The following is the established format for referencing this article:
Harding, L. E., and J. I. Mesler. 2022. Cormorant abundance, diet, and foraging habits in Arizona. Journal of Field Ornithology 93(3):6.ABSTRACT
In Arizona, USA, Double-crested Cormorants (Nannopterum auritum) primarily overwinter, whereas Neotropic Cormorants (N. brasilianum) are common residents year round. Concern exists that both species are increasing in numbers and may have a negative impact on fish populations. We collected data during January 2017–April 2019 to characterize cormorant numbers seasonally, describe their foraging patterns, and evaluate environmental factors associated with cormorant numbers at community fishing waters. With more than 100 trained volunteers, we surveyed water bodies across the state simultaneously once each quarter of the year during January 2017–March 2018. We also visited 36 community fishing waters and other locations to document cormorant foraging activities and to explore potential associations between environmental factors and the maximum number of cormorants counted at each site. We collected stomach contents from lethally sampled cormorants to examine size and species of consumed fish. Our seasonal counts showed the greatest cormorant numbers during the winter and spring, with larger concentrations in metropolitan areas and fewer birds at higher elevations. At community fishing waters, cormorants fed primarily on fish ≤12 cm long, though they did consume larger sport fish in reservoirs and along the Colorado River. At community fishing waters, cormorant numbers were positively associated with the number of artificial structures in/near the water’s edge and with the number of large trees within 200 meters of the water. To reduce cormorant numbers and their anticipated impacts, we recommend stocking larger, stout-bodied fish and removing woody vegetation close to shorelines while adding fully submerged structures to provide security cover for fish from cormorants. This study is unique and important in that it addresses cormorant populations located in the southwestern USA, an area that has not historically been viewed as waterbird habitat, and it presents an unbiased evaluation of cormorant foraging activities in desert areas manipulated by water. It also offers reasonable, non-lethal actions to reducing potential cormorant impacts on aquatic resources.RESUMEN
INTRODUCTION
Cormorant (Phalacrocorax and Nannopterum spp.) habits and impacts are relatively well documented in Europe and Asia (e.g., Russell et al. 2012, Klimaszyk and Rzymski 2016, and citations therein), eastern North America (USFWS 2003 and citations therein), and along the Pacific Coast (e.g., Carter et al. 1995, Adkins et al. 2014, Herbert et al. 2014), but very little is known of their habits and population dynamics in the southwestern USA. In the last 40 years, cormorant numbers have increased significantly throughout Europe (e.g., Russell et al. 2012, Klimaszyk and Rzymski 2016, and citations therein) and North America (e.g., Weseloh et al. 1995, Pacific Flyway Council 2012), creating a variety of concerns related to habitat changes, including guano killing trees (e.g., Hebert et al. 2014, Ayers et al. 2015, Klimaszyk and Rzymski 2016, Lafferty et al. 2016), potential negative impacts on fisheries (e.g., Rudstam et al. 2004, Dorr et al. 2012, van Eerden et al. 2012, Schultz et al. 2013), reduction of waterbird diversity (Farquhar et al. 2012, Dorr and Fielder 2017), property damage (Dorr et al. 2014), and sanitation concerns (Klimaszyk and Rzymski 2016).
Prior to 1998, conflict issues with cormorants in the USA were largely managed through non-lethal methods, with some limited lethal take authorized by the U. S. Fish and Wildlife Service (USFWS) under depredation permits on a case by case basis (Wires et al. 2001). After 1998, Double-crested Cormorants (Nannopterum auritum) in the eastern United States were managed under the USFWS Aquaculture Depredation Order (AQDO; 50 Code of Federal Regulations, i.e., CFR 21.47) and after 2003 also under the Public Resource Depredation Order (PRDO; formerly in 50 CFR 21.48), which allowed take without a permit to reduce conflicts with aquaculture and public resources, including fisheries. Both the AQDO and PRDO were vacated by lawsuit in 2016. However, these regulations had little or no effect on western populations because they did not apply to the western states. Currently, depredation permits can be obtained for Neotropic Cormorants (N. brasilianum) and there was a separate special depredation permit for Double-crested Cormorants established in February 2021 (50 CFR 21.123; USFWS 2020a).
Arizona is inhabited by two species of cormorants that have been protected under the Migratory Bird Treaty Act since 1972 (USFWS 2013). Neotropic Cormorants are present as resident populations, whereas Double-crested Cormorants are present in large numbers primarily during winter and spring. Double-crested Cormorants are the most abundant and widely distributed of the six species of cormorants in North America (Pacific Flyway Council 2012). Historically, Double-crested Cormorants were at low population levels and caused relatively little concern to resource managers in Arizona (USFWS 2003). Bird counts such as the Christmas Bird Count (CBC; National Aububon Society 2010), state waterbird surveys by the Arizona Game and Fish Department (hereafter AZGFD, unpublished data), and public birding observations (recorded in e.g., www.eBird.org) suggest that numbers of both species are increasing in Arizona, particularly for Neotropic Cormorants. Some data also suggest an expanding distribution for Neotropic Cormorants in Arizona, although they are still relatively rare in other western states (eBird 2018).
Cormorants are piscivorous and often form sizeable colonies that can collectively consume large numbers of fish (Dorr et al. 2014, Klimaszyk and Rzymski 2016, Telfair and Morrison 2020). Cormorants may forage up to 62 km (38.5 mi) from nesting or nighttime roosts, and their daily foraging patterns are dynamic, with colonies changing feeding sites in response to changes in fish availability (Anderson et al. 2004, Lyons et al. 2007). In Arizona, both species have been observed in large numbers at daytime feeding and loafing sites, night roosts, and nesting areas. Their apparent expansion in Arizona has caused concerns over their perceived or real impacts on both the sport fish industry and native fish conservation. For example, anglers in Arizona have voiced concerns that cormorant numbers and their nearly year-round residency have negatively impacted sport fishing opportunities and success across the state. However, few studies have been conducted on cormorants in the Southwest, particularly for Neotropic Cormorants, and the lack of empirical data on cormorant populations in the southwestern USA creates challenges in knowing what impacts the birds may have, but also for developing timely and effective management strategies to address them (Dorr and Somers 2012).
The goal of our study was to collect empirical data to inform management decisions related to cormorant population management, fish stocking, and aquatic habitat enhancement practices in Arizona. Our first objective was to seasonally estimate a minimum count of both Double-crested and Neotropic Cormorants in Arizona. Second, we sought to investigate the movement of individual birds across the landscape. Our third objective was to describe the composition of consumed fish by size, shape, and species, and our last objective was to evaluate potential associations between environmental factors and the number of cormorants at community fishing waters.
