The following is the established format for referencing this article:
Gunn, C. 2022. A surveillance camera reveals season-long nesting activities and behaviors at a nest of the Northern Black Swift (Cypseloides niger borealis). Journal of Field Ornithology 93(4):10.ABSTRACT
This study employed a high-resolution camera to capture a visual record of an active Northern Black Swift (Cypseloides niger borealis) nest throughout an entire breeding season to document nesting activities and behaviors which are incompletely known in this species. The camera captured footage 24 hours a day from arrival of the adults at Box Canyon Falls, Ouray, Colorado, to fledging. Nest repairs began a week after arrival and were conducted 83% of the time by the female. Egg-laying occurred three weeks after arrival. The adults nearly equally shared incubation, brooding, and chick provisioning duties; the male performed 49% of effective incubation, 47% of brooding, and 51% of provisioning while the female contributed 51%, 53%, and 49%, respectively. Adults fed the nestling intermittently both day and night until age 15 days, at which time brooding ended, the nestling spent days alone, and provisioning occurred primarily at night. The nestling started wing exercises at age 13 days and they continued in increasing frequency and duration until the day prior to fledging at 51 days of age. The nesting pair exhibited cathemeral behavior, active night and day to conduct activities necessary for survival and to successfully raise their offspring. Although only one nest was observed, the surveillance camera was an effective tool for recording otherwise difficult-to-observe activities, especially those occurring at night, and revealed many previously unknown breeding activities and behaviors for this species.RESUMEN
INTRODUCTION
Observations of nesting Northern Black Swifts (Cypseloides niger borealis, hereafter Black Swift) are typically sporadic and incomplete due to inaccessibility of colonies, small colony size, the cryptic nature of nest placement, and adults’ long absences from the nest. Continuous study is logistically difficult, resulting in deficiencies in the most basic information about this species’ life history, including breeding (Gunn et al. 2021). This study employed a surveillance camera to document season-long nesting activities and behaviors at an active Black Swift nest to discover and more fully understand nesting events in this species.
The Northern Black Swift is a neotropical swift found at breeding colonies scattered sparsely across western North America. Colonies occur exclusively near waterfalls and in a few caves where cool, moist conditions provide favorable nesting habitat (Knorr 1961, Foerster 1987, Marín and Stiles 1992, Knorr 1993, Marín 1997b, Levad et al. 2008, Gunn et al. 2012). This long-lived species exhibits strong colony and nest site fidelity (Foerster 1987, Collins and Foerster 1995, Marín 1997b, Hirshman et al. 2007, Gunn et al. 2020). They produce a single clutch consisting of one egg annually and the breeding season is protracted, on average lasting 89 days from laying to hatching (Gunn et al. 2020). Diet consists of aerial insects and ballooning spiders (Marín 1999a, Potter et al. 2015). The species exhibits sexually dimorphic plumage characteristics (Gunn et al. 2018). Migration paths and wintering areas in western Brazil for 12 swifts from Colorado are described (Beason et al. 2012, Hedenström et al. 2022; C. Gunn unpublished data).
These demanding field studies provide basic information on some of the life history characteristics of Black Swifts, but none provide detailed information on nesting behaviors and activities. The purpose of this study was to reveal previously unknown aspects of adult time budgets, copulation, nest-building, laying, incubation, brooding, nestling provisioning and development, preening, fledging, and any previously undocumented activities.
METHODS
Study area
This study was conducted at Box Canyon Falls Park, Ouray, Colorado (38°01′4″ N, 107°40′44″ W, elevation 2380 m), which has supported a colony of 8–20 Black Swift nesting pairs annually since its discovery in 1950 (Knorr 1961, Gunn et al. 2020). The 30 m falls plunge into a deeply incised, overhanging cleft in fault-weakened limestone (Chronic and Williams 2002). This site is unique in that it affords an excellent view of nests, a nearby power source, Internet, a secure location for camera installation, and indoor space for associated equipment.
Field methods
An ALIBI ALI-NS7004R-B four-megapixel camera with pan-tilt-zoom and 150 m distance infrared (IR) lighting (ALIBI, Austin, Texas, USA) was bolted to rock 5 m above a metal walkway and aimed into the cup of a historic nest ~15 m away (Appendix 1). This nest was active 22 out of the past 25 years indicating it had a good chance of again successfully raising offspring (S. Hirshman, personal communication). The camera’s IR illuminators emitted light at a wavelength of 850 nm, above the 700 nm visible to birds (Bowmaker et al. 1997). Category 6 ultraviolet-resistant gel-filled Ethernet cable (Micro Connectors Inc., Hayward, California, USA) ran from the camera to an Ethernet connection at the Box Canyon Falls Park Nature Center. To prevent rodent damage, the cable was buried 0.2 m underground where possible; in some rocky areas where the cable ran aboveground, it was covered with Gardner Bender 12.7 mm Split Flex Tubing (Gardner Bender, Milwaukee, Wisconsin, USA). Because the cable was 140 m long, a Cudy 1 gigabit power-over-Ethernet extender (Shenzhen Cudy Technology Co., Ltd., Shenzhen, China) in a Cantex 20 x 20 x 10 cm weatherproof junction box (Cantex Inc., Ft. Worth, Texas, USA) installed at the mid-point of the cable boosted the signal. The cable connected to an ALIBI ALI-NVR3308P network video recorder (NVR) with an eight-terabyte hard drive (Western Digital Corporation, San Jose, California, USA), to an APC 650VA battery backup and surge protector (Schneider Electric, USA Inc., Boston, Massachusetts, USA), and to a Ubiquiti Networks UniFi eight-port switch (Ubiquiti Inc., San Jose, California, USA). A high-definition multimedia interface (HDMI) cable connected the NVR to an ASUS full HD backlit LED 60 cm monitor (ASUSTek Computer Inc., Taipei, Taiwan) so visitors to the Nature Center could view real-time nest activities (Appendix 2). The system was in place and recording 11 May, before anticipated arrival of Black Swifts at Box Canyon based on previous breeding phenology studies at that colony (Hirshman et al. 2007, Gunn et al 2020). Recording ended 15 September 2021, five days after fledging, to ensure we captured last attendance at the nest. No sound recordings were made as the distance from the nest to the camera and noise from the waterfall prevented capturing vocalizations.
