The following is the established format for referencing this article:
Juhasz, C.-C., J. Dubos, P. Pinet, Y. Soulaimana Mattoir, P. Souharce, C. Caumes, M. Riethmuller, F. Jan, and M. Le Corre. 2022. Discovery of the breeding colonies of a critically endangered and elusive seabird, the Mascarene Petrel (Pseudobulweria aterrima). Journal of Field Ornithology 93(4):11.ABSTRACT
Seabirds are the most endangered group of birds and among them, the gadfly petrels (genera Pseudobulweria and Pterodroma) are the most threatened and least known. The Mascarene Petrel (Pseudobulweria aterrima) is endemic to Réunion Island and is one of the rarest birds in the world. This species was considered extinct in the mid-20th century but was rediscovered in 1970. The population is thought to be in decline because of predation by invasive predators, habitat destruction, and light-induced mortality. The first goal of this paper is to detail the methods that we used to discover the breeding colonies of this species and to determine the threats at these sites. The second goal is to present characteristics of the colonies we found, the threats occurring at these colonies, and the first conservation actions implemented at these sites. We first conducted an island-scale acoustic survey using autonomous recording units (ARUs) to locate the breeding colonies. We then used infrared thermal binoculars to precisely locate the places where birds displayed and landed. Because all discovered breeding sites were on vertical cliffs, we abseiled these cliffs to access the nests. Once burrows were discovered, we deployed infrared camera traps to determine the presence of alien predators or competitors (rats, cats, tenrecs) and to study the behaviors of the birds at the colony. The large-scale acoustic survey revealed the presence of 17 vocally active sites, 16 of which were investigated with infrared thermal binoculars. We observed petrel landings at five of these sites. Two of them were accessible and we abseiled to find the nests. We found 14 occupied burrows at one of these sites and eight at the other. Camera traps revealed the presence of rats and tenrecs at both sites, and cats were detected close to the colonies. The two colonies are on tall vertical cliffs covered with native vegetation, at elevations of 650 m a. s. l. and 1250 m a. s. l., respectively. These findings allowed us to implement conservation actions, such as invasive mammal control, and to start long-term monitoring and applied research for conservation. We are confident that the methods we developed could be used with great success at other places where finding colonies of a cryptic, rare, and nocturnal seabird is particularly challenging.RESUMEN
INTRODUCTION
Seabirds are the most endangered group of birds (Croxall et al. 2012, Dias et al. 2019). More than 30% of the 359 seabird species are considered globally endangered (Dias et al. 2019). A global study investigating population trends of seabirds worldwide has shown a decline of 69.7% in monitored colonies between 1950 and 2010 (Paleczny et al. 2015). This decline is due to numerous man-related threats occurring at sea and on land. The threats affecting most seabird species are predation by invasive alien species at breeding colonies, fishery bycatches, overfishing, hunting/trapping, and climate change/severe weather (Dias et al. 2019). Among seabirds, pelagic species such as albatrosses and petrels are the most endangered (Paleczny et al. 2015, Dias et al. 2019).
The gadfly petrels (genera Pseudobulweria and Pterodroma) constitute one of the most threatened and least known groups of seabirds. Gadfly petrels are threatened by invasive alien species at breeding colonies and light pollution (Rodríguez et al. 2017, Dias et al. 2019). Eradication or control of invasive alien species is one of the major priorities in seabird conservation (Dias et al. 2019), but represents a huge challenge, particularly when several alien species are present simultaneously. For instance, the cumulative effects of feral cat (Felis catus) predation on adults and rat (Rattus spp.) predation on eggs and chicks result in rapid population declines (Dias et al. 2019, Rodríguez et al. 2019). The mass mortality of fledglings caused by artificial lights is a major issue in urbanized islands and archipelagos such as the Canaries, Hawaii, and Réunion Island (Rodríguez et al. 2017). Reduction of light pollution combined with rescue programs of grounded birds are currently the only solutions to significantly reduce this mortality (Troy et al. 2013, Gineste et al. 2017, Rodríguez et al. 2017, 2019).
Among gadfly petrels, the genus Pseudobulweria includes the most threatened seabird species in the world (International Union for Conservation of Nature [IUCN] 2021). It includes five species, of which one is extinct (P. rupinarum), three are on the verge of extinction (the critically endangered P. becki, P. macgillivrayi, and P. aterrima), and one is near threatened (P. rostrata; Warham 1996, Croxall et al. 2012, IUCN 2021). These species are distributed in the tropical Indian and Pacific oceans (Gangloff et al. 2012). The three critically endangered species are endemic to one island or a group of islands (Gangloff et al. 2012). Because of their rarity and very secretive way of life (nocturnal behaviors, burrow nesting on remote and inaccessible cliffs), the biology of these birds is extremely poorly known. Consequently, the breeding sites of all of these species except the Tahiti Petrel (P. rostrata) are still unknown.
The Mascarene Petrel (P. aterrima) is one of the rarest, least known, and most endangered birds of the world (Jouanin 1987, Warham 1990, Le Corre et al. 2003, IUCN 2021). This critically endangered petrel is endemic to Réunion Island and is the only representative of the Pseudobulweria genus in the Indian Ocean. Although eight birds were collected in 1880 and 1891 (Jouanin 1970), the species was believed to be extinct by the mid-20th century (Jouanin 1987). It was rediscovered in 1970 with a dead grounded bird. Two more grounded Mascarene Petrels were found (dead or dying) in 1973 and 1995 (Jouanin 1987, Attie et al. 1997). In 1996, Le Corre et al. (2002, 2003) discovered the extent of the impact of urban light on the mortality of fledgling petrels and shearwaters at Réunion Island. This very important mortality is due to the fact that most petrel and shearwater colonies at Réunion Island are located inland and most cities are coastal, so birds fly over illuminated areas each time they commute from their colonies to the sea (Le Corre et al. 2002, 2003, Chevillon et al. 2022).
Since that time, a rescue operation based on large-scale public awareness campaigns has been made annually by a local non-governmental organization (the Societé d’Etudes Ornithologiques de La Réunion). These campaigns have resulted in the finding of 58 Mascarene Petrels between 1996 and 2021, of which 49 were released successfully (Chevillon et al. 2022). A first estimate of the population size on the basis of sightings at sea between 1978 and 1995 indicated a range of 45 to 400 pairs, with an average estimate of 250 breeding pairs (Attie et al. 1997). The population is believed to have declined because of predation by rats and cats at breeding colonies, habitat destruction (Virion et al. 2020), and light-induced mortality (Le Corre et al. 2003, Chevillon et al. 2022). However, no colony was known until 2016 (this study).
The first objective of this article is to present the different methods used to locate the breeding colonies of the Mascarene Petrel, because we are confident that these methods could be used with success at other places where finding a cryptic, rare, and nocturnal seabird is particularly challenging. Our second objective is to present, for the first time for this species, the main characteristics of its colonies, the threats that are occurring there, and the first conservation measures that we implemented.
METHODS
Automated acoustic survey and record analysis
From July 2015 to April 2017, 21 autonomous recording units (ARUs; six SM4, 11 SM3, and four SM2+ from Wildlife Acoustics, Inc.) were deployed at 68 locations. Recording sites were chosen on the basis of previous acoustic surveys conducted by Riethmuller et al. (2012) and the location of grounded birds attracted to artificial lights (Le Corre et al. 2003, Virion et al. 2020, Chevillon et al. 2022). ARUs were tied to trees within 300 meters of the targeted cliffs. During the first season of searching (July 2015 to April 2016), the ARUs were programmed to record from 8 PM to 5 AM (i.e., from 1h00–2h00 after sunset to 0h30–2h00 before sunrise). During the second season (July 2016 to April 2017), the ARUs were programmed to record from 2 AM to 4 AM (i.e., from 3h30–5h00 before sunrise to 1h30–3h00 before sunrise) on the basis of knowledge acquired during the previous season.
In order to describe temporal changes in vocal activity at the scale of a year and at the scale of a night, we placed an ARU at a site (named Malabar; MB) where Mascarene Petrels were acoustically detected in 2014 (M. Le Corre, P. Pinet, M. Riethmuller, and J. Dubos, unpublished data; see also Results). This ARU was deployed from July 2015 to January 2016 and from July 2016 to April 2017, with the same settings as previously described. First, we calculated the mean number of calls per hour from 1 November 2015 to 12 December 2015. These dates were selected because previous observations suggested that they correspond to a period of intense vocal activity (Riethmuller et al. 2012). This allowed us to determine the time of night when birds are most active vocally. Second, we described the annual pattern of changes of vocal activity during this part of the night using all records obtained on this site.