METHODS
Study area
Well known for its vast desert landscapes, Arizona, USA, is also home to numerous large reservoirs and hundreds of man-made lakes and ponds, many of which are stocked seasonally with sport fish. In addition, Arizona’s land mass is bounded by the Colorado River on its west side, and rivers such as the Gila, Salt, and Verde flow through much of the state’s interior. Ecosystems and elevations in Arizona vary from low-elevation Sonoran Desert (~21 m) to alpine systems above 3658 m. Mean temperatures may range from -7°C at high elevations in winter to 41°C, and frequently exceed 43°C in the low deserts in summer (http://www.ncei.noaa.gov). Precipitation may come as snowfall in the mountains, as monsoonal moisture in the summer, and as winter rains from the Pacific. Land use in Arizona is partitioned between metropolitan centers, ranching and agricultural communities, Native American lands, and vast expanses of state and federal land.
To address our objectives, our study included both naturally occurring and artificial waters on non-tribal land throughout Arizona. Natural waters and larger reservoirs, such as the Colorado River and its tributaries, were included because they presented important waterbird habitat. Cormorant stomach contents were also sampled from the Colorado River and other large reservoirs because these waters contained both native fish (e.g., razorback sucker, Xyraucben texanus; bonytail chub, Gila elegans) and sport fish. The last objective focused on artificial waters that support AZGFD’s community fishing program (CFP) because of concerns related to increased cormorant presence at these sites. To address this objective, we selected CFP waters (n = 36) located in the Phoenix, Prescott, Tucson, and Yuma metropolitan areas (Fig. 1, Table A1.1). All CFP waters were stocked every 2-4 weeks by AZGFD and, given that all waters in the CFP were artificial, many had limited cover and little to no natural or artificial habitat suitable for self-sustaining populations of sport fish (e.g., trout, Oncorhynchus, Salmo spp.; largemouth bass, Micropterus salmoides; and channel catfish, Ictalurus punctatus). Many sites also contained other forage or prey species, like tilapia (Oreochromis spp.), threadfin or gizzard shad (Dorosoma petenense, D. cepedianum), and fathead minnows (Pimephales promelas). For CFP waters developed in the last decade, AZGFD commonly stocked a “starter package” containing fathead minnows, small largemouth bass, and small bluegill (Lepomis macrochirus) that were allowed to grow for several months before larger sport fish species were stocked. Shad species and tilapia were often introduced to waters by municipalities or communities as control agents for algae or aquatic insects and larvae.
Estimating minimum population size
To generate minimum counts of each cormorant species in Arizona, we recruited more than 100 citizen observers to assist in conducting seasonal statewide counts for cormorants in January, March, May, and September 2017, and again in January and March 2018. We trained citizen observers in cormorant species identification and survey protocols. In efforts to visit as many waters as possible each season, we assigned observers specific, non-overlapping geographic areas within metropolitan areas and asked them to count cormorants at all accessible waters within those areas during each quarterly count. We also assigned volunteers to count at large reservoirs and wildlife refuges that contained water bodies across the state. We conducted each count on one day per season between 0700 and 1200, assuming that counts conducted in the same five-hour window would reduce the potential of double-counting birds as they moved from night roosts or between daily feeding and loafing sites. At each water body, observers counted adult and juvenile cormorants by species, as well as any cormorants they could not identify to species. For January counts, when it was still dark at 0700, we shifted observation start times to correspond with sunrise. We compiled and plotted count locations and conducted all subsequent spatial analyses in a GIS platform (ArcGIS, v. 10.4.1; ESRI 2018). We standardized an observation rate (birds/hour) each season by dividing the total number of observed cormorants by the sum of all reported observer hours.
Cormorant movements
We captured cormorants to document their movements across Arizona. Between August 2017 and April 2018, we used modified noose mats akin to bal-chatri traps (Foster and Fitzgerald 1982, Schemnitz 1996), bow nets and small throw nets, and nighttime netting from kayaks (King et al. 1994) to capture and mark a subset of cormorants within Arizona. While set, traps remained under constant surveillance so that snared birds could be quickly processed and released. All animals were handled using standardized AZGFD avian handling practices in adherence with stipulations outlined in the 50 CFR 21 guidelines (USFWS 2020b) and the Federal Bird Banding Lab (BBL) for permitting purposes. Only staff authorized on AZGFD’s Federal Bird Banding permit (USDA-BBL permit 06613) tagged cormorants. We placed captured cormorants in damp pillowcases for holding and covered their eyes and heads during handling to prevent injury and reduce stress. We minimized bird handling time and tagged each bird with a colored, uniquely numbered patagial tag on each wing and released tagged birds at their capture sites.
Composition and size of consumed fish
We examined characteristics of consumed fish in three ways. First, AZGFD staff and trained citizen observers documented cormorants’ feeding (i.e., actively diving for fish and swallowing or manipulating a fish in the bill upon resurfacing). Each observer was assigned an area and asked to visit any water bodies within it at least once each quarter. On site, an observer recorded the location, time, and date of the observation. When cormorants were present, observers were asked to watch a group of cormorants for 15 minutes and note the following each time they saw a cormorant with a fish: (1) cormorant species and age (adult, juvenile), (2) size of the fish, as estimated relative to the length of the bird’s bill in half-bill length increments (e.g., ≤ 1 x, 1.5 x, 2.0 x bill length), (3) fish shape (e.g., slim and elongated versus short and round), (4) fish color and species, if feasible, and (5) whether or not the bird was successful in swallowing the fish. At the end of the 15 minutes, if more than 1 group of cormorants was present, observers were asked to select another group of cormorants and repeat the process. Our observers were asked to record at least 10 15-minute sessions each quarter during their visits to various waters. Each observation of a cormorant feeding was considered an independent event, even if observers knowingly or incidentally watched the same bird or birds from the same group consume or attempt to swallow several fish.
As a second means of characterizing cormorant prey, we collected samples that captured cormorants regurgitated in the traps noted above. We collected and froze the regurgitant to visually examine and identify prey species as feasible. Cormorant species and age, number of fish, total fish length (cm) or estimated length of partial fish (cm; compared to intact reference fish in the sample), fish species (if possible), body girth (cm), body shape, and color were also recorded.
Lastly, we obtained a scientific collection permit from the USFWS (Permit MB02476D-0) that allowed the lethal collection of a sample (n = 10-120 birds in specified counties) of each of the 2 species to examine stomach contents. We identified potential collection sites from our quarterly counts and contracted the U. S. Department of Agriculture Animal and Plant Health Inspection Service Wildlife Services (hereafter Wildlife Services) to collect cormorants using shotguns from January through March 2019 when cormorants were abundant in Arizona. Wildlife Services agents were instructed to collect birds after they had opportunities to forage for fish in the morning hours. Upon collection, carcasses were frozen immediately and we later necropsied birds to remove any fish or fish parts from the gullet and stomach of each individual. Fish samples were identified and documented as above. No attempt was made to identify fish bones or substantially digested stomach contents. We also measured and calculated an average bill length (cm) for each cormorant species on a subsample of the birds that were lethally collected and used mean bill lengths to estimate length for fish captured/consumed by cormorants during feeding observations relative to the length of the bird’s bill.