Data analysis
The author viewed all 2520 hours of recorded data; no footage was eliminated from analysis. Slow-motion was employed to scrutinize certain activities in greater detail. The author recorded details of phenology, adult daily time budget, possible copulation attempts, nest-building, laying, incubation, brooding, nestling provisioning and development, adult preening, and miscellaneous activities.
RESULTS AND DISCUSSION
The network video recorder captured 5.9 terabytes of data from arrival of the swifts on 29 May 2021, through fledging of the nestling and abandonment of the nest by adults on 10 September, a total of 105 days. The forked tail of the male and square tail of the female enabled sex-specific activities to be determined (Fig. 1; Gunn et al. 2018).
Phenology
The swifts arrived at the nest 29 May within two hours of each other, the female arriving first at 03:45. Laying occurred on 24 June and hatching on 21 July. The male left the nest the afternoon of 8 September and did not return; the female left the nest 15 minutes after the nestling fledged on 10 September. These dates correlate well with those previously reported for this subspecies in Colorado (Hirshman 1998, Hirshman et al. 2007, Gunn et al. 2020) and at other locations (Foerster 1987, Campbell et al. 1990, Bailey 1993, Marín 1997b, 1999a).
Adult daily time budget
The daily time budget spent at the nest for each adult from arrival through fledging varied depending on phenology events, but was nearly equal for the male and female (Fig. 2). Prior to laying and after brooding, adults showed diurnal and crepuscular habits, leaving the nest to presumably forage before dawn and returning after dusk (Sparks 2018, Gunn et al. 2021; R. A. Sparks, Lois Webster Fund Presentation, Denver Field Ornithologists, https://www.youtube.com/watch?v=inzuiv0BlJ4). Nest-building, provisioning the nestling, and preening were primarily nocturnal activities and adults only slept a maximum of about three uninterrupted hours each night.
The Black Swift has traditionally been considered a diurnal species, but this study revealed characteristics of a cathemeral species, with activity patterns which are not exclusively nocturnal, diurnal, nor crepuscular, but irregularly active at any time of night or day (Tattersall 1987). Species adopt a cathemeral lifestyle for purposes of food acquisition, reproduction, socialization, self-care, predator avoidance, and other livelihood activities. A number of cathemeral mammal species are known, but there are few cathemeral bird species, the Burrowing Owl (Athene cunicularia) being one example (Poulin et al. 2020). Further illustration of cathemerality in the Black Swift includes non-stop migratory flights (Beason et al. 2012) and continuous flight without landing to roost while in the wintering areas in Brazil (Hedenström et al. 2022).
Copulation
The male swift may have attempted copulation at the nest (Appendix 3, Video S1), but footage from six interactions reviewed at one-quarter speed revealed that no copulation took place. These interactions occurred from arrival on 29 May to 7 July, 13 days after the egg was laid, lasted from 6 to 28 seconds, and occurred between 20:50 and 05:30, in darkness. In three cases, the swifts flew from the nest simultaneously after the interaction and it is possible that copulation occurred when the swifts flew beyond the frame of the camera. In the other three cases, the adults remained at the nest after the encounter with no further interaction. The encounters were unlike any other behaviors observed, the male being very aggressive and the female attempting to avoid the exchange. Timing and manner of copulation in the Black Swift has not been studied (Gunn et al. 2021). Foerster (1987) interpreted an aerial exchange as copulation in one instance and Campbell et al. (1990) cites a pair seen copulating in the air in British Columbia. Other observers interpreted interactions in the air as equivocal in proving that copulation had taken place (Rathbun 1925, Marín and Stiles 1992, Marín 1997a).
Nest-building
The previous year’s nest was present but dilapidated when the swifts arrived, so repairs were made to bolster the existing structure. Repairs began on 5 June, seven days after adults arrived at the colony and continued until 6 August when the nestling was 16 days old, a total of 62 days. No nest-building activities occurred by either adult for four days prior to and one day after laying. All nest substrate collection and incorporation occurred in darkness. Nest-building activities varied between the male and female, with the female performing the majority of nest substrate collection and incorporation (Table 1). All nest repair items were vegetation, consisting of moss, fern tips (Polypodium sp.), fine grasses, a Gambel oak leaf (Quercus gambelii), a few spruce needles (Picea sp.), and a few unidentified pieces of vegetation. The swift delivering nesting substrate immediately incorporated it into the nest. Substrate was carried in the beak, never in the feet. The adults incorporated substrate by first placing it at the desired location, and then by using the beak to drive it into the existing nest structure by rapidly moving the head back and forth (Appendix 3, Video S2). Adults usually dropped dry grass stems, spruce needles, and fern tips into the nest without incorporating them. The swifts sat in the cup of the nest for all building activities, even if they had to push the other adult out of the way.
The swifts often returned with droplets of water on the head and body feathers, indicating they had gathered the materials near spray from the waterfall. The short collection times and types of substrate collected indicate materials were obtained in the immediate vicinity of the nest. All gleaning and deliveries occurred between 20:17 and 05:47 (N = 270; Fig. 3) which explains the paucity of previous observations of this activity. eBird shows a three-second video of a Black Swift collecting nesting material at Box Canyon (Macauley Library ML 476959). In southern California, a researcher observed a swift gathering moss with its bill while clinging to a wall with wings extended, but did not observe nest-building (Marín 1997b).
Laying and incubation
Both adults shared the nest cup for roosting and sleeping at night prior to laying. As laying approached, the female spent more time in the nest in the mornings rather than leaving at dawn to forage. In the hour prior to laying, the female appeared agitated, could not settle in one position on the nest, and defecated very liquid feces frequently without moving to the side of the nest to eject feces over the edge as usual. Normal resting respirations of 60–70/min increased to ~80/min as observed by tail dipping (Arnall and Petrak 1982). She laid the egg at 11:30 on 24 June (Appendix 3, Video S3) and remained on the egg until mid-afternoon.