ARUs were set at 16 kHz level (8 kHz for each microphone) with high pass filter at 220 Hz, because the fundamental frequency of Mascarene Petrel calls is approximately 3 kHz (Riethmuller, personal communication). ARUs used for acoustic surveys were deployed on average for 54 days (minimum: 3; maximum: 213) at a given location, except the ARU at the MB site. Batteries and SD cards were changed at least every two months. The direction of each microphone was recorded and used to determine the direction of the vocalizations. Recordings were retrieved in WAV format. We developed a recognizer with the Kaleidoscope Pro software (Wildlife Acoustics, version 4.5.4) to automatically detect the calls. Each record with positive automated detections was subsequently manually analyzed to determine the direction of the calls and to count the calls.
Nocturnal observations with infrared thermal binoculars
We used infrared thermal binoculars (“Matis” type, Safran) with a video recorder (Espionner Angel Eye Mini DVR 2.5”) to detect petrels at night. Binoculars were used in December 2015 (three weeks), from February to May 2016 (15 weeks), and from October 2016 to March 2017 (24 weeks). Nocturnal observations were made at places where acoustic surveys had revealed the presence of a colony. We chose vantage points with a large view of the cliffs where calls were previously recorded.
For each bird detected, observers noted the species, flight direction, and behaviors (commuting, prospecting, or landing). Birds were observed until they left the vision field or landed. When a bird landed, suggesting the presence of a burrow, we located the landing place precisely on a picture of the site using visual landmarks (tree, rocks, etc.). Identification of the species was made on the basis of flight pattern and vocalizations. The only other nocturnal seabird known to be present at the places where we prospected is the Tropical Shearwater (Puffinus bailloni). Compared to the Mascarene Petrel, the Tropical Shearwater is smaller and has a very rapid flight with almost continuous wing flapping and short gliding. The two other nocturnal seabirds (Barau’s Petrel [Pterodroma baraui] and Wedge-tailed Shearwater [Ardenna pacifica]) have never been recorded on the sites that we prospected. The Barau’s Petrel breeds higher in the mountains (Probst et al. 2000) and the Wedge-tailed Shearwater breeds mostly on coastal cliffs (Faulquier et al. 2017).
Field expeditions and description of the colonies
On the basis of the combined results of acoustic surveys and nocturnal observations, we selected sites with high acoustic activity and observations of landing birds. All of these sites were on vertical cliffs; thus, accessing the spots where landing birds were observed required abseiling. On one of these sites field workers were lowered by helicopter.
Once discovered, the colonies were carefully prospected and all nests were labeled, geo-localized, checked, and measured (height, width of the entrance, and minimum depth to the nearest 5 mm), and their contents noted. In order to start a long-term population monitoring, all accessible birds were banded, weighed (to the nearest 5 g), and measured (wing chord to the nearest mm; culmen length, bill depth, crochet length, and tarsus length to the nearest 0.1 mm). We also described elevation, slope, vegetation type, vegetation structure, and orientation of the cliff of each colony.
Rapid assessment of the threats and conservation actions implemented at breeding colonies
In order to detect invasive predators (cats, rats) or burrow competitors such as the tenrec (Tenrec ecaudatus), we deployed five camera traps (Bushnell Trophy Cam Wireless HD 119598) at the first colony discovered. The tenrec is a hedgehog-like mammal endemic to Madagascar and introduced to Réunion Island. It frequently uses natural cavities and burrows of other species, including seabirds (M. Le Corre and J. Dubos, personal observations). Camera traps were configured with a night-only, motion-trigger sensor recording mode, with 30 seconds of video and sound recording for each triggering.
Because rats and cats were suspected to be present at the colony or in its immediate vicinity, we implemented active rat and cat control as soon as the colony was discovered. At the first colony, rat control was carried out by dispersing blocks of poison containing bromadiolone 0.005% (n°CAS 28772-56-7). To do so, 25 bait stations were set with two blocks of poison (30 g each) per station, 20 of which were along the path to access the colony (one bait station every 30 m) and five were at the colony. Bait consumption at each station was monitored at each visit to the colony and a consumption rate was calculated by dividing the number of consumed blocks by the total number of spread blocks. Consumed blocks were replaced on a regular basis. In total, 2.2 kg of bait were spread at the bait stations. In addition, 17 kg of rat poison was manually spread in the most inaccessible parts of the cliff (12 kg) and at the colony (5 kg). Two automated, rechargeable percussion traps (A24, Goodnature) equipped with a percussion counter were set at the colony and baited with peanut butter and chocolate. These two traps were deployed at places unreachable by a petrel. The number of percussions was checked during the last visit at the colony.
Indices of the presence of cats (e.g., scats) were observed near the first colony discovered. All scats were removed and cat control was carried out with two cages (Havahart x-large 1-door trap), baited with oiled sardines and deployed on access tracks. Cages were automatically monitored with wireless camera traps (Bushnell Trophy Cam Wireless HD 119598) configured with a motion-trigger sensor. A group of three pictures was recorded for each triggering. Traps were checked remotely and automatically every morning through a picture sent to cell phones. Because of the late discovery of the second breeding colony (see Results) and the presence of rats being confirmed with infrared thermal binoculars, we manually dispersed 10 kg of rat poison at this colony and in its vicinity.
Other methods that were rapidly abandoned
We deployed VHF beeper tags (Biotrack, 3.5 g) on adults stranded in towns as a consequence of light pollution. The tags were attached to three tail feathers with nylon thread and were set with a beep every two seconds. Four telemetry receivers (Lotek SRX800) were installed in Grand Bassin in order to detect the presence of the equipped birds. Because no birds were detected and other methods resulted in the finding of two colonies, we rapidly abandoned this method.
We also used two trained small-sized sniffer dogs (a dachshund and a mongrel dog, both about 10 kg and under three years old) to look for burrows at targeted sites. The dogs were trained with scents of Barau’s and Mascarene petrels (linen impregnated with the smell of stranded birds). The dachshund was particularly lazy and inefficient, and the mongrel, though very active and determined, was totally inefficient in the vertical cliffs where Mascarene petrels were suspected to be. For these reasons, and because we found the colonies using the other methods, we rapidly abandoned this method.
RESULTS
Automated acoustic survey
From July 2015 to April 2017, ARUs were deployed at 68 sites (18 in 2015–2016 and 50 in 2016–2017), representing 14,903.5 h of recordings, of which 75.2% were analyzed (see details in Table 1 and Appendices 1–2). Mascarene Petrels were detected at 17 of these sites (25%; Fig. 1). Most sites with vocal activities (n = 15) were located at Grand Bassin, a remote and deep canyon with almost no human settlement. Two other sites with vocal activities were located at Rivière des Remparts, another remote area with huge vertical cliffs and almost no habitation. On average, the vocal activity was low (mean ± sd = 0.66 ± 2.03 voc/h; range [min; max] = 0.003; 8.410 voc/h) suggesting that most of these sites had small numbers of birds (Fig. 1). However, the vocal activity was greater at two sites (> 1 voc/h): Rein de Dimitile (RD) and Le Trou (see Appendix 3).
Temporal changes of vocal activity at Malabar site
We recorded 1290.75 h at the MB site (966.75 h in 2015–2016 and 324.00 h in 2016–2017; see Appendix 1B). In November and December 2015, the time of the night with the highest vocal activity was between 2 AM and 3 AM (26.5 ± 1.5% of vocalizations; Fig. 2). At the scale of a year, the maximum vocal activity was observed in December and January (Fig. 3).
Infrared thermal binoculars survey
We used infrared thermal binoculars at 16 sites where Mascarene Petrels were detected during the acoustic surveys. This represented 50 sessions totaling 205 h of observations. All sessions took place during the breeding season (see details in Table 2; Fig. 1). At five sites no Mascarene Petrels were observed. In the remaining 11 sites, we made 94 observations of Mascarene Petrels, all of them in flight (Appendix 4). On the basis of these observations, we characterized three types of flight: (1) commuting flight, with the bird flapping almost continuously and flying in a straight line (Appendix 4A); (2) prospecting flight, with the bird flapping, gliding, and circling close to cliffs (Appendix 4B and C); and (3) landing flight, with the bird circling above an area until it landed (Appendix 4D).
We observed landing petrels at five sites. On three of these sites, the number of observations was extremely low despite considerable observation effort, suggesting that very few, if any, birds bred there (Appendix 5). On the two other sites, 12 and 17 birds, respectively, were observed landing, suggesting a more active colony (Appendix 5). These two sites were considered accessible and were selected for cliff surveying by abseiling.