To examine potential cormorant take of fish specifically at CFP sites, we used a mean value from published ranges of daily dietary requirements for fledged Double-crested Cormorants in the northeastern USA (0.750 kg/bird/day; Schultz et al. 2013) and Neotropic Cormorants in South America (0.425 kg/bird/day; Barquete et al. 2008) to estimate take by each cormorant species per day, regardless of what fish species were present. We multiplied the daily dietary estimate by the maximum number of cormorants by species we observed during any one quarterly count at each CFP site, assuming all birds to be fledged, to generate an estimate of potential mass of fish consumed during any one day cormorants were present in a quarter at a site. We then divided potential mass of fish consumed by the surface area (in hectares) of the CFP site to standardize the estimate across sites that varied in size. We acknowledge this may under- or overestimate the total mass of fish consumed across time because bird numbers at any given site likely fluctuate.
Cormorants at community fishing waters
To evaluate environmental factors potentially associated with the numbers of cormorants at CFP waters (Fig. 1), we visited operating CFP waters (n = 36; Table A1.1) in the Phoenix area, Prescott, Tucson, and Yuma to collect covariate data on site. These waters were stocked with sport fish by the AZGFD, had high angling demand and activity, and some experienced high numbers of cormorants whereas others had low numbers of cormorants. We initially identified covariates based on proposed environmental variables in existing literature or postulated by AZGFD personnel working at CFP waters to affect cormorant abundance at waters. Covariate data collected at each site (Table A1.2) included the number of artificial features (e.g., docks, ‘reef’ blocks, low head dams, buoys) floating, partially submerged or visible under the water, or on the ground within three meters of the shoreline, as well as the number of natural structures (e.g., aquatic vegetation, boulders, deadfall) and any islands present in the water. Usually, water clarity and depth visually obscured any fully submerged features farther than the first few meters from the shoreline, and AZGFD had scant records on what types or numbers of underwater features were available to fish, so we did not quantify underwater features beyond what we could see from the shore. We also visually estimated primary plant cover as the percent cover of bare ground, herbaceous vegetation, shrubs, or trees within three meters of shorelines and recorded vegetation types on islands.
Cormorants use large trees for roosting, nesting, and perhaps as perching structures to view fish (e.g., USFWS 2003, 2017, Koh et al. 2012), so we also visually counted the number of trees > 2 m tall within 200 m of shorelines that could be used for roosting or loafing between feeding bouts and the number of active nests in the same area (Table A1.2). We relied on published descriptions of CFP waters from AZGFD’s community fishing guidebook (https://www.azgfd.com/fishing/community/) to characterize water size and depth and the stocked fish species at each site. We also included days since fish stocking events, based on AZGFD fish stocking records. We used standardized criteria for visually estimating covariates, and whenever possible, the same observer scored covariates at CFP sites to minimize any observer bias. We also collected data on human activity, e.g., the number of people and dogs present, because we postulated they could significantly alter cormorant numbers at a site.
We then took the maximum number of cormorants counted at a CFP water site during any of our quarterly counts as the dependent variable and used multivariate regression analyses to examine potential relationships between the above covariates and the maximum number of cormorants counted. To do so, we first removed covariates that we observed were greatly influenced by the time of day or season recorded (e.g., the number of people and dogs present) or with lower biological relevance to cormorant presence (e.g., tree cover). Then we performed a correlation test on remaining habitat covariates using the Pearson correlation coefficient and considered variables to be correlated when r² ≥ 0.50. For correlated covariates, we included the variable we deemed most biologically relevant to cormorants in the model. However, too many variables still remained that were below the cut-off value (r² ≥ 0.50), so we removed additional parameters that we deemed redundant to variables included in the final model (e.g., we removed covariates for the number of roost trees and the number of trees > 2 m tall within 3 m of shore because we included a variable for the number of trees > 2 m tall within 200 m of shore, which contained all the trees covered by the former 2 covariates).
We determined that our data were overdispersed by comparing model residual deviance to the residual degrees of freedom (Harrison 2014), possibly as a result of zero inflation in our count data caused by the tendency of birds to clump (Schneider and Duffy 1985, Lindén and Mäntyniemi 2011). A negative binomial GLM was selected because of its higher performance when using overdispersed data compared to Poisson models (White and Bennetts 1996, Warton et al. 2016). We conducted all statistical analyses in program R and tested all possible combinations (n = 120) of selected covariates using the “MuMIn_dredge” package (Barton 2013) and ranked models by comparing Akaike’s Information Criterion (AIC) values (Burnham and Anderson 2002).
RESULTS
Estimated minimum abundance
We surveyed 1048 unique bodies of water between January 2017 and March 2018. Our quarterly statewide counts suggested that cormorants were most prevalent during winter 2017 (Fig. 2). The total number of cormorants then declined by more than half in the spring, was lowest during summer, increased during fall, and peaked again in winter 2018. Overall cormorant abundance (i.e., both species and unidentified cormorants) was 4.4 times greater in winter than in summer. Neotropic Cormorant numbers were 9.5 times more abundant in winter than summer and remained higher in summer than in fall, and Double-crested Cormorants were 9.3 times more abundant in winter than summer, when they were at their lowest numbers. Observer effort varied by season, but observation rate (birds per hour) followed the same general trend as cormorant numbers, being highest in winter and spring and lower in summer and fall (Fig. 2).
Geographically, we observed higher numbers of Neotropic Cormorants in all seasons within the Phoenix and Tucson metropolitan areas than in other areas of the state. In winter, Double-crested Cormorants were observed in relatively high numbers within the Phoenix area, near Yuma, and along the Colorado River in western Arizona, but also occurred in isolated lakes and at higher elevations during the spring and fall seasons.
Cormorant movements
We captured and tagged 23 Neotropic Cormorants (17 adults, 6 juveniles) and 3 juvenile Double-crested Cormorants. We trapped all birds from three sites in the Phoenix metropolitan area despite our efforts to trap at several locations. We caught 23 birds using the modified noose mats and flushed 3 from night roosts and netted them on the water. We asked citizen observers, birdwatchers, and the general public to report tagged birds from September 2017-December 2018. We observed 13 tagged individuals a total of 51 times at 12 different water bodies throughout Phoenix from September 2017-April 2018. Eight tagged individuals were reported on greater than one occasion (four were observed twice after capture, and four individuals were seen an additional three, four, five, and six times after capture), whereas five cormorants had only one sighting after capture. We documented mortality of one tagged cormorant a few days post release. Time between consecutive sightings of tagged individuals averaged 92 days (SD = 66.7) and ranged 12-210 days. Assuming linear distances between observation points, tagged cormorants traveled a mean minimum distance of 17.5 km (SD = 25.1 km) and ranged 0.47-78.1 km from their original capture locations. Unfortunately, we were unable to trap and mark a sufficient number of cormorants to inform our second objective farther than we have reported with the estimates here.
Composition and size of fish
In total, we completed 7634 observations of cormorants. Although we considered each observation of a cormorant as an independent event, our data may include repeat observations of some individual birds. Of the 7634 observations, 6110 observations (of 198 Double-crested, 5718 Neotropic, and 194 unknown cormorant species) were of non-feeding birds. We observed cormorants feeding or attempting to feed during 1524 observations (of 41 Double-crested and 1483 Neotropic Cormorants; Table 1).