Adults left the egg uncovered for three to 11 hours per day until consistent incubation began on 1 July. Without variation, the male and female alternated daytime incubation (from dawn to dusk) from 1 July to 17 July (Fig. 2). At night, both adults intermittently covered the egg without a set pattern, exchanging incubation duties to collect nesting material, preen, or sleep. The adults took turns covering the egg for several hours both day and night from 18 July until hatching on the evening of 21 July. Total incubation, effective incubation, mean daily egg neglect, and longest incubation bouts were nearly equally shared between the male and female (Table 1). Daily brooding and incubation on/off bouts for both adults are referenced (Appendix 4). The adults rolled the egg by backing up and using the beak to manipulate the egg which was kept between the legs. Frequency of this activity was difficult to track if the view was obscured, but it occurred hourly to several hours apart, often triggered when the incubating adult changed positions on the nest, defecated, or preened. Eggshells remained in the nest until they disintegrated; the swifts did not ingest or eject them from the nest.
Brooding
The male and female alternated brooding duties daily from 22 July until 27 July. After that, one of the adults was almost constantly brooding the nestling while the other was foraging, but they switched duties one or more times during the day and night (Fig. 2, Table 1, Appendix 4). The nestling was brooded between the legs of the adults, usually with its posterior facing the head of the adult. On two occasions, the brooding adult ate a fecal sac produced by the chick (Appendix 3, Video S4). By day 10, the nestling was too large to fit between legs of adults and was brooded between the body and one wing of the adult. On 5 August when the nestling was 15 days old and covered in dense semiplumes, it spent its first 10-hour day alone. After that date, the nestling remained alone during the daytime; adults arrived for short daytime provisioning bouts which became less frequent and ended as the nestling grew.
In California, Foerster (1987) found that adults brooded the nestling continuously for the first 10 days post-hatching; brooding then decreased as the nestling developed a thick semiplume covering and was able to thermoregulate. Marín (1997b) contends that in Cypseloidine swifts, brooding seems to be a critical factor in the first 13–14 days of life, or until acquisition of thermoregulatory ability. Collins (1968) found that the body temperature of the Chestnut-collared Swift (Streptoprocne rutila), also a Cypseloidine, approached thermoregulatory ability by age 12–13 days, coinciding with the full growth of the down-like semiplumes.
Provisioning the nestling
A “feeding episode” is defined here as a discrete bout of feeding the nestling, occurring when the adults regurgitate and feed one or more individual “portions” of the bolus of insects carried in the upper esophagus. Adults attempted feeding episodes soon after arriving at the nest after foraging. The adults moved their heads in a forward thrusting motion as they regurgitated part of the bolus and the floor of the mouth distended. The adult inserted its beak into the open beak of the nestling, and with a rapid pumping motion, delivered the insects to the nestling (Appendix 3, Videos S5 and S6). The nestling begged by pecking at the breast, throat, beak, and face of the adult, sometimes quite aggressively. When the bolus was depleted, the adult turned its head or moved away from the nestling to avoid the pecking/begging behavior. The inside of the nestling’s mouth was pink, with no pigmented marks or patterns.
Adults first fed the hatchling 25 h 30 min after hatching, the yolk sac remnants nourishing the hatchling during that time. Feeding episodes were frequent and the number of individual portions were low for the first eight days after hatching. Feeding episodes gradually decreased in number after day nine, but the number of individual portions per feeding episode increased until day 34 when both feeding episodes and individual portions began to decrease prior to fledging on day 51 (Fig. 4). The begging nestling performed a wing-raising display twice, lifting both wings over its back when the female arrived at the edge of the nest after foraging.
The adults fed the nestling almost equally (Table 1). The male returned to the nest during the day to feed the nestling more than the female. The male primarily fed the nestling between 22:00 and 03:00 and the female primarily fed the nestling between 18:00 and 01:00, demonstrating a predominantly nocturnal provisioning strategy (Fig. 5). In the three days prior to fledging, feeding episodes decreased.
Adults attempted to feed the hatchling insects too large to swallow on several occasions. In these instances, either the adult removed the insect from the hatchling’s mouth and re-ingested it, or the hatchling shook its head until it ejected the incompletely swallowed insect. The male regurgitated and spit out three large noctuid moths rather than feed them to the hatchling. Pellets of undigested insect parts were never regurgitated by either the adults or nestling.
The long delay between hatching and the first meal given to the hatchling in this study is a well-recognized trait in hatchlings as they assimilate remnants of the yolk for nourishment (Romanoff 1944). A few short studies reported nocturnal provisioning of Black Swift nestlings (Collins 1998, Collins and Peterson 1998, Marín 1999a, Gunn 2010). There are also daytime observations of Black Swift nestling provisioning (Michael 1927, Grant 1966; S. Hirshman, personal observation). Marín (1997a) documented nestling wing-raising when an adult arrived at the nest to feed the nestling. It was notable that the nestling received very few feedings in the last three days prior to fledging which likely affected body mass. Foerster (1987) reported that nestlings lost a few grams of weight before fledging.
This study found that the male and female almost equally shared incubation, brooding, and chick provisioning duties. Knorr and Baily (1950) believed that males spent most of the day foraging far from the nesting area, returning at infrequent intervals while females incubated. Michael (1927) stated that he believed the female performed all the duties of incubation and feeding of the young.
Nestling development and fledging
The nest was inaccessible and the nestling was not handled, but footage revealed much about daily development and behavior of the nestling (Table 2); the table can serve as a visual guide for aging Black Swift nestlings. When the nestling was alone in the nest, it picked at the nest cup as if gleaning dropped insects, preened, exercised its wings, defecated, yawned, and moved around within the nest cup. Observation did not detect a time period in which the chick was inactive long enough to enter/exit torpor, although only readings of core body temperature could prove this. Previous reports of nestling growth and plumage development agree, in general, with the findings in this study (Foerster 1987, Marín 1997b, 1999b), taking into account that nestling growth varies, even among nests at a single colony (Gunn et al. 2020).