Discovery of the breeding colonies
An initial climbing expedition of three people was conducted on 15 November 2016 at RD to reach the landing area. A team placed at the base of the cliff guided the climbers with walkie-talkies and binoculars to help them to approach the landing area. The colony was found after three hours of searching and abseiling. After this discovery, two expeditions were conducted in December 2016 and February 2017 to complete the census and to monitor the colony. Fourteen burrows were found and 13 birds were banded and measured (Appendix 6). Two burrows contained an egg and one contained a downy chick (wing chord = 31.3 mm; weight = 275 g) during the first visit in November. These three burrows were found empty in December, suggesting a breeding failure, presumably because of rat predation.
The second colony was discovered at Rond des Chevrons (RDC) on 28 February 2017. Four people were first lowered to the cliff by helicopter. They reached the landing area by abseiling 40 m down the cliff. Eight burrows were found containing nine adult birds. Seven of them were caught, banded, and measured (Appendix 6), although two were inaccessible. No chicks or eggs were found during this expedition. Because this discovery occurred toward the end of the breeding season, no other expedition was organized during that season.
Characteristics of the colonies and burrows
The breeding colony of RD is located on the lower part of a tall cliff, 690 m high, facing west. The colony is at an elevation of about 640 m a. s. l. and 100–150 m above a riverbed (Fig. 4A). The colony contains 14 burrows in an area of 800 m² (burrow density: 0.0175 burrow/m²; Table 3). The minimum and maximum distance between burrows is 0.65 m and 40 m, respectively.
The breeding colony at RDC is located on the lower part of a tall cliff, 1050 m high, facing southeast. The colony is at an elevation of 1200 m and 100–150 m above a riverbed (Fig. 4B). Eight burrows are spread into four patches of one to four burrows each, with a total surface of 10,000 m² (burrow density: 0.0008 burrow/m²; Table 3). The minimum and maximum distance between burrows is 0.5 m and 140 m, respectively.
For the two colonies, the habitat is characterized by young indigenous sub-humid forest of the megatherm hygrophilous stage (Cadet 1977), dominated by Olea lancea and the endemic Monimia rotundifolia (Huré 2019). Burrows are dug in humus, under tree roots or rocks. They typically have one to three entries and a single incubation chamber. The mean dimensions of the burrows are presented in Table 3.
Rapid assessment of the threats
The two breeding colonies presented a low level of invasion by exotic plants. However, 15 exotic species were identified at the colonies or in their immediate vicinity, of which five are potentially invasive and worrying: the west Indian lantana (Lantana camara), the Brazilian pepper tree (Schinus terebinthifolia), the Cattley guava (Psidium cattleyanum), the coastal she-oak (Casuarina equisetifolia), and the giant cabuya (Furcraea foetida).
Camera traps at the RD colony recorded 616 videos between November 2016 and January 2017. Mascarene Petrels were the main species observed on videos (95.3%, n = 587), followed by rats (3.2%, n = 20; Appendix 7A and 8), tenrecs (1.3%, n = 8; Appendix 7B), and shrews (Suncus murinus; 0.2%, n = 1). The rats were observed foraging with no sign of interference with petrels. Tenrecs were observed entering active burrows occupied by breeding Mascarene Petrels on three occasions, suggesting possible interference and competition for burrows. Furthermore, one tenrec was observed attacking a Mascarene Petrel before entering a burrow containing a chick. No cats were detected at the breeding colony.
The consumption rate of poison at the bait stations between November 2016 and February 2017 was 83.11% at RD. One percussion from a A24 was recorded in February 2017. No cats were captured and no new feces were found. Cage-traps were retrieved during the last expedition at the RD colony.
VHF tracking of stranded birds and use of sniffer dogs at potential breeding sites
We found two stranded adults that were fit enough for being equipped with VHF transmitters (on 13 and 16 November 2015, at Le Tampon and Sainte-Marie, respectively). Although four receivers were simultaneously deployed at a suspected breeding site (Grand Bassin; Fig. 1), no detection was made. We had no success with the sniffer dog method because all suspected breeding sites were on vertical cliffs that dogs could not access.
DISCUSSION
Advantages of new technologies for seabird research and conservation
Our study shows that the combination of new technologies such as ARUs and infrared thermal binoculars is of particular interest by facilitating the discovery of remote breeding colonies of secretive nocturnal seabirds. It allowed us to locate colonies very efficiently with limited physical effort, searching time, financial costs, and human risks. Infrared thermal binoculars observations and camera traps at colonies were essential for the initial investigations of behaviors of birds and their predators, as well as for providing first assessments of the threats.
ARUs are used in avian research for species richness, abundance estimates (Shonfield and Bayne 2017, Pérez-Granados and Traba 2021), phenology (Blumstein et al. 2011), and behavioral ecology (Blumstein et al. 2011). This technology is also lauded as a useful tool for monitoring rare and elusive species (Blumstein et al. 2011, Holmes et al. 2015) and is recommended when conducting large-scale monitoring of birds in remote locations (see Hill et al. 2006, Venier et al. 2012). This non-invasive and easy-to-use tool reduces disturbance caused by human presence and increases the spatial and temporal scale of studies with minimum effort and time invested in the field. In addition, with current technological advances, ARUs are becoming more affordable with greater portability and longevity in the field (see Hill et al. 2006). However, several limitations have to be considered when using ARUs. First, for bird species that mainly vocalize from the ground (as Mascarene Petrels do; J. Dubos, unpublished observations), the distance and the quality of call detections may be reduced, leading to possible false negatives or impeding the ability to locate the origin of a sound. Second, the very important quantity of data produced leads to painstaking and time-consuming data management and processing (Edney and Wood 2021). Although the use of automated recognition can shorten the process (Knight et al. 2017), the low number of good quality recordings may limit the efficiency of automated detection.
Infrared thermal binoculars are increasingly used in seabird ecology (e.g., Syposz et al. 2021). In comparison to classic nocturnal detection tools such as night vision goggles or radar (Swift and Burt-Toland 2009, Galase 2019), infrared thermal binoculars present strong advantages. For instance, they do not require minimal ambient light, as is the case with night vision goggles (Galase 2019), because detection is based on body temperature only. In contrast to radars, infrared thermal binoculars are a low-cost technology, easy to use and to carry in the field with low maintenance (Orben et al. 2019). Furthermore, at close range (< 500 m) most detected birds can be identified to the species level. Infrared thermal binoculars can be combined with an image recorder so that recorded videos can be reviewed for further analysis. However, detection range of infrared thermal binoculars can be reduced in poor environmental conditions such as foggy or rainy weather, rough topography, or dense vegetation cover. The accuracy and the portability of this technology continues to improve, making it increasingly suitable for rough fieldwork (Gade and Moeslund 2014). This tool was a key element in the success of the present study and we strongly recommend it.
Several other methods are commonly used when looking for secretive seabird species (Gummer et al. 2015, Rayner et al. 2015, 2019, Galase 2019). For instance, tracking birds can help to find a breeding site. Rayner et al. (2015) discovered the first breeding colony of the New Zealand Storm-Petrel (Fregetta maoriana) by radio-tagging 24 birds captured at sea, of which 11 were detected from land by using remote base stations and hand-held telemetry receivers. Two of these equipped birds were found in burrows (Rayner et al. 2015). We tentatively used this method during our project but no equipped birds were detected. This lack of success is probably because of the fact that too few birds were equipped. Because the other methods resulted in the finding of colonies, we rapidly abandoned this method.
Satellite telemetry (Argos transmiters or GPS) has been used more or less successfully to locate breeding colonies of elusive and rare seabirds, with tags deployed on birds captured at sea after having been attracted with chumming (see, e.g., Rayner et al. 2019). However, in our context, we quickly discarded these options because of the enormous difficulties in attracting birds close enough to the boat to be catchable (M. Le Corre, P. Pinet, and J. Dubos, unpublished data).
Conservation scent dogs (so-called “sniffer dogs”) are a powerful olfactory tool for the detection of rare species (Bennett et al. 2020), including cavity-nesting and nocturnal seabirds (reviewed in Bolton et al. 2021). In 2018, Galase identified the first colony of Band-rumped Storm-Petrel (Oceanodroma castro) on Hawaii Island after a conservation scent dog detected 18 potential burrows over an area of 3.4 km² (Galase 2019). In our context, it became apparent that sniffer dogs were unusable because of the topography of the breeding sites.
Implications for conservation
Before the discovery of the two breeding colonies, conservation actions targeting the Mascarene Petrel were limited to rescue campaigns to reduce light-induced mortality (Le Corre et al. 2003, Chevillon et al. 2022) and to small-scale cat controls at suspected breeding sites (Riethmuller et al. 2012; authors, unpublished data). The National Park of Réunion Island was established in 2007 (Fig. 1). Twelve of the 18 potential breeding sites (including the two discovered colonies) are located within this park.