We captured 26 cormorants, but we only collected 10 regurgitant samples from Neotropic Cormorants. Birds often vomited while caught in the nooses, but if other cormorants were nearby, free birds quickly consumed regurgitated fish. We collected 154 intact or partially digested fish in samples. We did not include regurgitant samples in the totals reported in Tables 1-3 because only one-third of the fish (n = 51/154) were intact. Mean length of regurgitated fish, including both actual or estimated length, was 4.5 cm (n = 154, range = 0.4-11.4 cm); mean girth of regurgitated fish, including both actual or estimated girth, was 2.6 cm (n = 154, range = 0.2-9.6 cm). None of the intact fish in regurgitant samples measured > 11.4 cm in length or > 9.6 cm in girth. Estimated average mass of fish using length and girth was 2.7 g (n = 154, range 0.001-34.2 g). Fish that could be identified included bluegill (n = 59), threadfin shad (n = 5), tilapia (n = 9), fathead minnows (n = 2), mosquitofish (Gambusia affinis; n = 26), and unknown shad (n = 17).
We lethally collected 68 cormorants from 6 counties and 11 water bodies in Arizona. Wildlife Services agents collected all cormorants between 1030 and 1645, so birds had time to forage prior to being removed. Firearm restrictions precluded efficient sampling in populated residential and commercial areas. As such, lethally sampled cormorants were removed primarily from large rivers and reservoirs rather than CFP waters as initially planned. We removed stomach contents from 43 Double-crested Cormorants (27 adults, 16 juveniles), of which 32 contained partial or whole fish. One additional bird’s stomach had only fish bones and a second had indeterminate digested material. We examined stomach contents from 25 Neotropic Cormorants (20 adults, 5 juveniles), 16 of which contained partial or whole fish. Two more contained fish bones and endoparasites, a third had only endoparasites, and a fourth contained indeterminate material. Fish species included sport fish like bluegill, largemouth bass, and channel catfish, as well as smaller forage fish like shad and minnows (Tables 2 and 3). Double-crested Cormorants ate fish primarily elongate in shape (98%; Table 2); three birds consumed fish with round body shape. One fish was a black crappie (Dorosoma cepedianum; 27.0 cm); the other 2 were a gizzard shad (32.0 cm) and an unknown shad (23.0 cm). Lethally sampled Neotropic Cormorants consumed only fish with elongate body shape (Table 3). Overall, fish eaten by Double-crested Cormorants had a mean length of 10.0 cm (range 4.0-32.5 cm, n = 134), whereas those taken by Neotropic Cormorants were smaller, with an average length of 8.5 cm (range 5.0-13.0 cm, n = 68). Approximately half the samples of forage fish species were partially digested but intact enough to estimate a minimum length, so measurements reported include minimum estimates of total length for some fish and actual total length on others. We did not measure girth of fish removed from cormorant stomachs, but they generally fell within the range of sizes we observed in both regurgitant samples and feeding observations.
We measured bill length on 38 lethally sampled cormorants. Mean bill length was 9.8 cm (range 8.4-10.5 cm, n = 32) for adult and juvenile Double-crested Cormorants combined and 8.1 cm (range 7.5-9.0 cm, n = 6) for Neotropic Cormorants. We rounded mean bill lengths to 10 cm and 8 cm, respectively, and binned fish lengths from cormorant feeding observations into half-bill increments.
During our cormorant feeding observations, we observed that many birds actively swallowed small fish as they surfaced from diving trips, suggesting cormorants likely also consumed other small prey underwater (Grémillet et al. 2006). Of the fish seen by observers when the birds surfaced (Table 1), Double-crested Cormorants primarily consumed fish estimated ≤ 10 cm in length (71.2%, n = 52 observations), whereas Neotropic Cormorants fed primarily on fish ≤ 8 cm long (92.3%, n = 1970 observations). We were unable to estimate the sizes of fish eaten by 9 birds. Only 143 of the observations made of cormorants to look for feeding activity occurred at CFP waters, though we frequently visited CFP waters throughout the year. Of the 143, 91 were non-feeding observations, leaving 52 observations of cormorants feeding or attempting to feed. At CFP waters, 4 Double-crested Cormorants successfully swallowed 2 fish estimated at 10 cm, 1 at 15 cm, and 1 elongate fish 20-cm long. A fifth Double-crested Cormorant attempted to swallow an elongate fish 20 cm long but was unsuccessful. At CFP sites, 47 observations of Neotropic Cormorants showed the birds had increasing difficulty swallowing larger and more round-shaped fish. For instance, only 1 of 19 attempts to swallow fish ≤ 8 cm long was unsuccessful, whereas 11 of 28 attempts to swallow a fish > 8 cm were unsuccessful, particularly if the fish were round (n = 9 of 11) in shape.
Estimated total mass/hectare of fish potentially consumed in a single day each quarter when cormorants were present at a CFP site (Fig. 3, Table A1.1) suggested that most CFP waters (~95%) potentially lost no more than 5 kg/ha of fish to cormorants on a given day. However, a few of the CFP sites could potentially experience significant losses to depredation by cormorants (e.g., 23 kg/ha/day at West Wetlands in winter, 36 kg/ha/day at Eldorado in spring; Table A1.1). These data also suggested that smaller waters (i.e., < 5 ha size) may experience greater proportional losses, even though larger lakes (e.g., Tempe Town Lake at 91 ha) may see more birds.
Cormorants at community fishing waters
We visited 36 CFP waters in Phoenix, Tucson, Yuma, and Prescott and collected site-specific information at each. The same observer visually estimated covariates at 75% of all CFP sites in an effort to reduce observer bias. Almost half (46%) of CFP sites lacked natural structures in the water (e.g., living vegetation, deadfall, woody debris) and 46% had natural structures present, whereas 8% only had rock features/boulders in the shallows or along the shoreline. Of the CFP sites, 89% had artificial structures in or over the water (e.g., piers, dams, bridges, sign posts, fountains). Nearly all sites (92%) had trees > 2 m tall within 3 m of the banks, ranging in number from 8 to more than 100 trees close to the water. In addition, 62% of the CFP sites had trees within 200 m of the water that we deemed as roost trees, either for cormorants or other piscivorous birds.
In our analysis of environmental variables potentially influencing cormorant numbers at CFP waters, several covariates were correlated (Table A1.2). After removing correlated covariates, we included the following covariates in our analysis of environmental factors that may influence cormorant abundance: maximum depth of CFP water at construction, time since the previous fish stocking, number of trees > 2 m tall present within 200 m of the water, distance from the water’s edge to open ground, the number of natural structures present in the water, and the number of artificial structures present within 3 m of the shoreline in/around the water. Our top models from regression analyses were within 2 AIC (Table 4), however, the only difference was the parameter for natural structures in the water. We considered this additional parameter to be uninformative because model selection by AIC does not sufficiently penalize models for the inclusion of additional parameters that do not proportionally improve model fit (Arnold 2010). Thus, the top model suggested cormorants were more likely to be present at CFP waters with a higher number of trees > 2 m tall near the water (β = 0.028, SE = 0.0068, 95% CI = 0.015, 0.042) and more artificial features in/around the water (β = 0.085, SE = 0.02, 95% CI = 0.046, 0.12).