The nestling first exercised its wings on 3 August, at 13 days of age. As it grew older, the exercises increased in frequency and duration and lasted until the day before fledging. (Fig. 6; Appendix 3, Videos S7 and S8). During each wing exercising bout, the nestling intermittently rested the wings by leaving them fully outstretched before flapping them again. The nestling only exercised wings when it was alone at the nest. From age 13 days to age 38 days, the nestling always remained in the nest cup and faced the back corner of rock to exercise wings, but by day 39 it moved onto the rim of the nest to exercise wings. By age 43 days, the nestling started to move to the side of the nest to exercise wings and by days 49–50 the nestling moved to rocks below the base of the nest to exercise wings. Playback at one-quarter speed of 10 separate wing exercise bouts indicated 8–12 wingbeats/second, and this rate stayed the same despite age of the chick. The nestling did not exercise its wings prior to fledging at 05:15 on day 51 (Appendix 3, Video S9). Previous reports stated that nestling wing exercises occurred just prior to fledging to build up the flight muscles, but no details were given (Smith 1928, Hunter and Baldwin 1962, Grant 1966). In this study, the nestling fledged before dawn and did not return to the nest, which agrees with a previous study (Marín 1999b).
Adult preening
Preening times included onset of the preening bout, scratching, stretching, and rousing. Preening was mainly confined to the contour feathers of the breast, scapulars, and back; rarely did the adults preen the remiges or rectrices or appear to obtain oil from the preen gland (Appendix 3, Video S10). Adults scratched their heads directly by bringing a foot up between the body and wing to reach the head. No contour feathers were lost and no flight feathers were molted during the breeding season; when the swifts stretched and revealed the wings and tail, there were no missing or partially grown feathers. Adults did not allopreen and neither adult preened the nestling. Both adults preened at a fairly constant daily rate throughout the season and preening occurred mainly in darkness (Fig. 7, Table 1). It is possible that the adults preened when away from nest such as when roosting on rocks out of view of the camera or possibly during flight. Common Swifts (Apus apus) are regularly observed lifting wings vertically in flight and losing height, while turning the head backwards and gripping a flight feather with the beak and this has been interpreted as aerial preening (A. Hedenström, personal communication). Considering the Common Swift may stay airborne for up to ten months (Hedenström et al. 2016) and the Black Swift for eight months (Hedenström et al. 2022), it seems likely they have to preen feathers in flight. The nestling started preening at 10 days of age. Nestling preening was not recorded because adults often obscured the view.
No information on preening in this species has been reported (Gunn et al. 2021). Data collated on 62 different bird species across seven orders showed that on average, birds spend 9.2% of the day in maintenance activities including preening, scratching, dusting, sunning, anting, bill wiping, water bathing, smoke bathing, and comfort movements such as stretching (Cotgreave and Clayton 1994). These same authors found the major component of these maintenance activities (92.6%) was preening and scratching; male birds devoted more time to maintenance than females, except in the case of ducks. These figures are much larger than the daily average of 1.1–1.4% of time spent preening in this study, although the male swift spent more time grooming than the female.
Miscellaneous behaviors
A previously undocumented behavior, described here as a “beak tussle,” was exhibited by the adults toward each other. It involved mild to intense pecking by one adult on the beak, face, and head of the other when one adult arrived at the nest or if one adult disturbed the other. The response was to peck back, or to turn and lower the head to deflect or avoid pecks (Appendix 3, Video S11). Beak tussles at times were quite violent, did not seem ritualized, and appeared to be agonistic behavior between the adults. This behavior was never displayed toward the nestling.
The swifts performed wing-raising displays, in which both wings were held almost touching above the body, as described in Marín (1997a). The female occasionally performed a subtle wing-raise toward the male when he arrived at the nest or engaged her in a beak tussle. On two occasions, the nestling performed a wing-raising display when an adult arrived at the nest to provision it.
It was not unusual for other swifts, both male and female, to fly very close to the adults on the nest, to perch on the rocks near the nest, to approach the nest closely enough to tug on feathers of the adults, or to attempt to get into the nest. In some cases, neither adult took notice of these intrusions, continuing to sleep, preen, or pick at the nest. At other times, the female, in particular, was aware of the visiting swift, stared at it intently, moved toward it or performed a wing-raising display. If the intruder did not fly off, the adult would actively chase it off, then immediately return to the nest. Most of these encounters were during darkness and especially near dawn when many swifts were flying about the colony.
Neither adult fed the other during this study. From 1996 to 2017, one observer logged approximately 13,500 observation hours at Box Canyon and never observed one adult feeding the other (S. Hirshman, personal communication). Michael (1927) reported what appeared to be one adult feeding another at a nest. Murphy (1951) reported that occasionally both adults would be at a nest at the same time, one giving food to the other. Several times during incubation and the early part of brooding, Marín (1997a) observed one adult giving food to another.
Occasionally, the adults left the nest during darkness for variable amounts of time and returned with water droplets on their head and feathers without bringing back nesting material, but indicating they had been near spray from the falls. In one instance, dried urates present on the tail of the female disappeared after one of these short absences which suggests she could possibly have been bathing. Several times, the female appeared to drink from a rain-filled crack in the rocks where she often roosted.
CONCLUSIONS
Although this study examined a single nest, it revealed many previously unknown aspects of the breeding activities and behaviors for this species, forming a baseline of observations. The camera system used here performed well and was effective obtaining documentation of activities and behaviors that would otherwise be difficult or impossible to obtain. Researchers could employ similar techniques or adapt the system to solar power and wireless connectivity to document other species which are difficult to observe with usual field methods.
RESPONSES TO THIS ARTICLE
Responses to this article are invited. If accepted for publication, your response will be hyperlinked to the article. To submit a response, follow this link. To read responses already accepted, follow this link.ACKNOWLEDGMENTS
The Colorado Field Ornithologists' Joe Roller Memorial Grant (#CFO202102) and private donations supported this citizen science project. No funders had input into the content of the manuscript, and no funder approval was required before submission or publication. The City of Ouray, Colorado, and R. Noll fully supported the project by allowing installation of the camera and associated equipment and by providing wireless Internet service. R. Willis, City of Ouray I.T. Specialist, helped technical aspects of the project. The Southwest Conservation Youth Corp volunteers and E. Hendrickson helped install cable conduits. J. Fleetwood made installation and operation of the camera and equipment possible. We thank the Macaulay Library at the Cornell Lab of Ornithology for the video citing (ML 476959). I am indebted to Dr. J. I. Areta and an anonymous reviewer for their careful review and invaluable comments which greatly improved an earlier version of the manuscript.