Now that we have discovered two breeding colonies and know how to access them on a regular basis, we are able to implement specific conservation actions that more efficiently target threats at these colonies. Four species of introduced mammals have been detected at the colonies or in their immediate vicinity (rats, cats, tenrecs, and shrews). At least two of them are known to prey upon petrel chicks or adults (rats and cats), and the tenrec is suspected of competing with petrels for burrows. We implemented permanent cat and rat control with an early detection system, using a network of camera traps, to prevent any reinvasion. This strategy was very efficient and resulted in a rapid increase of the breeding success at both colonies. Rat control is now extended to other unmonitored and inaccessible breeding sites by using unmanned aircraft systems (Réunion National Park, personal communication).
Many studies have demonstrated the efficiency of artificial breeding colonies as an active conservation measure, especially for petrels (e.g., Pterodroma cahow, Wingate 1977; Ardenna pacifica, Byrd et al. 1983; Pterodroma leucoptera, Priddel et al. 2006; Pterodroma axillaris, Gummer et al. 2015). Artificial nest boxes limit intra- and inter-specific competition for nests (see Gummer et al. 2015) and reduce predation risk. In 2018, two artificial breeding colonies, consisting of 19 and 20 underground nesting boxes, respectively, were settled near each breeding site (Pinet 2020). The dimensions of these boxes were comparable to those of the natural burrows (see Table 3). These two artificial colonies were equipped with solar-powered acoustic social attraction systems and were permanently freed of rats and cats. The first Mascarene Petrels were observed prospecting at one of these sites within one year (Pinet 2020). We are now monitoring the two sites in order to detect the first breeding attempt.
In the future, the use of predator-proof fences could be explored to prevent colonies from being re-invaded by exotic mammals, but we expect to quickly encounter limitations because of (1) the characteristics of the breeding sites (i.e., high elevation, deep slope, and rough field), and (2) the cost of installing and maintaining the fences. In the longer term, we could translocate chicks from natural colonies impacted by introduced predators to artificial sites where predators have been permanently removed. However, to date this has been impossible because the natural colonies that would be accessible for translocation have produced too few chicks. Removing these chicks may have limited positive demographic impact while significantly jeopardizing existing colonies (Fischer et al. 2022). In addition, there are plans to establish other artificial colonies at places farther from light-polluted areas (Virion et al. 2020).
We found 18 other sites where birds were vocally active. Each of these potential breeding sites probably gathers a small number of pairs. All of these sites are on vertical cliffs, inaccessible to humans but not to rats and cats. Furthermore, these sites are extremely unstable and subject to permanent erosion and collapse. This situation leads to a conservation paradox for this critically endangered species. On the one hand, protecting 18 sites simultaneously against introduced predators is extremely challenging. On the other hand, the fact that the species breeds on 18 discreet sites protects it from environmental stochasticity (cliff collapses, fire, cyclonic events). We intend to implement long-term acoustic monitoring to detect any drop of vocal activity, which may indicate a decline of a given colony. This may allow us to implement targeted conservation actions at these colonies.
Implications for applied research
Applied research is an essential step for a precise assessment of the threats and viability of a population. Since the discovery of the colonies, the phenology, biology, and population dynamics of the species are now studied by using acoustic and mark-recapture tools at the two breeding colonies. First estimations of demographic parameters, such as breeding success and adult survival, are now used to estimate population viability and to measure impacts of the initial conservation actions at colonies. We also described the at-sea distribution (Saunier 2019) and marine habitat selection (Fernandez 2021) of Mascarene Petrels during the non-breeding period using geolocators. Genetic analyses have allowed us to describe the genetic diversity, estimate contemporary effective population size, and search for evidence of population bottleneck (Lopez et al. 2021). Further genetic studies will look for potential genetic structures between colonies that will be used to identify adapted conservation units (Danckwerts et al. 2021).
CONCLUSION
To our knowledge, Mascarene Petrel is now the only critically endangered species of the genus Pseudobulweria whose burrows have been discovered. This discovery represents the cornerstone on which knowledge of the ecology of the Mascarene Petrel is now tremendously increasing, allowing the implementation of effective conservation actions, crucial for the protection of the species. In addition, this study demonstrates the efficiency and the complementarity of non-invasive methods for the discovery of one of the most elusive, rare, and endangered seabird species. Although searching for breeding sites of elusive nocturnal seabirds may present different local challenges, depending on each situation, we are convinced that our research strategy is reproducible and adaptable to other situations and would be of great interest for the conservation of other species facing the same gap of knowledge, particularly endemic nocturnal petrels and shearwaters.
RESPONSES TO THIS ARTICLE
Responses to this article are invited. If accepted for publication, your response will be hyperlinked to the article. To submit a response, follow this link. To read responses already accepted, follow this link.AUTHOR CONTRIBUTIONS
PP & MLC conceived and designed the study. JD, YSM, MR, PS, CC, FJ, MLC, & PP collected data and performed the analyses. CCJ, JD, & MLC participated in the writing. All authors reviewed and contributed to the final version of the manuscript.
ACKNOWLEDGMENTS
This study was funded by the SMAC program, Seabird Multidisciplinary Applied Research for Conservation, co-funded by the European Union (ERDF) and the Région Réunion. This study is a production of the European project LIFE + Petrels (grant number: LIFE13 BIO/FR/000075) co-led by the National Park of Réunion Island, the Réunion University, the Société d’Etudes Ornithologiques de La Réunion (SEOR), and the Office National de la Chasse et de la Faune Sauvage, with financial support from the European Union, the Direction de l’Environnement l’Aménagement et du Logement (DEAL), and the Conseil Départemental of Réunion Island. We thank Gérard Millischer of National Park of Mercantour for lending us the infrared thermal binoculars and for his help in the field.
DATA AVAILABILITY
Data/code sharing not applicable.
LITERATURE CITED
Attie, C., J.-C. Stahl, and V. Bretagnolle. 1997. New data on the endangered Mascarene Petrel Pseudobulweria aterrima: a third twentieth century specimen and distribution. Colonial Waterbirds 20(3):406-412. https://doi.org/10.2307/1521590
Bennett, E. M., C. E. Hauser, and J. L. Moore. 2020. Evaluating conservation dogs in the search for rare species. Conservation Biology 34(2):314-325. https://doi.org/10.1111/cobi.13431
Blumstein, D. T., D. J. Mennill, P. Clemins, L. Girod, K. Yao, G. Patricelli, J. L. Deppe, A. H. Krakauer, C. Clark, K. A. Cortopassi, et al. 2011. Acoustic monitoring in terrestrial environments using microphone arrays: applications, technological considerations and prospectus. Journal of Applied Ecology 48(3):758-767. https://doi.org/10.1111/j.1365-2664.2011.01993.x
Bolton, M., G. Morgan, S. E. Bolton, J. R. F. Bolton, S. Parmor, and L. Bambini. 2021. Teaching old dogs and young dogs new tricks: canine scent detection for seabird monitoring. Seabird 33:35-52. http://www.seabirdgroup.org.uk/seabird-33-35
Byrd, G. V., D. I. Moriarty, and B. G. Brady. 1983. Breeding biology of Wedge-tailed Shearwaters at Kilauea Point, Hawaii. Condor 85(3):292-296. https://doi.org/10.2307/1367063
Cadet, L. J. T. 1977. La végétation de l’Ile de La Réunion: Etude phytoécologique et phytosociologique. Thesis. Université Aix-Marseille III, Marseille, France.