DISCUSSION
Our quarterly survey data showed that Neotropic Cormorants in Arizona are common residents year round, with a substantial influx of migratory flocks during the winter and spring. However, Neotropic Cormorants are relative newcomers to the interior continental USA. Until the early 1970s, all known breeding colonies were restricted to coastal areas in Texas and Louisiana (Morrison 1977, Portnoy 1977, Clapp et al. 1982). It was nearly 30 years later that the first Neotropic Cormorant breeding colony was reported in southern Arizona (Stevenson and Rosenburg 2004), and now their breeding and nesting colonies are found north of the Phoenix metropolitan area into central Arizona (AZGFD, unpublished data). Likewise, our data suggested that Arizona now has annual resident populations of Double-crested Cormorants, with greater numbers during winter and spring as migratory flocks arrive. Whereas Double-crested Cormorants were formerly reported to migrate to Mexico and coastal areas of the USA to winter (Dorr et al. 2014, USFWS 2017), we observed large concentrations of cormorants during the winter and spring months in Arizona’s metropolitan areas. Both species were commonly observed at lower elevations during the winter months, not only during nesting and breeding periods. Double-crested Cormorants were also observed during winter at higher elevations, at lakes where ice did not frequently persist. As temperatures rose in the spring and early summer, our quarterly counts also showed numbers declined for both species in desert areas and increased above observed winter numbers at higher-elevation lakes and ponds.
Increased aquaculture and artificial waters inland now support migratory populations wintering and breeding inland in several southern states (USFWS 2017). Similarly, with the proliferation of artificial aquatic ecosystems, cormorants in Arizona have greatly expanded habitat available in which to reside, winter, and nest in the state. Also, several of the artificial waters contain Tilapia species to control aquatic vegetation, and tilapia mature quickly and begin reproducing as early as six months (McBay 1961, Duponchelle and Panfili 1998), thus providing increased forage of appropriately sized prey for cormorants.
Although we were unable to trap and mark sufficient numbers of cormorants to collect adequate data on movement or where individual birds spent their time, we suggest this type of data would be useful to resource managers. Such information would help in understanding flock dynamics, how cormorants move around within the state, and whether or not they move out of the state on a seasonal basis.
Our data suggested that cormorants, and particularly Neotropic Cormorants, fed primarily on smaller fish (< 12 cm) rather than taking larger stocked sport fish preferred by anglers. The majority (92.6%) of fish collected from lethally collected birds in our study were relatively small lengths (< 16.2 cm), although longer fish were taken by Double-crested Cormorants. As well, very few cormorants observed in foraging bouts attempted to take fish > 16 cm (1.3%), which are smaller than stocked sizes, and nearly one-third of those observed attempts were unsuccessful. We estimated that > 70% of Double-crested Cormorant prey were ≤ 10 cm and > 90% of Neotropic Cormorant prey were ≤ 8 cm long. Indeed, we observed many birds swallowing small fish within seconds of surfacing from dives before we could estimate fish length or see more than a flash of color in the bird’s bill. Considering how easy it may be to miss seeing or misjudge size for a fish smaller than a cormorant’s bill, the numbers of small fish consumed are likely to be underestimated. For Neotropic Cormorants, our results are consistent with those of Telfair and Morrison (2020), who also reported that about 90% of prey taken by Neotropic Cormorants in Texas were ≤ 8 cm. Likewise, Barquete et al. (2008) reported that Neotropic Cormorants typically consumed fish 5-10 cm long in Brazil and that fish 30 cm in length might represent the upper limit of what a Neotropic Cormorant is capable of swallowing.
Our results suggested that although Double-crested Cormorants may occasionally take larger fish, they more frequently consume small prey. This contrasts findings in Texas that they consumed farmed channel catfish up to 42 cm long (Campo et al. 1993) but supports other evidence that Double-crested Cormorants on aquaculture ponds in the eastern USA primarily selected for smaller fish 10-15 cm long (Schramm et al. 1984, Stickley 1991, Stickley et al. 1992), e.g., selecting schooling species like gizzard shad that were present in ponds, over channel catfish of any size (Glahn et al. 1995). Likewise, in natural aquatic habitats, Double-crested Cormorants in the eastern USA ate both native and stocked fish that averaged approximately 12 cm, even when larger fish were available (Durham 1955, Hirsch 1986, Hobson et al. 1989, Campo et al. 1993, Glahn et al. 1998, USFWS 2017). By comparison, Great Cormorants (Phalacrocorax carbo) in Europe, which are very similar to Double-crested Cormorants, consumed fish of equivalent sizes as well (e.g., Opačak et al. 2004, van Eerden et al. 2012). That cormorants fed on smaller fish also demonstrates that they may perhaps be capitalizing on prey from the most prolific phases of fish growth (i.e., juveniles), where available numbers are highest, rather than on less abundant adult fish (Opačak et al. 2004, van Eerden et al. 2012, and references therein). We acknowledge that knowing the abundance and age structure of both sport fish and forage species in each water would have been useful covariates in our analyses, but we were unable to include information for either because records of their actual abundance and ages were lacking.
Lengths of fish stocked by contracted suppliers to Arizona’s CFP waters are generally > 25-30 cm (AZGFD, unpublished data). However, trout from AZGFD hatcheries that are also stocked into CFP waters may be smaller than fish from vendors, but they still commonly exceed 20 cm in length before stocking (AZGFD, unpublished data). Similarly, channel catfish that are stocked in the Phoenix metropolitan area in the warmer months are often delivered at longer lengths and heavier sizes than the vendor contract stipulates (AZGFD, unpublished data) and may be unappealing because cormorants likely have significant difficulty manipulating and swallowing a large fish with prominent pectoral fin spines (Glahn et al. 1995).
Although we did not document cormorants frequently handling or consuming large fish, anglers and site managers at CFP waters have voiced concerns that cormorants damaged or killed large fish without swallowing them. However, research using underwater video to film foraging Double-crested Cormorants suggested that such damage was extremely rare (0.4%) when cormorants pursued small prey (i.e., prey 15-22 cm long in Grémillet et al. 2006) and that birds often successfully ingested larger fish even after an initial failed attempt underwater, rather than fatally injuring and leaving them. However, the same study also indicated that the rate of potential fish loss and injury from foraging Great Cormorants may increase with fish size in dense aquaculture areas (Grémillet et al. 2006).
