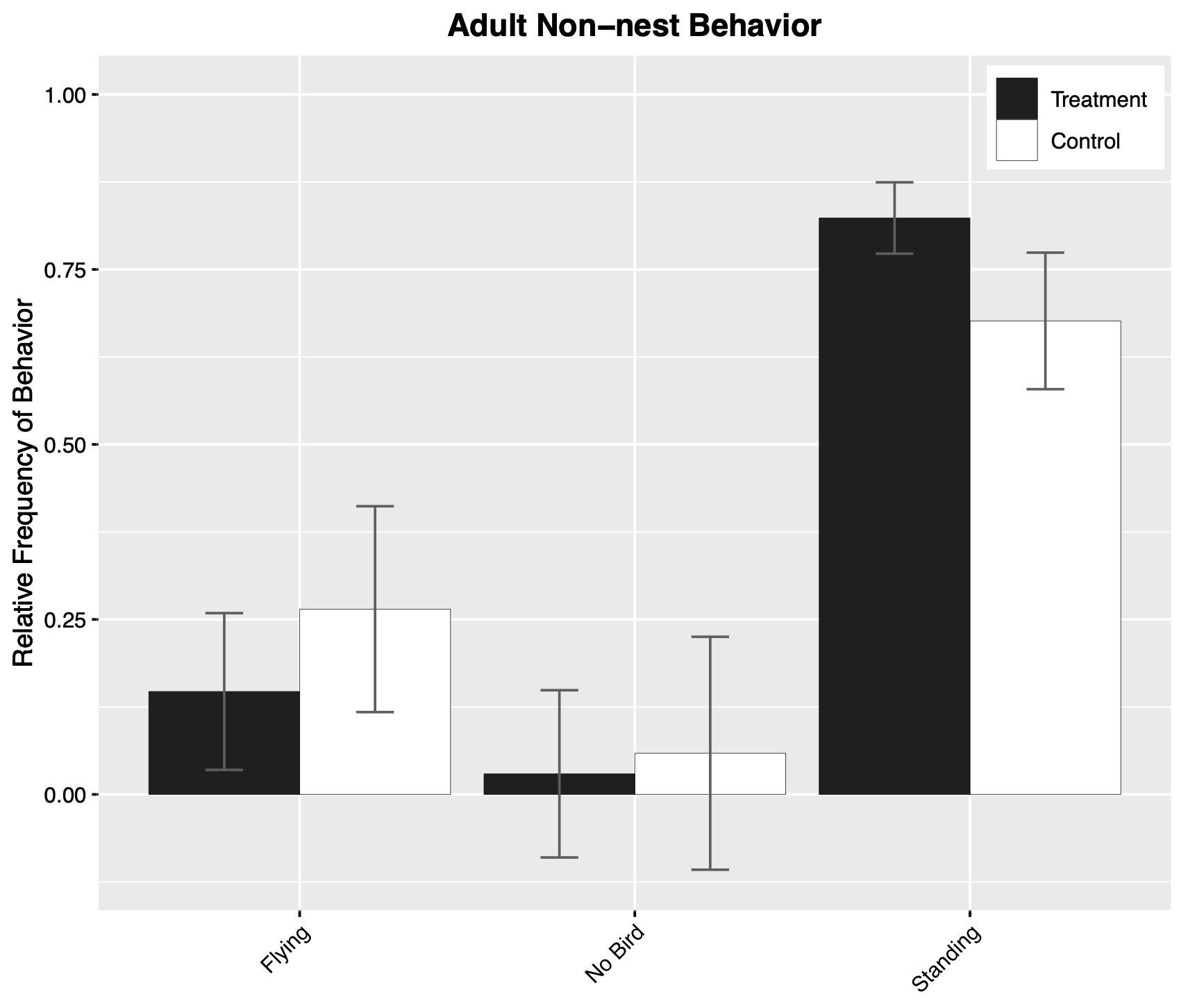
Fig. 3. Estimated frequency of behavioral categories, as observed away from the nest for adult Common Terns (Sterna hirundo) marked with transmitters (treatment) and opportunistically chosen control birds. Estimated frequencies are derived from a multinomial model that estimated behavioral category frequency as a function of treatment group and sex (not shown). Error bars show one standard error of the mean. Note that while proportions are displayed in the plots, unequal amounts of time were spent observing birds from each category.