Chevillon, L., J. Tourmetz, J. Dubos, Y. Soulaimana-Mattoir, C. Hollinger, P. Pinet, F.-X. Couzi, M. Riethmuller, and M. Le Corre. 2022. 25 years of light-induced petrel groundings in Reunion Island: retrospective analysis and predicted trends. Global Ecology and Conservation 38:e02232. https://doi.org/10.1016/j.gecco.2022.e02232
Croxall, J. P., S. H. M. Butchart, B. Lascelles, A. J. Stattersfield, B. Sullivan, A. Symes, and P. Taylor. 2012. Seabird conservation status, threats and priority actions: a global assessment. Bird Conservation International 22(1):1-34. https://doi.org/10.1017/S0959270912000020
Danckwerts, D. K., L. Humeau, P. Pinet, C. D. McQuaid, and M. Le Corre. 2021. Extreme philopatry and genetic diversification at unprecedented scales in a seabird. Scientific Reports 11:6834. https://doi.org/10.1038/s41598-021-86406-9
Dias, M. P., R. Martin, E. J. Pearmain, I. J. Burfield, C. Small, R. A. Phillips, O. Yates, B. Lascelles, P. G. Borboroglu, and J. P. Croxall. 2019. Threats to seabirds: a global assessment. Biological Conservation 237:525-537. https://doi.org/10.1016/j.biocon.2019.06.033
Edney, A. J., and M. J. Wood. 2021. Applications of digital imaging and analysis in seabird monitoring and research. Ibis 163(2):317-337. https://doi.org/10.1111/ibi.12871
Faulquier, L., L. Solier, F. X. Couzi, J. Tourmetz, M. Saliman, and M. Le Corre. 2017. Evaluation du Plan De Conservation en faveur du Pétrel de Barau Pterodroma baraui 2008-2016. UMR ENTROPIE, Université de La Réunion/Société d'Etudes Ornithologiques de La Réunion/Parc National de La Réunion, La Réunion, France.
Fernandez, R. 2021. Modélisation de la distribution océanique du pétrel noir de Bourbon (Pseudobulweria aterrima) dans l’océan Indien pendant sa période internuptiale. Université de La Réunion, Saint-Denis, La Réunion, France.
Fischer, J. H., H. U. Wittmer, C. F. Kenup, K. A. Parker, R. Cole, I. Debski, G. A. Taylor, J. G. Ewen, and D. P. Armstrong. 2022. Predicting harvest impact and establishment success when translocating highly mobile and endangered species. Journal of Applied Ecology 59(8):2071-2083. https://doi.org/10.1111/1365-2664.14219
Gade, R., and T. B. Moeslund. 2014. Thermal cameras and applications: a survey. Machine Vision and Applications 25:245-262. https://doi.org/10.1007/s00138-013-0570-5
Galase, N. K. 2019. First confirmed Band-rumped Storm Petrel Oceanodroma castro colony in the Hawaiian Islands. Marine Ornithology 47:25-28.
Gangloff, B., H. Shirihai, D. Watling, C. Cruaud, A. Couloux, A. Tillier, E. Pasquet, and V. Bretagnolle. 2012. The complete phylogeny of Pseudobulweria, the most endangered seabird genus: systematics, species status and conservation implications. Conservation Genetics 13:39-52. https://doi.org/10.1007/s10592-011-0261-6
Gineste, B., M. Souquet, F.-X. Couzi, Y. Giloux, J.-S. Philippe, C. Hoarau, J. Tourmetz, G. Potin, and M. Le Corre. 2017. Tropical Shearwater population stability at Reunion Island, despite light pollution. Journal of Ornithology 158:385-394. https://doi.org/10.1007/s10336-016-1396-5
Gummer, H., G. Taylor, K.-J. Wilson, and M. J. Rayner. 2015. Recovery of the endangered Chatham Petrel (Pterodroma axillaris): a review of conservation management techniques from 1990 to 2010. Global Ecology and Conservation 3:310-323. https://doi.org/10.1016/j.gecco.2014.12.006
Hill, G. E., D. J. Mennill, B. W. Rolek, T. L. Hicks, and K. A. Swiston. 2006. Evidence suggesting that Ivory-billed Woodpeckers (Campephilus principalis) exist in Florida. Avian Conservation and Ecology 1(3):2. https://doi.org/10.5751/ACE-00078-010302
Holmes, S. B., K. Tuininga, K. A. McIlwrick, M. Carruthers, and E. Cobb. 2015. Using an integrated recording and sound analysis system to search for Kirtland’s Warbler (Setophaga kirtlandii) in Ontario. Canadian Field-Naturalist 129(2):115-120. https://doi.org/10.22621/cfn.v129i2.1688
Huré, M. 2019. Caractérisation et modélisation spatiale des habitats de reproduction des pétrels endémiques menacés de La Réunion, le pétrel de Barau (Pterodroma baraui) et le pétrel noir de Bourbon (Pseudobulweria aterrima): implication pour la conservat. Université de La Réunion, Saint-Denis, La Réunion, France.
International Union for Conservation of Nature (IUCN). 2021. The IUCN red list of threatened species. Version 2021-3. IUCN, Cambridge, UK. https://www.iucnredlist.org.
Jouanin, C. 1970. Note taxonomique sur les Petits Puffins, Puffinus lherminieri, de l’Océan Indien occidental. Oiseau et RFO 40:303-306.
Jouanin, C. 1987. Notes on the nesting of Procellariiformes in Réunion. Pages 359-363 in A. W. Diamond, editor. Studies of Mascarene Island birds. Cambridge University Press, Cambridge, UK.
Knight, E. C., K. C. Hannah, G. J. Foley, C. D. Scott, R. M. Brigham, and E. Bayne. 2017. Recommendations for acoustic recognizer performance assessment with application to five common automated signal recognition programs. Avian Conservation and Ecology 12(2):14. https://doi.org/10.5751/ACE-01114-120214
Le Corre, M., T. Ghestemme, M. Salamolard, and F.-X. Couzi. 2003. Rescue of the Mascarene Petrel, a critically endangered seabird of Réunion island, Indian Ocean. Condor 105(2):387-391. https://doi.org/10.1093/condor/105.2.387
Le Corre, M., A. Ollivier, S. Ribes, and P. Jouventin. 2002. Light-induced mortality of petrels: a 4-year study from Réunion Island (Indian Ocean). Biological Conservation 105(1):93-102. https://doi.org/10.1016/S0006-3207(01)00207-5
Lopez, J., N. Nikolic, M. Riethmuller, J. Dubos, P. Pinet, P. Souharce, F.-X. Couzi, M. Le Corre, A. Jaeger, and L. Humeau. 2021. High genetic diversity despite drastic bottleneck in a critically endangered, long-lived seabird, the Mascarene Petrel Pseudobulweria aterrima. Ibis 163(1):268-273. https://doi.org/10.1111/ibi.12864
Orben, R. A., A. B. Fleishman, A. L. Borker, W. Bridgeland, A. J. Gladics, J. Porquez, P. Sanzenbacher, S. W. Stephensen, R. Swift, M. W. McKown, and R. M. Suryan. 2019. Comparing imaging, acoustics, and radar to monitor Leach’s Storm-Petrel colonies. PeerJ 7:e6721. https://doi.org/10.7717/peerj.6721
Paleczny, M., E. Hammill, V. Karpouzi, and D. Pauly. 2015. Population trend of the world’s monitored seabirds, 1950-2010. PLoS ONE 10(6):e0129342. https://doi.org/10.1371/journal.pone.0129342
Pérez-Granados, C., and J. Traba. 2021. Estimating bird density using passive acoustic monitoring: a review of methods and suggestions for further research. Ibis 163(3):765-783. https://doi.org/10.1111/ibi.12944
Pinet, P. 2020. A new success story for Mascarene Petrel conservation, Réunion Island. Indian Ocean Seabird Group Newsletter 7:5-6.
Priddel, D., N. Carlile, and R. Wheeler. 2006. Establishment of a new breeding colony of Gould’s Petrel (Pterodroma leucoptera leucoptera) through the creation of artificial nesting habitat and the translocation of nestlings. Biological Conservation 128(4):553-563. https://doi.org/10.1016/j.biocon.2005.10.023
Probst, J.-M., M. Le Corre, and C. Thébaud. 2000. Breeding habitat and conservation priorities in Pterodroma baraui, an endangered gadfly petrel of the Mascarene archipelago. Biological Conservation 93(1):135-138. https://doi.org/10.1016/S0006-3207(99)00114-7
Rayner, M. J., K. A. Baird, J. Bird, S. Cranwell, A. F. Raine, B. Maul, J. Kuri, J. Zhang, and C. P. Gaskin. 2019. Land and sea-based observations and first satellite tracking results support a New Ireland breeding site for the Critically Endangered Beck’s Petrel Pseudobulweria beckii. Bird Conservation International 30(1):58-74. https://doi.org/10.1017/S0959270919000145
Rayner, M. J., C. P. Gaskin, N. B. Fitzgerald, K. A. Baird, M. M. Berg, D. Boyle, L. Joyce, T. J. Landers, G. G. Loh, S. Maturin, et al. 2015. Using miniaturized radiotelemetry to discover the breeding grounds of the endangered New Zealand Storm Petrel Fregetta maoriana. Ibis 157(4):754-766. https://doi.org/10.1111/ibi.12287
Riethmuller, M., F. Jan, Y. Giloux, and M. Saliman. 2012. Plan national d’actions en faveur du Pétrel noir de Bourbon Pseudobulweria aterrima (2012-2016). Ministère de l’Ecologie, du Développement durable et de l’Energie/Direction de l’Environnement de l’Aménagement et du Logement de La Réunion, La Réunion, France.