Though greater fish length may hinder cormorant predation, fish shape and girth appear to be larger deterrents because they determine the mass of the largest fish a bird can handle (Riedel et al. 2007). Cormorants in our feeding observations swallowed small, round-shaped fish generally no larger than 10 cm. Regurgitated fish also were generally < 10 cm in length or girth and estimated < 35 g in mass, so their size did not likely present an obstacle to being swallowed. In contrast, Riedel et al. (2007) suggested avian predators, including cormorants, might struggle significantly more to ingest a 1000-g stout-bodied fish than to consume a large fusiform fish of equal mass. Given that larger size and shape may help fish to escape cormorant predation, fish that quickly attain a large or stout size may be preferable to stock in waters with avian predators (Riedel et al 2007). Fish species, such as gizzard shad, which grow from elongate fingerlings into larger stout-bodied fish, might be eaten easily in their smaller form but become more difficult to swallow at larger sizes. If waters in Arizona are stocked routinely with fish > 20 cm in length, it may be worthwhile to consider stocking larger fish as well as fewer fusiform fish and more stout-bodied species to reduce predation by cormorants.
Because of their piscivorous and social habits, cormorants will continue to cause concerns about potential adverse impacts to aquatic resources as their populations increase. Our estimates of fish losses at CFP sites varied widely, reflecting differences in daily cormorant numbers and should not be interpreted as exact amounts. Because cormorants are monotypic, it is often not possible to tell how many individuals forage at a site on a given day. This might be accomplished if counts could be adjusted for turnover rates of individuals at each location via individual bird identification. We had attempted to tag cormorants with unique tags, but our efforts to do so were largely unsuccessful. Therefore, we lacked the means to distinguish individuals at day to day foraging locations. Given the dynamic nature of cormorant feeding activities and movements, we caution against assuming the mass of fish consumed by birds on any given day at a CFP site represents a defined loss; rather, losses are dependent on the number of cormorants present at a particular CFP water. Values reported here thus represent a range of values in fish consumption by cormorants and may likely underestimate the total number of foraging birds and subsequent mass of fish consumed in a given area.
We expected that time since stocking might have been an important covariate in explaining cormorant numbers at CFP sites. Although cormorants do shift from site to site seeking resources (Dorr et al. 2012, USFWS 2017), the timing of stocking events was relatively unimportant in our regression models, suggesting that stocking events may not drive cormorant abundance at CFP waters.
Our data suggested that higher cormorant numbers observed at CFP waters were associated with lower numbers of underwater structures. Fully submerged natural structures, including deadfall or aquatic vegetation, were mostly lacking or could not be seen in the majority of CFP waters. Many of the CFP sites also appeared to lack fully submerged reef blocks or other artificial hiding cover for fish because they may create obstacles to angling, particularly from the shore. Perhaps stocked fish at sites without underwater cover are then particularly susceptible to cormorant predation because they may be naïve to predators and are more surface oriented and poorer swimmers than resident fish (Boström et al. 2009, Jackson and Brown 2011). Submerged structures are likely to benefit fish by providing hiding cover from aquatic or avian predators (Barlow and Bock 1984, Russell et al. 2008) and increased food resources with vegetation or by attracting aquatic macroinvertebrates and zooplankton (Dibble et al. 1997, Stahr and Kaemingk 2017). Of particular importance to fish is the amount of structural complexity any submerged or emergent vegetation may provide for security cover (Miranda and Hubbard 1994, Stahr and Shoup 2015). Conversely, if structures are floating, partially submerged, or submerged in shallow water where piscivorous birds can stand and hunt, as were many of the artificial structures (e.g., buoys, floating docks, low head dams) at CFP waters, they may negatively influence fish security by permitting cormorants to perch in open water away from shoreline predators and harassment. In particular, if fish congregate around such water features, they may provide easier predation opportunities in which birds are able to concentrate (Wires et al. 2001). As well, fish species that school, like gizzard shad, may be more visible in this behavior and easily captured by an avian predator (Glahn et al. 1995). Indeed, we observed that the largest numbers of fish taken by sampled cormorants in Arizona were small shad (Dorosoma spp.) < 10 cm in length.
Within the metropolitan areas, our regression analyses showed that cormorants were more abundant at CFP waters that had taller trees near the water’s edge. Cormorants roost, nest, and perhaps hunt fish from perches in large trees (USFWS 2003, 2017, Koh et al. 2012). Our observations suggested that trees on islands may provide even more enticing areas for cormorants, particularly when they allow birds to roost and forage with minimal harassment. Although water depth was not a significant variable in our regression analyses, previous literature suggests that it can influence cormorant foraging behaviors. For instance, cormorants may be more inclined to employ social foraging methods (Coleman 2009), or flock feed, by herding fish in shallow water (Bent 1922, Morrison and Slack 1977). We often observed both species of cormorants, particularly Neotropic Cormorants, herding fish to shallow inlets or edges of deeper water bodies to feed on them. Perhaps shallow inlets and areas near islands with trees thus permit cormorants to more easily view, pursue, and capture prey (Wires et al. 2001, Braun 2019), as well as providing warmer underwater conditions where macroinvertebrates, zooplankton and vegetation are likely more abundant to attract fish.
CONCLUSION
Our primary intent with this research was to provide empirical data to inform management of cormorants in Arizona and suggest reasonable alternatives to mitigate and manage for anticipated impacts to aquatic resources in desert environments. Although our data suggest cormorants in Arizona generally forage on small fish species rather than larger stocked sport fish, we recognize that there are localized situations in which cormorants may exert undue pressure on fish populations, particularly in smaller lakes or reservoirs with few alternative prey, so impacts are likely site-specific (USFWS 2017).
Extensive endeavors to address cormorant-fish conflicts elsewhere have resulted in pan-European collaborations (e.g., Russell et al. 2012) that address Great Cormorants, a species very similar to Double-crested Cormorants, and many discuss various harassment or exclusion techniques that may be better suited to waters that are not maintained as angling sites. However, for waters in which fisheries are maintained for recreational angling, our research suggests the following might be considered in efforts to reduce numbers of cormorants or their anticipated impacts at stocked waters:
- removing stiff or woody vegetation and trees close to the water to eliminate perch sites in which birds are free from harassment or predation pressure;
- adding fully submerged underwater structures and/or vegetation having good structural complexity to provide security cover for fish from diving cormorants; research cited in Russell et al. (2012) suggests that a refuge volume as low as 0.5-1.0% of pond volume can have marked positive effects on fish survival, particularly in the absence of other cover;
- stocking fish with lengths > 20 cm and stocking stout-bodied fish rather than fusiform fish;
- delay stocking fusiform-shaped fish until the majority of cormorants have migrated out of areas where they tend to concentrate.
Despite increasing concerns about growing cormorant populations, consensus is lacking on how many cormorants are too many cormorants in an area. Human social tolerance for cormorants varies across resource users. Resource and wildlife managers must therefore carefully evaluate available information and weigh management actions against public sentiments to set biologically and socially realistic population goals for cormorants.