DATA AVAILABILITY
Data/code sharing is not applicable to this article because no data/code were analyzed in this study.
LITERATURE CITED
Arnall, L., and M. L. Petrak. 1982. Diseases of the respiratory system. Page 400 in M. L. Petrak, editor. Diseases of cage and aviary birds. Second edition. Lea and Febiger, Philadelphia, Pennsylvania, USA.
Bailey, S. F. 1993. Black Swift. Pages 176-177 in D. Roberson and C. Tenney, editors. Atlas of the breeding birds of Monterey County California. Monterey Peninsula Audubon Society, Monterey, California, USA.
Beason, J. P., C. Gunn, K. M. Potter, R. A. Sparks, and J. W. Fox. 2012. The Northern Black Swift: migration path and wintering area revealed. Wilson Journal of Ornithology 124(1):1-8. https://doi.org/10.1676/11-146.1
Bowmaker, J. K., L. A. Heath, S. E. Wilkie, and D. M. Hunt. 1997. Visual pigments and oil droplets from six classes of photoreceptor in the retinas of birds. Vision Research 37(16):2183-2194. https://doi.org/10.1016/S0042-6989(97)00026-6
Campbell, R. W., N. K. Dawe, I. McTaggert-Cowan, J. M. Cooper, G. W. Kaiser, and M. C. E. McNall. 1990. The birds of British Columbia, Volume 2: nonpasserines (diurnal birds of prey through woodpeckers). Royal British Columbia Museum, Victoria, Canada.
Chronic, H., and F. Williams. 2002. Roadside geology of Colorado. Second edition. Mountain Press Publishing Company, Missoula, Montana, USA.
Collins, C. T. 1968. Distributional notes on some Neotropical swifts. Bulletin of the British Ornithologists’ Club 88:133-134.
Collins, C. T. 1998. Food delivery and chick provisioning in cypseloidine swifts. Bulletin of the British Ornithologists’ Club 118:108-112.
Collins, C. T., and K. S. Foerster. 1995. Nest site fidelity and adult longevity in the Black Swift (Cypseloides niger). North American Bird Bander 20:11-14. https://www.researchgate.net/publication/237632937
Collins, C. T., and B. M. Peterson. 1998. Nocturnal chick provisioning by Black Swifts. Western Birds 29(3):227-228. https://sora.unm.edu/node/122667
Cotgreave, D. H., and P. Clayton. 1994. Comparative analysis of time spent grooming by birds in relation to parasite load. Behaviour 131(3-4):171-187. https://doi.org/10.1163/156853994X00424
Foerster, K. S. 1987. The distribution and breeding biology of the Black Swift (Cypseloides niger) in Southern California. Thesis. California State University, Long Beach, California, USA.
Grant, J. 1966. A Black Swift nest in British Columbia. Canadian Field-Naturalist 80:60-61. https://www.biodiversitylibrary.org/item/89101#page/70/mode/1up
Gunn, C. 2010. Use of a game surveillance camera to capture nesting activity of Black Swifts. Colorado Birds 44:172-179.
Gunn, C., K. J. Aagaard, K. M. Potter, and J. P. Beason. 2018. Sexually dimorphic plumage characteristics in the Northern Black Swift. Western Birds 49:214-225. https://doi.org/10.21199/WB49.3.4
Gunn, C., S. E. Hirshman, and K. J. Aagaard. 2020. Trends in Black Swift (Cypseloides niger) breeding phenology, productivity, and nest success in southwest Colorado, 1996-2017. Wilson Journal of Ornithology 132:317-326. https://doi.org/10.1676/1559-4491-132.2.317
Gunn, C., P. E. Lowther, C. T. Collins, J. P. Beason, K. Potter, and M. Webb. 2021. Black Swift (Cypseloides niger), version 2.0. In S. M. Billerman and B. K. Keeney, editors. Birds of the World. Cornell Lab of Ornithology, Ithaca, New York, USA. https://birdsoftheworld.org/bow/species/blkswi/cur/introduction
Gunn, C, K. M. Potter, and J. P. Beason. 2012. Nest microclimate at Northern Black Swift colonies in Colorado, New Mexico, and California: temperature and relative humidity. Wilson Journal of Ornithology 124(4):797-802. https://doi.org/10.1676/1559-4491-124.4.797
Hedenström, A., G. Norevik, K. Warfvinge, A. Andersson, J. Backman, and S. Åkesson. 2016. Annual 10-month aerial life phase in the Common Swift Apus apus. Current Biology 26(22):3066-3070. https://doi.org/10.1016/j.cub.2016.09.014
Hedenström, A., R. A. Sparks, G, Norevik, C. Woolley, G. J. Levandoski, S. Åkesson. 2022. Moonlight drives nocturnal vertical flight dynamics in black swifts. Current Biology 32(8):1875-1881. https://doi.org/10.1016/j.cub.2022.03.006
Hirshman, S. 1998. Black Swifts (Cypseloides niger) in Box Canyon, Ouray, Colorado. Journal of Colorado Field Ornithology 32:53-60.
Hirshman, S. E., C. Gunn, and R. G. Levad. 2007. Breeding phenology and success of Black Swifts in Box Canyon, Ouray, Colorado. Wilson Journal of Ornithology 119(4):678-685. https://doi.org/10.1676/06-112.1
Hunter, W. F., and P. H. Baldwin. 1962. Nesting of the Black Swift in Montana. Wilson Bulletin 74:409-416. https://www.jstor.org/stable/4159102
Knorr, O. A. 1961. The geographical and ecological distribution of the Black Swift in Colorado. Wilson Bulletin 73(2):155-170. https://www.jstor.org/stable/4158914
Knorr, O. A. 1993. Black Swift (Cypseloides niger) nesting site characteristics: some new insights. Avocetta 17(2):139-140. https://www.avocetta.org/articles/vol-17-2-bva-black-swiftnbsp-icypseloides-nigerinbsp-nesting-site-characteristicssome-new-insights/
Knorr, O. A., and A. L. Baily. 1950. First breeding record of Black Swift (Nephoecetes n. borealis) in Colorado. Auk 67:516.