Rodríguez, A., J. M. Arcos, V. Bretagnolle, M. P. Dias, N. D. Holmes, M. Louzao, J. Provencher, A. F. Raine, F. Ramírez, B. Rodríguez, et al. 2019. Future directions in conservation research on petrels and shearwaters. Frontiers in Marine Science 6:1-27. https://doi.org/10.3389/fmars.2019.00094
Rodríguez, A., N. D. Holmes, P. G. Ryan, K.-J. Wilson, L. Faulquier, Y. Murillo, A. F. Raine, J. F. Penniman, V. Neves, B. Rodríguez, et al. 2017. Seabird mortality induced by land-based artificial lights. Conservation Biology 31(5):986-1001. https://doi.org/10.1111/cobi.12900
Saunier, M. 2019. Etude de la phénologie, de la distribution et de l’activité en mer des pétrels noirs de Bourbon (Pseudobulweria aterrima). Thesis, Université de La Réunion, Saint-Denis, La Réunion, France.
Shonfield, J., and E. M. Bayne. 2017. Autonomous recording units in avian ecological research: current use and future applications. Avian Conservation and Ecology 12(1):14. https://doi.org/10.5751/ACE-00974-120114
Swift, R., and E. Burt-Toland. 2009. Surveys of procellariiform seabirds at Hawaiˋi Volcanoes National Park, 2001-2005. Pacific Cooperative Studies Unit, University of Hawaiˋi at Mânoa, Honolulu, Hawaiˋi, USA.
Syposz, M., O. Padget, J. Willis, B. M. Van Doren, N. Gillies, A. L. Fayet, M. J. Wood, A. Alejo, and T. Guilford. 2021. Avoidance of different durations, colours and intensities of artificial light by adult seabirds. Scientific Reports 11:18941. https://doi.org/10.1038/s41598-021-97986-x
Troy, J. R., N. D. Holmes, J. A. Veech, and M. C. Green. 2013. Using observed seabird fallout records to infer patterns of attraction to artificial light. Endangered Species Research 22:225-234. https://doi.org/10.3354/esr00547
Venier, L. A., S. B. Holmes, G. W. Holborn, K. A. McIlwrick, and G. Brown. 2012. Evaluation of an automated recording device for monitoring forest birds. Wildlife Society Bulletin 36(1):30-39. https://doi.org/10.1002/wsb.88
Virion, M.-C., L. Faulquier, M. Le Corre, F.-X. Couzi, M. Salamolard, B. Lequette, P. Pinet, J. Dubos, M. Riethmuller, Y. Soulaimanana Mattoir, et al. 2021. Plan national d’action en faveur des pétrels endémiques de La Réunion 2021-2030. UMR ENTROPIE, Université de La Réunion, Société d’Études Ornithologiques de La Réunion, Parc national de La Réunion, La Réunion, France.
Warham, J. 1990. The petrels: their ecology and breeding systems. Academic, London, UK.
Warham, J. 1996. The behaviour, population biology and physiology of the petrels. Academic, London, UK.
Wingate, D. B. 1977. Excluding competitors from Bermuda petrel nesting burrows. Pages 93-102 in S. A. Temple, editor. Proceedings of symposium on management techniques for preserving endangered birds. University of Wisconsin-Madison, Madison, Wisconsin, USA.
Fig. 1
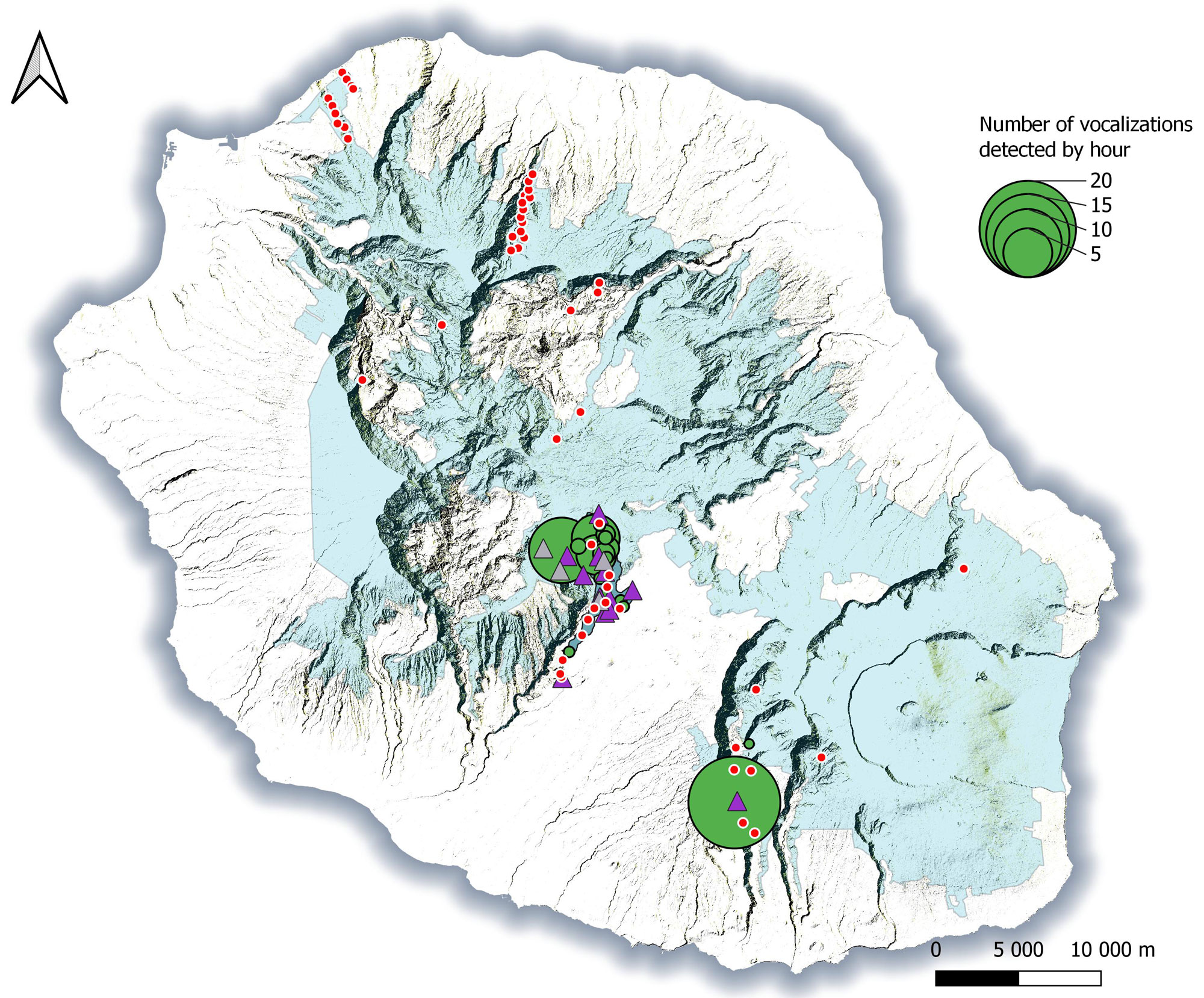
Fig. 1. Locations of autonomous recording units (ARUs; circles) used for acoustic prospection and sites of nocturnal visual prospections with infrared thermal binoculars (triangles), in 2015–2017, Réunion Island. Green and red circles represent sites with and without Mascarene Petrel (Pseudobulweria aterrima) calls recorded, respectively. The size of the green circles is proportionate to the vocalization rate during a month of records. Purple and gray triangles show sites with and without Mascarene Petrel observations, respectively. The blue area indicates the National Park of Réunion Island.