RESPONSES TO THIS ARTICLE
Responses to this article are invited. If accepted for publication, your response will be hyperlinked to the article. To submit a response, follow this link. To read responses already accepted, follow this link.ACKNOWLEDGMENTS
We thank our large contingent of citizen observers for their diligent help. Without their tremendous efforts, we could not cover all areas included in this study. Many thanks to B. Russo, T. Brtis, S. Eno, C. Gray, and C. Mitchell for monumental efforts taken to trap and observe cormorants. We thank E. Rubin for help in project design, permits, and manuscript reviews. We thank J. Hill, A. Clark, S. Gurtin, K. Peterson, and T. Corman for data on AZGFD aquatic resources and practices at CFP waters and at roosting sites statewide. We thank many AZGFD staff who counted cormorants and reviewed this manuscript, as well as anonymous reviewers who helped improve the manuscript. This project was funded by The Federal Aid in Wildlife Restoration Act through grants to AZGFD.
DATA AVAILABILITY
Supplementary electronic datasets are available on Figshare https://doi.org/10.6084/m9.figshare.20264529.
LITERATURE CITED
Adkins, J. Y., D. D. Roby, D. E. Lyons, K. N. Courtot, K. Collis, H. R. Carter, W. D. Shuford, and P. J. Capitolo. 2014. Recent population size, trends, and limiting factors for the Double-crested Cormorant in Western North America. Journal of Wildlife Management 78:1131-1142. https://doi.org/10.1002/jwmg.737
Anderson, C. D., D. D. Roby, and K. Collis. 2004. Foraging patterns of male and female Double-crested Cormorants nesting in the Columbia River estuary. Canadian Journal of Zoology 82:541-554. https://doi.org/10.1139/z04-019
Arnold, T. W. 2010. Uninformative parameters and model selection using Akaike’s information criterion. Journal of Wildlife Management 74:1175-1178. https://doi.org/10.1111/j.1937-2817.2010.tb01236.x
Ayers, C. R., K. C. Hanson-Dorr, S. O’Dell, C. D. Lovell, M. L. Jones, J. R. Suckow, and B. S. Dorr. 2015. Impacts of colonial waterbirds on vegetation and restoration of island habitats. Restoration Ecology 23:252-260. https://doi.org/10.1111/rec.12169
Barlow, C. G., and K. Bock. 1984. Predation of fish in farm dams by cormorants, Phalacrocorax spp. Australian Wildlife Research 11:559-566. https://doi.org/10.1071/WR9840559
Barquete, V., L. Bugoni, and C. M. Vooren. 2008. Diet of Neotropic Cormorant (Phalacrocorax brasilianus) in an estuarine environment. Marine Biology 153:431-443. https://doi.org/10.1007/s00227-007-0824-8
Barton, K. 2013. MuMIn: Multi-model inference. R package version RStudio 1.2.5033. R Foundation for Statistical Computing, Vienna, Austria. https://cran.r-project.org/web/packages/MuMIn/MuMIn.pdf
Bent, A. C. 1922. Life histories of North American petrels and pelicans and their allies. Bulletin of the United States National Museum 121. Smithsonian Institution, Washington, D.C., USA.
Boström, M. K., S.-G. Lunneryd, L. Karlsson, and B. Ragnarsson. 2009. Cormorant impact on trout (Salmo trutta) and salmon (Salmo salar) migrating from the river Dalälven emerging in the Baltic Sea. Fisheries Research 98:16-21. https://doi.org/10.1016/j.fishres.2009.03.011
Braun, D. P. 2019. Bonytail (Gila elegans) (BONY): basic conceptual ecological model for the Lower Colorado River, 2018 updates. Submitted to the Lower Colorado River Multi-Species Conservation Program. Sound Science, Boise, Idaho, USA.
Burnham, K. P., and D. R. Anderson. 2002. Model selection and multi-model inferences: a practical-theoretic approach. Springer-Verlag, New York, New York, USA. https://doi.org/10.1007/b97636
Campo, J. J., B. C. Thompson, J. C. Barron, R. C. Telfair, II, P. P. Durocher, and S. J. Gutreuter. 1993. Diet of Double-crested Cormorants wintering in Texas. Journal of Field Ornithology 64:135-144. https://sora.unm.edu/sites/default/files/journals/jfo/v064n02/p0135-p0144.pdf
Carter, H. R., A. L. Sowls, M. S. Rodway, U. W. Wilson, R. W. Lowe, G. J. McChesney, F. Gress, and D. W. Anderson. 1995. Population size, trends, and conservation problems of the Double-crested Cormorant on the Pacific coast of North America. Colonial Waterbirds 18:189-215. https://doi.org/10.2307/1521540
Clapp, R. B., D. Morgan-Jacobs, and R. C. Banks. 1982. Marine birds of the southeastern United States and Gulf of Mexico. Part I. Gaviformes through Pelicaniformes. FWS/OBS-82/01. U.S. Fish and Wildlife Service, Office of Biological Services, Washington, D.C., USA.
Coleman, J. T. H. 2009. Diving behavior, predator-prey dynamics, and management efficacy of Double-crested Cormorants in New York State. Dissertation. Cornell University, Ithaca, New York, USA.
Dibble, E. D., K. J. Killgore, and S. L. Harrel. 1997. Assessment of fish-plant interactions. Miscellaneous Paper A-97-6. U.S. Army Engineer Waterways Experiment Station, Vicksburg, Massachusetts, USA.
Dorr, B. S., and D. G. Fielder. 2017. Double-crested Cormorants: too much of a good thing? Fisheries 42:468-477. https://doi.org/10.1080/03632415.2017.1356121
Dorr, B. S., S. L. Hanisch, P. H. Butchko, and D. G. Fielder. 2012. Management of Double-crested Cormorants to improve sport fisheries in Michigan: three case studies. Human-Wildlife Interactions 6:17. https://doi.org/10.26077/myd1-wt71
Dorr, B. S., J. J. Hatch, and D. V. Weseloh. 2014. Double-crested Cormorant (Phalacrocorax auritus) In A. Poole, editor. The birds of North America [online]. Cornell Lab of Ornithology, Ithaca, New York, USA.
Dorr, B. S., and C. Somers. 2012. The direction of research and management of Double-crested Cormorants heading into the 2000s: symposium overview and future information needs. Waterbirds 35:138-148. https://doi.org/10.1675/063.035.sp115
Duponchelle, F., and J. Panfili. 1998. Variations in age and size at maturity of female Nile tilapia, Oreochromis niloticus, populations from man-made lakes of Côte d’Ivoire. Environmental Biology of Fishes 52:453-465. https://doi.org/10.1023/A:1007453731509
Durham, L. 1955. Effects of predation by cormorants and gars on fish populations of ponds in Illinois. Dissertation. University of Illinois Urbana-Champaign, Urbana, Illinois, USA.
eBird. 2018. eBird: an online database of bird distribution and abundance. eBird, Cornell Lab of Ornithology, Ithaca, New York, USA. https://ebird.org/home
Environmental Systems Research Institute (ESRI). 2018. ArcGis, Version 10.4.1. Environmental Systems Research Institute, Redlands, California, USA.