Levad, R., C. Shultz, K. Potter, C. Gunn, and J. Doerr. 2008. Distribution, abundance and nest-site requirements of Black Swift (Cypseloides niger borealis) in the Southern Rocky Mountains of Colorado and New Mexico. Wilson Journal of Ornithology 120(2):331-338. https://doi.org/10.1676/07-049.1
Marín, M. 1997a. On the behavior of the Black Swift. Condor 99(2):514-519. https://doi.org/10.2307/1369958
Marín, M. 1997b. Some aspects of the breeding biology of the Black Swift. Wilson Bulletin 109:290-306. https://www.jstor.org/stable/4163812
Marín, M. 1999a. Food, foraging, and timing of breeding of the Black Swift in California. Wilson Bulletin 111(1):30-37. https://www.jstor.org/stable/4164025
Marín, M. 1999b. Growth rates in the Black Swift: temperate versus tropical comparisons. Ornitologia Neotropical 10(2):179-192. https://sora.unm.edu/node/119337
Marín, M., and F. G. Stiles. 1992. On the biology of five species of swifts (Apodidae, Cypseloidinae) in Costa Rica. Proceedings of the Western Foundation of Vertebrate Zoology 4(5):387-351. https://www.researchgate.net/publication/44359639
Michael, C. W. 1927. Black Swifts nesting in Yosemite National Park. Condor 29(2):89-97. https://doi.org/10.2307/1363018
Murphy Jr., J. A. 1951. The nesting of the Black Swift. Natural History 60:446-449.
Potter, K. M., C. Gunn, and J. P. Beason. 2015. Prey items of the Black Swift (Cypseloides niger) in Colorado and a review of historical data. Wilson Journal of Ornithology 127(3):411-420. https://doi.org/10.1676/14-152.1
Poulin, R. G., L. D. Todd, E. A. Haug, B. A. Millsap, and M. S. Martell. 2020. Burrowing Owl (Athene cunicularia), version 1.0. In A. F. Poole, editor. Birds of the World. Cornell Lab of Ornithology, Ithaca, New York, USA. https://birdsoftheworld.org/bow/species/burowl/cur/introduction
Rathbun, S. F. 1925. The Black Swift and its habits. Auk 42(4):497-516. https://doi.org/10.2307/4074986
Romanoff, A. L. 1944. Avian spare yolk and its assimilation. Auk 61(2):235-241. https://doi.org/10.2307/4079367
Smith, E. 1928. Black Swifts nesting behind a waterfall. Condor 30(2):136-138. https://doi.org/10.2307/1363156
Sparks, R. 2018. Black Swifts: new horizons. Bird Conservancy of the Rockies, Brighton, Colorado, USA. https://birdconservancy.org/black-swifts-new-horizons/
Tattersall, I. 1987. Cathemeral activity in primates: a definition. Folia Primatologica 49:200-202. https://doi.org/10.1159/000156323
Fig. 1

Fig. 1. Identifying the male and female at the nest. The swifts’ heads are to the left. The male’s forked tail is distinguishable from the female’s square to rounded tail. Photo captured with infrared light on 4 June 2021, 22:04:58.

Fig. 2
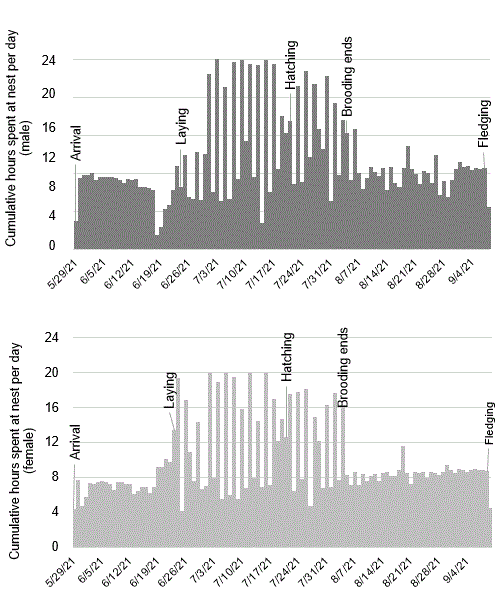
Fig. 2. Cumulative number of hours spent at the nest by the male and female throughout the breeding season with salient events labeled. From arrival to laying and from the end of brooding to fledging, both adults were away from the nest during daylight hours, most likely foraging.

Fig. 3
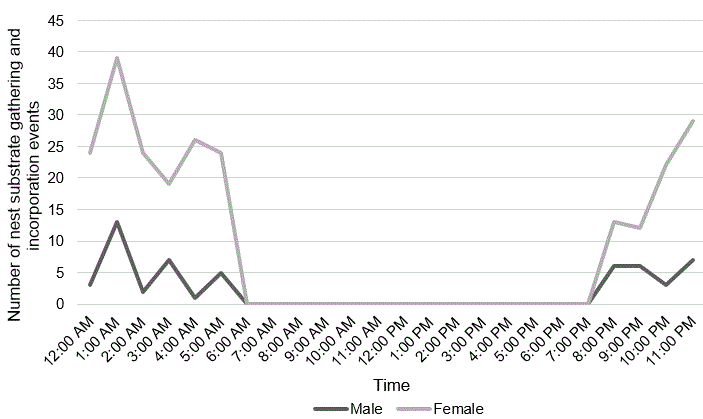
Fig. 3. Nest-building timescale for male and female. No nest-building activities occurred during daylight.

Fig. 4
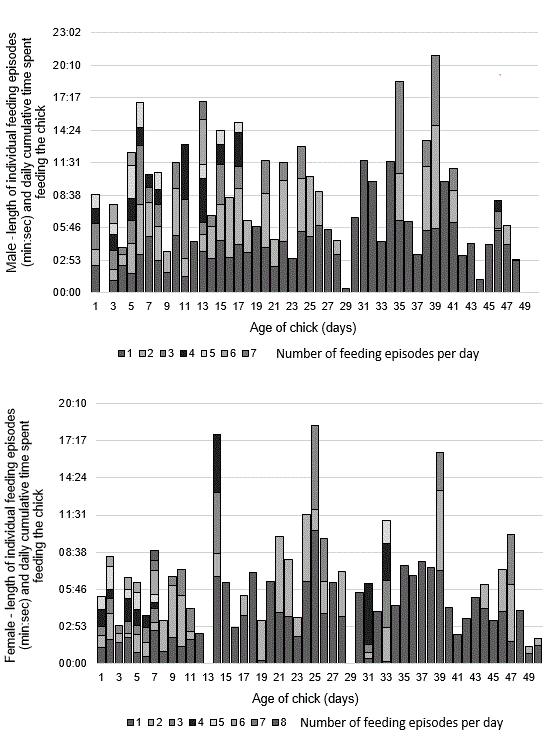
Fig. 4. Nestling provisioning. Each stacked vertical bar represents the number of feeding episodes per day from hatching to fledging for the male and female and cumulative amount of time per day spent feeding the chick.