Fig. 2
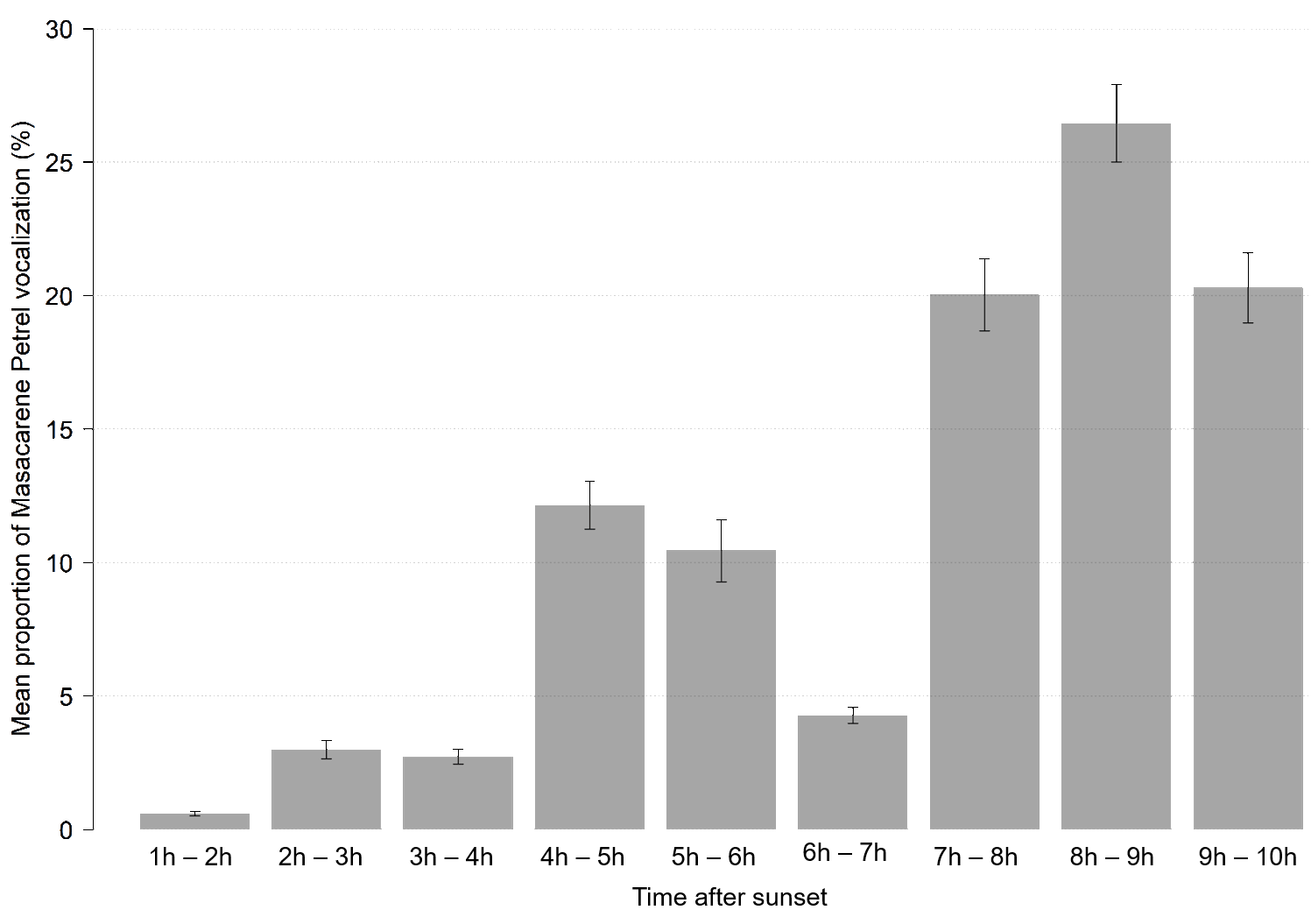
Fig. 2. Temporal change in vocal activity of Mascarene Petrel (Pseudobulweria aterrima) at Malabar, in November and December 2015, Réunion Island. Vocal activity is the proportion of calls recorded per hour after sunset (out of the total number of calls recorded during the period).

Fig. 3
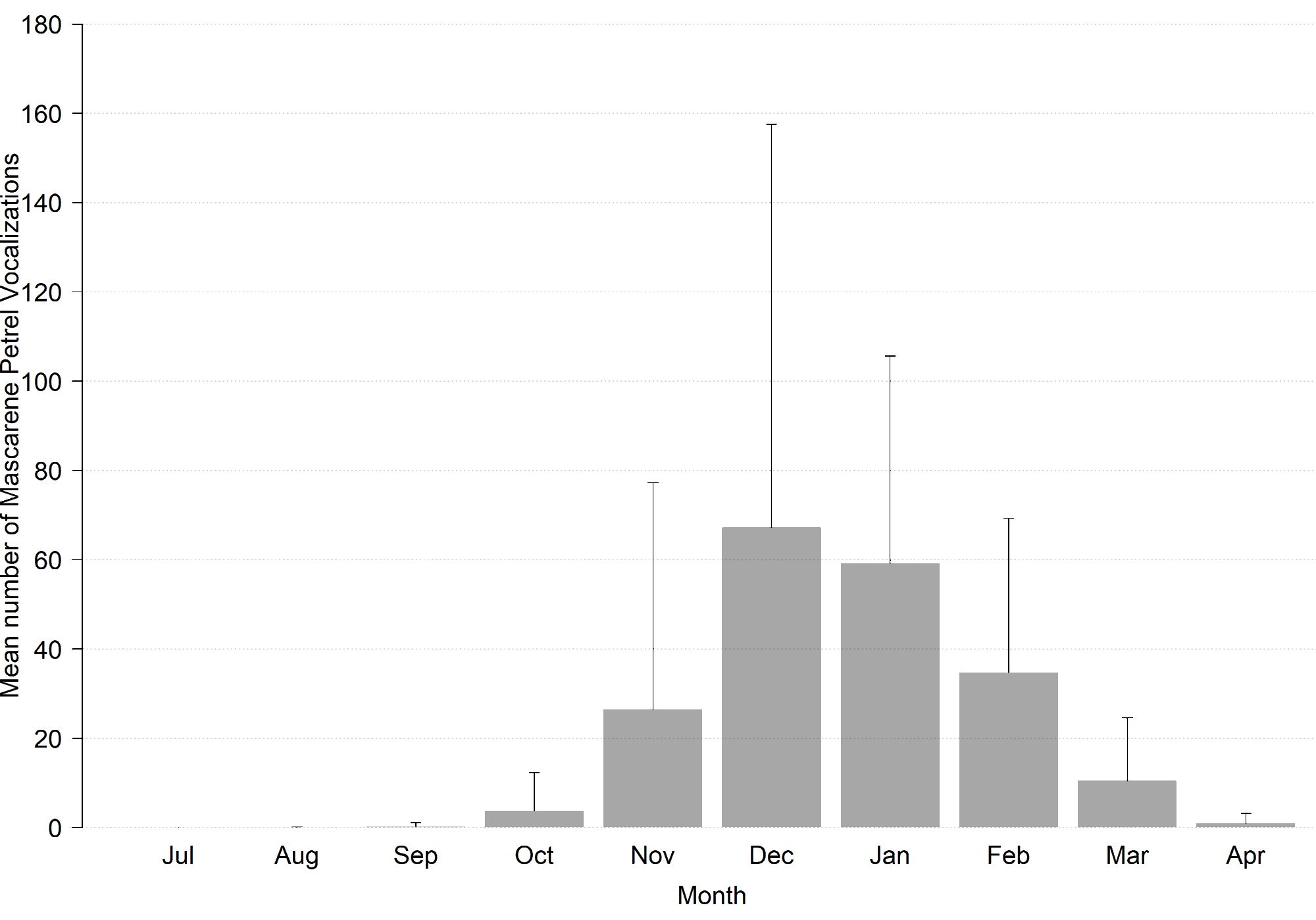
Fig. 3. Monthly variations in the mean number of calls per hour of Mascarene Petrel (Pseudobulweria aterrima) at Malabar (manual analysis) during the peak of daily vocal activity (eight to nine hours after sunset), from July 2015 to April 2017, Réunion Island.