Fig. 4
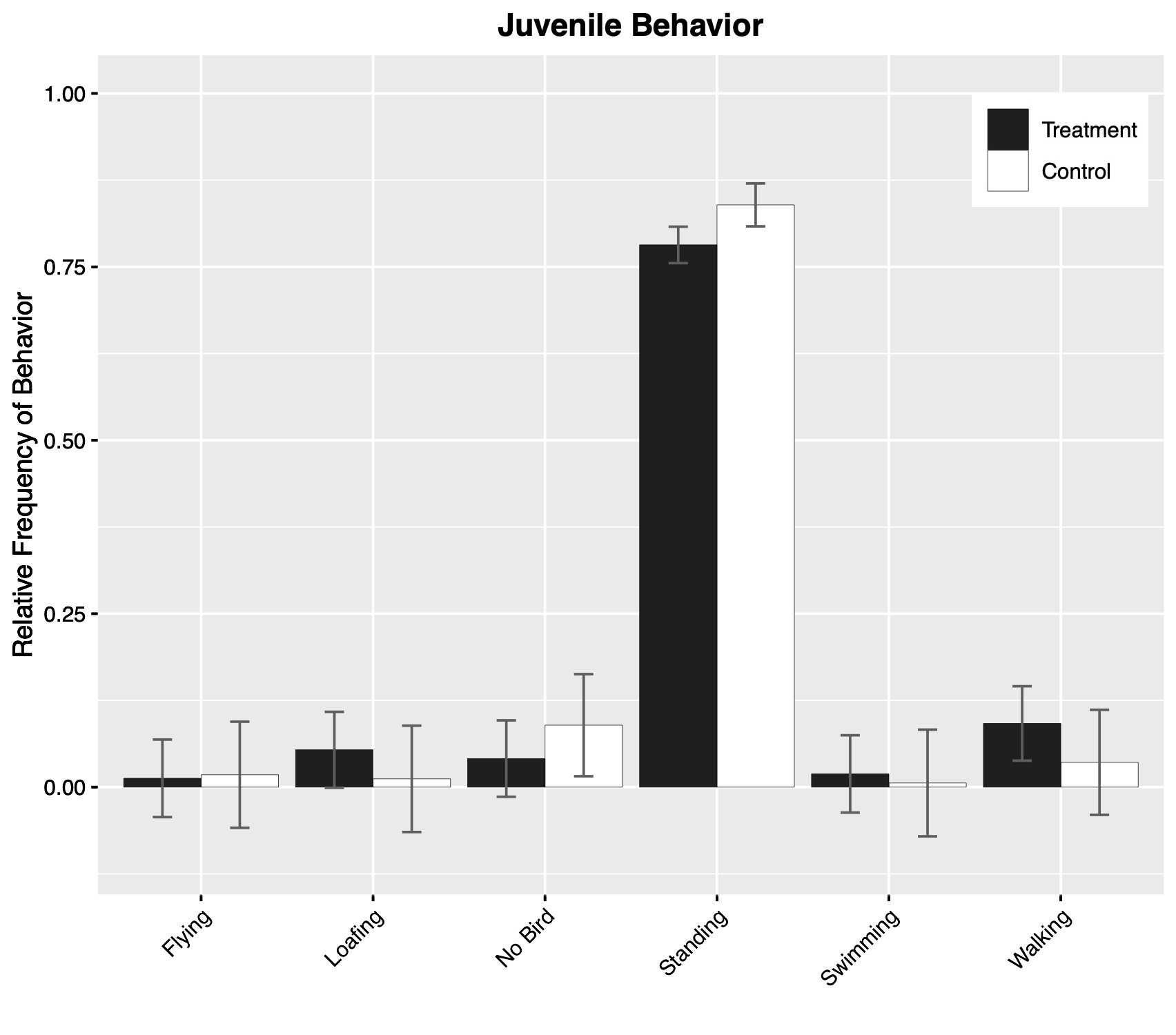
Fig. 4. Estimated frequency of behavioral categories for juvenile Common Terns (Sterna hirundo) marked with transmitters (treatment) and opportunistically chosen control juveniles. Estimated frequencies are derived from a multinomial model that estimated behavioral category frequency as a function of treatment group and sex (not shown). Error bars show one standard error of the mean. Note that while proportions are displayed in the plots, unequal amounts of time were spent observing birds from each category.