Fig. 5
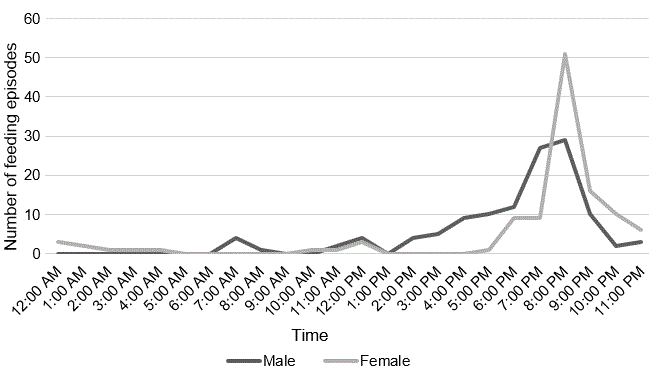
Fig. 5. Nestling provisioning timescale. Except for daytime feedings when the nestling was small, provisioning occurred after dusk when adults returned to the nest.

Fig. 6
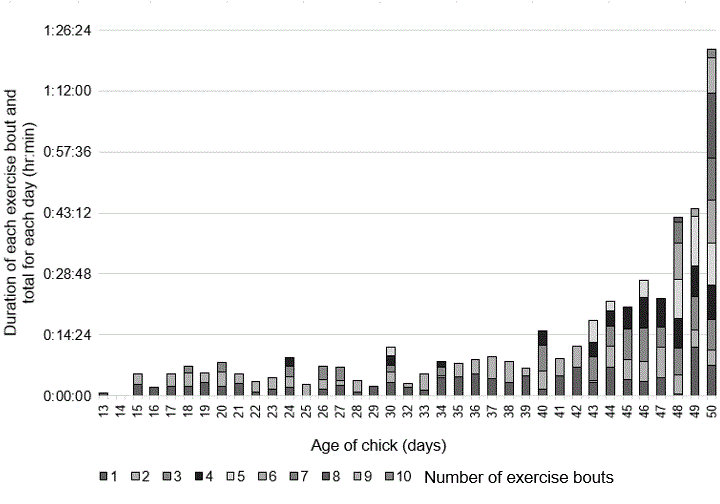
Fig. 6. Nestling wing exercises. Wing exercises increased in frequency and duration as the chick grew.

Fig. 7
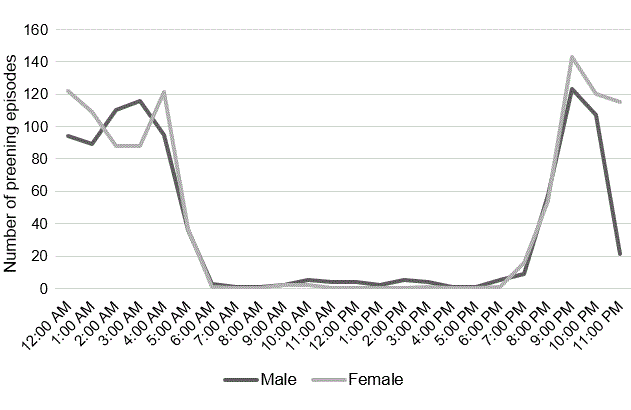
Fig. 7. Adult preening timescale. Most preening episodes occurred at dusk, nighttime, or before dawn; adults may have preened while away from the nest, either in flight or roosting on rocks near the nest but out of the view of the camera, or at some other location at the colony.

Table 1
Table 1. Comparisons between male and female activities during the nesting season.
| Activity | Male | Female |
| Nest-building | ||
| Vegetation gathering and delivery (from the time the swift left the nest until it returned) | Averaged 3 min 26 s (range 54 s to 5 min 59 s, for a total time of 2 h 42 min, N = 47) | Averaged 2 min 12 s (range 26 s to 4 min 47 s, for a total time of 8 h 11 min, N = 223) |
| Incorporating gathered substrate into the nest | Averaged 1 min 56 s (range 1 s to 4 min 50 s, for a total of 1 h 44 min) | Averaged 2 min 22 s (range 0.1 s to 7 min 56 s, for a total of 9 h 9 min) |
| Total - gathering and incorporating nest substrate | 4 h 26 min (20.4% of effort) | 17 h 20 min (79.6% of effort) |
| Substrates collected | 93% moss, 7% unidentified vegetation | 55% moss, 23% fine grass stems, 12% fern tips, 10% spruce needles, leaves, and mixed or unidentified vegetation |
| Incubation | ||
| Total incubation (from laying on 24 June until hatching on 21 July; a total of 27 days) | 267 h 43 min (45.8%) | 317 h 9 min (54.2%) |
| Effective incubation (from 1 July when consistent incubation began until hatching on 21 July; a total of 20 days) | 238 h 47 min (49%) | 246 h 53 min (51%) |
| Mean daily incubation (from 1 July when consistent incubation began though hatching on 21 July) | 11 h 22 min | 11 h 43 min |
| Mean daily egg neglect from laying on 24 June to hatching on 21 July | 2 h 33 min (total for both adults) | |
| Mean daily egg neglect from onset of consistent incubation on 1 July to hatching on 21 July | 42 min (total for both adults) | |
| Longest incubation bout without a break |
19 h 23 min | 18 h 24 min |
| Brooding | ||
| Brooding (from hatching on 21 July until the chick could thermoregulate at age 15 days on 5 August) | 143 h 37 min (47%) | 161 h 48 min (53%) |
| Longest brooding bout without a break |
16 h 21 min | 16 h 16 min |
| Provisioning the nestling | ||
| Feeding episodes (from the time the adult starting to regurgitate food until the last portion was given) | A total of 122 feeding episodes (51.5%), delivering 771 individual portions | A total of 115 feeding episodes (48.5%), delivering 593 individual portions |
| Duration of feeding episodes | Averaged 3 min 23 s (range 2 s to 11 min 45 s); intervals between feeding episodes (range 5 h 57 min to 26 h 24 min) | Averaged 2 min 45 s (range 8 s to 10 min 17 s); intervals between feeding episodes (range 1 min 25 s to 26 h 59 min) |
| Number of portions/single feeding episode |
1–17 | 1–16 |
| Preening | ||
| Number, of preening episodes by adults throughout the breeding season | 988 | 1018 |
| Duration | Averaged 2 min 8 s (range 5 s to 16 min 3 s) | Averaged 1 min 35 s (range 7 s to 7 min 35 s) |
| Total preening time at nest | 35 h 6 min (1.4% of the daily time budget) | 26 h 46 min (1.0% of the daily time budget) |
| Range of preening episodes per 24 h period | 0–27 | 0–33 |
| Maximum total time spent preening in a single 24 h period | 1 h 27 min (6% of that days’ time budget) | 1 h 4 min (4.4% of that days’ time budget) |
Table 2
Table 2. Nestling development from hatching to fledging including feather growth and behaviors.