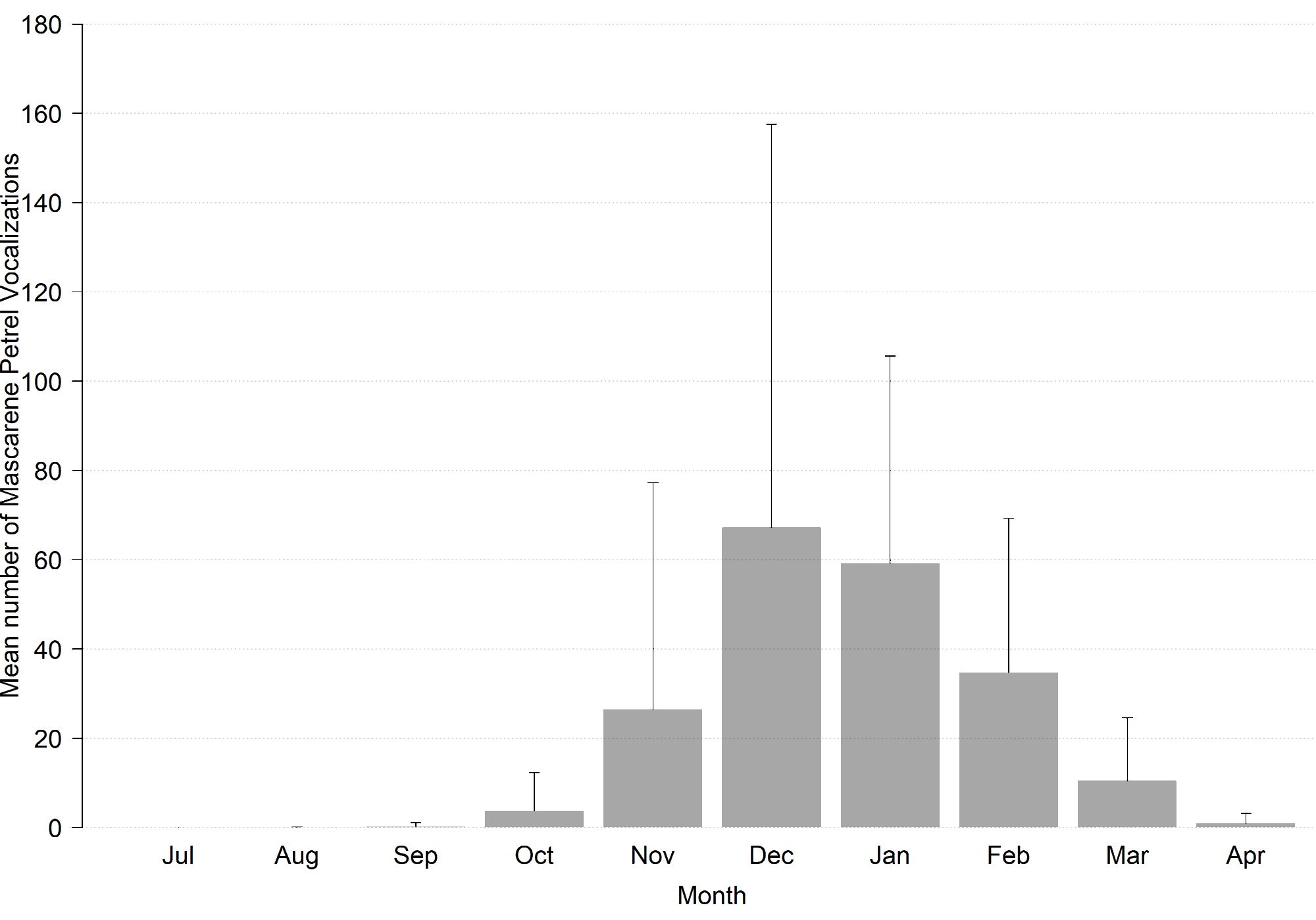
Fig. 4
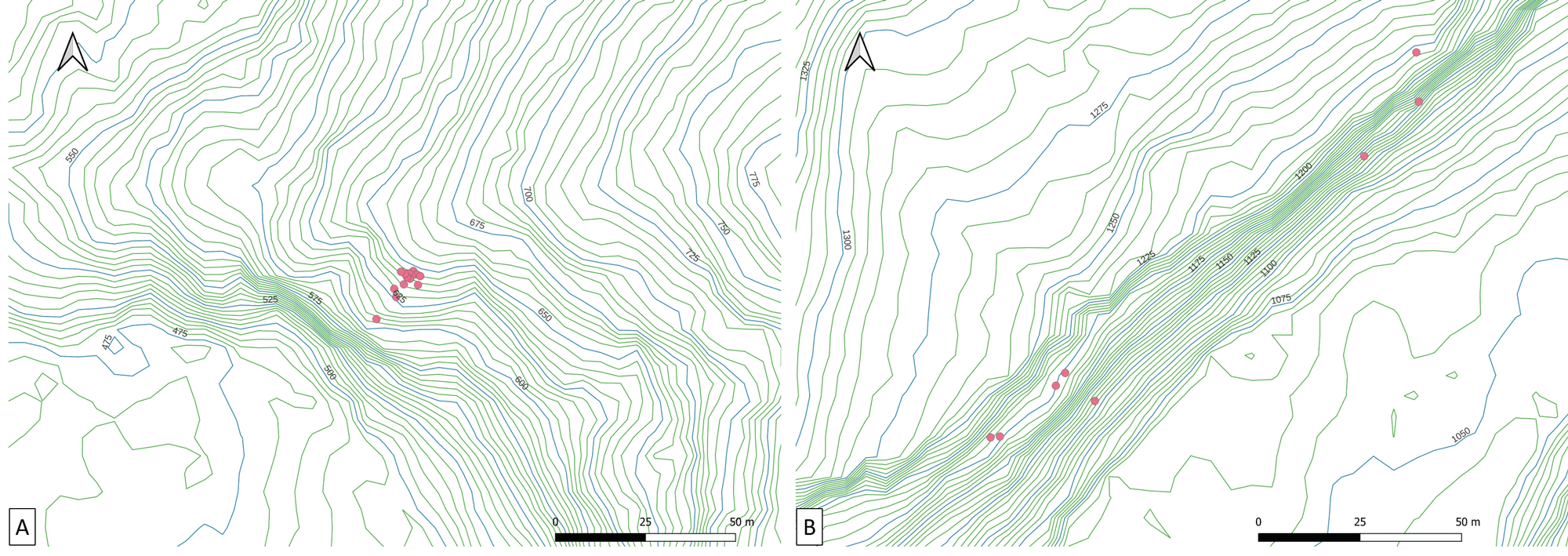
Fig. 4. Topographical details of Mascarene Petrel (Pseudobulweria aterrima) breeding colonies located at Rein de Dimitile (A) and Rond des Chevrons (B). The red dots correspond to the location of the burrows.

Table 1
Table 1. Characteristics of the automated acoustic survey conducted at Réunion Island for the detection of Mascarene Petrel (Pseudobulweria aterrima) breeding sites.
| Breeding season | 2015–2016 | 2016–2017 | Total |
| Sampling period | 1 Jul 2015–30 Apr 2016 | 1 Jul 2016–28 Apr 2017 | - |
| Total recording duration (h) | 9213.5 | 5690.0 | 14,903.5 |
| Analyzed recordings (h) | 5516.5 (59.87%) | 5690.0 (100%) | 11,206.5 |
| Number of surveyed sites | 18 | 50 (+ 3 same as 2015–2016) | 68 |
| Number of sites where Mascarene Petrels were detected | 7 | 10 (+ 3 same as 2015–2016) | 17 |
Table 2
Table 2. Characteristics of the infrared thermal binocular survey conducted at Réunion Island for the detection of Mascarene Petrel (Pseudobulweria aterrima) breeding sites.
| Breeding season | 2015–2016 | 2016–2017 | Total |
| Number of infrared thermal binocular sessions | 18 | 32 | 50 |
| Observations effort (h) | 102 | 103 | 205 |
| Number of surveyed sites | 6 | 14 | 16 (4 sites were surveyed during both seasons) |
| Sites with Mascarene Petrels | 4 | 9 | 11 (2 sites were surveyed during both seasons) |
| Sites with landing Mascarene Petrels | 0 | 5 | 5 |
Table 3
Table 3. Characteristics of the two Mascarene Petrel (Pseudobulweria aterrima) colonies discovered in 2016–2017, Réunion Island.
| Colony | Rein de Dimitile (RD) | Rond des Chevrons (RDC) |
| Elevation (m) | 640 | 1200 |
| Elevation from the riverbed (m) | 100–150 | 100–150 |
| Slope of the cliff | 50° to 90° | 50° to 90° |
| Vegetation height | Maximum 7 m | Maximum 7 m |
| Vegetation type | Hygrophile forest species of medium and high altitude, dominated by Olea lancea and Monimia rotundifolia | |
| Habitat structure | Continuous forest ridge | Patches of vegetation separated by rocky surfaces |
| Colony orientation | West | Southeast |
| Numbers of burrows found at the time of discovery | 14 | 8 |
| Mean width of burrow entrance (cm ± sd) |
17.7 ± 4.5 (n = 12) |
15.5 ± 1.9 (n = 4) |
| Mean height of burrow entrance (cm ± sd) |
12.8 ± 3.4 (n = 12) |
14.3 ± 4.2 (n = 4) |
| Mean depth of burrows (cm ± sd) |
100.0 ± 38.1 (n = 11) |
— |
| Distribution of the burrows | One single patch | 4 small patches containing 1 to 4 burrows each |
| Substrate where burrows are dug | Humus under rocks or roots | Humus under rocks or roots |