Table 1
Table 1. Sample sizes of transmittered Common Terns (Sterna hirundo) by colony, sex, age class, and transmitter type.
| Colony | Transmitter Type | Female | Male | Unknown† | Total |
| Cell 2C Colony | |||||
| Adult Center-weighted | 2 | 5 | 1 | 8 | |
| Adult Rear-weighted | 2 | 3 | 1 | 6 | |
| Adult Control | 7 | 1 | 6 | 14 | |
| Juvenile Rear-weighted | 1 | 3 | 0 | 4 | |
| Juvenile Control | 0 | 4 | 0 | 4 | |
| Cell 1B Colony | |||||
| Adult Center-weighted | 0 | 0 | 0 | 0 | |
| Adult Rear-weighted | 3 | 1 | 0 | 4 | |
| Adult Control | 3 | 1 | 0 | 4 | |
| Juvenile Rear-weighted | 3 | 12 | 2 | 17 | |
| Juvenile Control | 4 | 9 | 3 | 16 | |
| †Molecular sexing failed | |||||
Table 2
Table 2. Success metrics estimated by Tukey post hoc tests based upon the marking status of Common Terns (Sterna hirundo). For analyses of transmittered adults, the reference category includes only nests surviving to within seven days of hatch; for fledging success of transmittered juveniles, the reference category includes only juveniles that reached at least 14 days of age.
| Treatment | Control | Reference | ||||
| Metric | N | Mean [95% CI] | N | Mean [95% CI] | N | Mean [95% CI] |
| Hatching Success Low Estimate | 18 | 0.554 [0.324, 0.760] | 18 | 0.665 [0.425, 0.842] | 336 | 0.598 [0.545, 0.649] |
| 2C Colony | 14 | 0.557 [0.331, 0.762] | 14 | 0.668 [0.429, 0.843] | 176 | 0.601 [0.529, 0.670] |
| 1B Colony | 4 | 0.550 [0.313, 0.766] | 4 | 0.662 [0.411, 0.846] | 160 | 0.595 [0.518, 0.667] |
| Hatching Success High Estimate | 18 | 1.000 [<0.001, 1] | 18 | 0.932 [0.642, 0.991] | 336 | 0.895 [0.855, 0.925] |
| 2C Colony | 14 | 1.000 [<0.001, 1] | 14 | 0.962 [0.764, 0.995] | 176 | 0.940 [0.895, 0.967] |
| 1B Colony | 4 | 1.000 [<0.001, 1] | 4 | 0.882 [0.483, 0.983] | 160 | 0.822 [0.755, 0.873] |
| Juveniles Banded per Nest | 18 | 0.698 [0.404, 1.207] | 18 | 1.020 [0.647, 1.607] | 336 | 0.859 [0.765, 0.965] |
| 2C Colony | 14 | 0.741 [0.429, 1.278] | 14 | 1.083 [0.689, 1.701] | 160 | 0.912 [0.783, 1.062] |
| 1B Colony | 4 | 0.658 [0.371, 1.165] | 4 | 0.961 [0.593, 1.559] | 176 | 0.810 [0.684, 0.959] |
| Fledging Success Juveniles of Transmittered Adults† | 14 | 0.0 | 20 | 0.05 | 255 | 0.24 |
| 2C Colony | 13 | 0.0 | 14 | 0.0 | 152 | 0.09 |
| 1B Colony | 1 | 0.0 | 6 | 0.17 | 103 | 0.47 |
| Fledging Success of Transmittered Juveniles | 21 | 0.766 [0.517, 0.910] | 20 | 0.817 [0.560, 0.940] | 122 | 0.717 [0.617, 0.800] |
| 2C Colony | 4 | 0.667 [0.363, 0.875] | 4 | 0.731 [0.410, 0.914] | 27 | 0.608 [0.431, 0.760] |
| 1B Colony | 17 | 0.843 [0.634, 0.943] | 16 | 0.880 [0.672, 0.963] | 95 | 0.806 [0.718, 0.872] |
| † Mean values for the fledging success of juveniles from transmittered adults are calculated from raw data and confidence intervals are not available because this metric was not suited for analysis with GLM. All other metrics are model-derived means and confidence intervals. | ||||||
Table 3
Table 3. Return rates, nesting rates, and hatching success of Common Terns (Sterna hirundo) tagged with radio transmitters on Poplar Island observed during the following breeding season.
| Treatment Type | Originally Treated | Returned | Nested | Hatched at Least one Chick Low Estimate | Hatched at Least one Chick High Estimate |
| Control | 18 | 13 | 9 | 6 | 8 |
| Treatment | 18 | 12 | 10 | 8 | 9 |