| Date | Age (days) | Observations |
| 21 July | 0 | Hatching |
| 22 July | 1 | Skin shiny dusky pink except the eyes (closed) and wings which are gray; able to abduct wings; when alone it can rapidly rock from side to side with head down; sits back on pelvis and able to raise head and chest to beg with open beak but cannot stand. |
| 23 July | 2 | Able to elevate body and support itself on its wingtips for feeding. |
| 24–26 July | 3–5 | Skin is shiny dark dusky pink; forming semiplumes visible in tracts under skin; rests on ventrum and rocks from side to side for minutes at a time; on day 3 it is able to maneuver its posterior to produce a fecal sac on the rim of the nest which the adult immediately eats. |
| 27 July | 6 | Skin is shiny dusky pink, less dark on malar and ventral cervical area; eyes closed; movements weak and uncoordinated; eggshells still present in the nest. |
| 28 July | 7 | Able to position cloaca at the edge of nest and project feces past the rim of the nest by standing on legs and supporting itself on its wingtips; primary sheaths visible under skin of metacarpals and phalanges. |
| 29–30 July | 8–9 | Eyes partially open; primary sheaths emerging from skin 1 mm; semiplumes emerging along the capital, cervical, humeral, scapular, femoral, crural, and spinal tracts. |
| 31 July–August 1 | 10–11 | Semiplumes more dense, side of neck bare; turns head fully to right and left to preen behind wings and yawns; primary sheaths 2 mm long, secondary sheaths emerged through skin 1 mm; both eyes open on day 11. |
| 2 August | 12 | Only a few apteria now visible as semiplumes are denser and longer; no semiplumes in the propatagium, neck, lores, or around eyes; primary and secondary sheaths 2–4 mm long. |
| 3–4 August | 13–14 | Primary sheaths 5 mm and secondary sheaths 4 mm, no evidence of rectrices; body and most of head covered in dense black semiplumes; able to preen its back, stretches wings backwards and alongside the body; exercises wings briefly; see text for further descriptions of wing exercises. |
| 5–6 August | 15–16 | Primary and secondary feathers emerging from sheaths; greater and primary covert sheaths 2 mm long; no down around beak, eyes, or metacarpals; no rectrix visible beyond semiplumes; digs around in nest cup as if gleaning dropped insects. |
| 7–8 August | 17–18 | Primary and secondary feather sheaths 5–6 mm long with feathers extending 2 mm beyond sheaths; greater and primary covert sheaths 4 mm; long, thick black semiplumes covering body and face; white facial markings visible. |
| 9–10 August | 19–20 | Primary and secondary feathers emerging 6–8 mm beyond sheaths with white tips visible; rectrices just visible extending beyond dense semiplumes; egg tooth still present. |
| 11–12 August | 21–22 | Primary and secondary feathers extend 10–12 mm beyond sheaths with white chevron tips clearly visible; feathers extending 2–4 mm beyond greater and primary covert sheaths; rectrices extend 3–4 mm beyond semiplumes. |
| 13–14 August | 23–24 | Primaries extend 14–16 mm beyond sheaths which are beginning to fray; difficult to gauge length of secondaries; rectrices extend 4–5 mm beyond semiplumes and have white tips; frontal region of capital tract feather sheaths just emerging above beak; egg tooth not visible. |
| 15–17 August | 25–27 | Primaries 18–20 mm long; greater and primary coverts with white tips filling in and covering remaining feather sheaths; feather sheaths of coronal region of capital tract and cervical and scapular region 2 mm long; semiplumes still in the superciliary area and rest of body; rectrices extend 5–6 mm beyond semiplumes; upper tail coverts 3–4 mm long. After this age, it was difficult to determine length of exposed primaries, secondaries, and rectrices, and was not documented. |
| 18–20 August | 28–30 | Lesser and median coverts emerged; more frontal and coronal pinfeathers emerging giving the nestling the appearance of a sloping forehead; semiplumes still present in the superciliary, auricular, occipital, cervical, interscapular, dorsal, and pelvic regions. |
| 21–23 August | 31–33 | Semiplumes in superciliary, auricular, post auricular, malar, submalar, and dorsal region are replaced by emerging contour feathers; semiplumes still present in cervical area. |
| 24–26 August | 34–36 | Semiplumes replaced by contour feathers over body except in cervical and interscapular regions; undertail coverts with 2–3 mm white tips. |
| 27–31 August | 37–41 | Contour feathers emerging from sheaths on frontal and coronal region of capital tract with frosted tips present. |
| 1–5 September | 42–46 | All semiplumes now replaced by contour feathers. |
| 6–10 September | 47–51 | Nestling indistinguishable from adults except for white chevrons on tips of primaries and extensive white tipping of head, wing, rump, and tail coverts. |