Farquhar, J. F., I. M. Mazzocchi, R. D. McCullough, R. B. Chipman, and T. L. DeVault. 2012. Mitigation of Double-crested Cormorant impacts on Lake Ontario: from planning and practice to product delivery. Waterbirds 35 (Special Publication 1):56–65. https://doi.org/10.1675/063.035.sp107
Foster, M. S., and L. A. Fitzgerald. 1982. A technique for live-trapping cormorants. Journal of Field Ornithology 53:422-423.
Glahn, J. F., P. J. Dixson, G. A. Littauer, and R. B. McCoy. 1995. Food habits of Double crested-Cormorants wintering in the Delta Region of Mississippi. Colonial Waterbirds 18 (Special Publication 1):158-167. https://doi.org/10.2307/1521536
Glahn, J. F., J. B. Harrel, and C. Vyles. 1998. The diet of wintering Double-crested Cormorants feeding at lakes in the southeastern United States. Colonial Waterbirds 21:445-452. https://doi.org/10.2307/1521657
Grémillet, D., M. Enstipp, M. Boudiffa, and H. Liu. 2006. Do cormorants injure fish without eating them? An underwater video study. Marine Biology 148:1081-1087. https://doi.org/10.1007/s00227-005-0130-2
Harrison, X. A. 2014. Using observation-level random effects to model overdispersion in count data in ecology and evolution. PeerJ e616.https://doi.org/10.7717/peerj.616
Hebert, C. E., J. Pasher, D. V. C. Weseloh, T. Dobbie, S. Dobbyn, D. Moore, V. Minelga, and J. Duffe. 2014. Nesting cormorants and temporal changes in island habitat. Journal of Wildlife Management 78:307-313. https://doi.org/10.1002/jwmg.659
Hirsch, K. 1986. Colony expansion and food habits of Double-crested Cormorants in Minnesota. Pacific Seabird Group Bulletin 13:32-33.
Hobson, K. A., R. W. Knapton, and W. Lysack. 1989. Population, diet and reproductive success of Double-crested Cormorants breeding on Lake Winnipegosis, Manitoba, in 1987. Colonial Waterbirds 12:191-197. https://doi.org/10.2307/1521340
Jackson C. D., and G. E. Brown. 2011. Differences in antipredator behaviour between wild and hatchery-reared juvenile Atlantic salmon (Salmo salar) under seminatural conditions. Canadian Journal of Fisheries and Aquatic Sciences 68:2157-2165. https://doi.org/10.1139/f2011-129
King, D. T., K. J. Andrews, J. O. King, R. D. Flynt, J. F. Glahn, and J. L. Cummings. 1994. A night-lighting technique for capturing cormorants. Journal of Field Ornithology 65:254-257. https://sora.unm.edu/sites/default/files/journals/jfo/v065n02/p0254-p0257.pdf
Klimaszyk, P., and P. Rzymski. 2016. The complexity of ecological impacts induced by Great Cormorants. Hydrobiologia 771:13-30. https://doi.org/10.1007/s10750-015-2618-1
Koh, S., A. J. Tanentzap, G. Mouland, T. Dobbie, L. Carr, J. Keitel, K. Hogsden, G. Harvey, J. Hudson, and R. Thorndyke. 2012. Double-crested Cormorants alter forest structure and increase damage indices of individual trees on island habitats in Lake Erie. Waterbirds 35 (Special Publication 1):13-22. https://doi.org/10.1675/063.035.sp103
Lafferty, D., K. C. Hanson-Dorr, A. M. Prisock, and B. S. Dorr. 2016. Biotic and abiotic impacts of Double-crested Cormorant breeding colonies on forested islands in the southeastern United States. Forest Ecology and Management 369:10-19. https://doi.org/10.1016/j.foreco.2016.03.026
Lindén, A., and S. Mäntyniemi. 2011. Using the negative binomial distribution to model overdispersion in ecological count data. Ecology 92:1414-1421. https://doi.org/10.1890/10-1831.1
Lyons, D. E., D. D. Roby, and K. Collis. 2007. Foraging patterns of Caspian Terns and Double-crested Cormorants in the Columbia River Estuary. Northwest Science 81:91-103. https://doi.org/10.3955/0029-344X-81.2.91
McBay, L. G. 1961. The biology of Tilapia nilotica Linnaeus. Pages 208-218 in Proceedings of the 15th Annual conference Southeastern Association of Game and Fish Commissioners. Southeastern Association of Game and Fish Commissioners, Frankfurt, Kentucky, USA.
Miranda, L. E., and W. D. Hubbard. 1994. Winter survival of age-0 largemouth bass relative to size, predators, and shelter. North American Journal of Fisheries Management 14:790-796.
Morrison, M. L. 1977. Life history and status of the Olivaceous Cormorant. Thesis. Texas A&M University, College Station, Texas, USA.
Morrison, M. L., and R. D. Slack. 1977. The role of flock feeding in Olivaceous Cormorants. Bird-Banding 48:277-279.
National Audubon Society. 2010. The Christmas Bird Count historical results. National Audubon Society, New York, New York, USA. https://netapp.audubon.org/cbcobservation/
Opačak, A., T. Florijančić, D. Horvat, S. Ozimec, and D. Bodakoš. 2004. Diet spectrum of Great Cormorants (Phalacrocorax carbo sinensis L.) at the Donji Miholjac carp fishponds in eastern Croatia. European Journal of Wildlife Research 50:173–178. https://doi.org/10.1007/s10344-004-0059-7
Pacific Flyway Council. 2012. Pacific Flyway plan: a framework for the management of Double-crested Cormorant depredation on fish resources in the Pacific Flyway. Pacific Flyway Council, U.S. Fish and Wildlife Service, Portland, Oregon, USA. https://pacificflyway.gov/Documents/Dcc_plan.pdf
Portnoy, J. W. 1977. Nesting colonies of seabirds and wading birds, coastal Louisiana, Mississippi, and Alabama. FWS/OBS-77/07. U.S. Fish and Wildlife Service, Biological Services Program, Washington, D.C., USA.
Riedel, R., L. M. Caskey, and S. H. Hurlbert. 2007. Length-weight relations and growth rates of dominant fishes of the Salton Sea: implications for predation by fish-eating birds. Lake and Reservoir Management 23:528-535. https://doi.org/10.1080/07438140709354036
Rudstam, L. G., A. J. VanDeValk, C. M. Adams, J. T. H. Coleman, J. L. Forney, and M. E. Richmond. 2004. Cormorant predation and the population dynamics of walleye and yellow perch in Oneida Lake. Ecological Applications 14:149-163. https://doi.org/10.1890/03-5010
Russell, I., B. Broughton, T. Keller, and D. Carss. 2012. The INTERCAFE cormorant management toolbox. Methods for reducing cormorant problems at European fisheries. Final Report. European Cooperation in Science and Technology (COST), Brussels, Belgium. http://www.intercafeproject.net/pdf/Cormorant_Toolbox_web_version.pdf
Russell, I., D. Parrott, M. Ives, D. Goldsmith, S. Fox, D. Clifton-Dey, A. Prickett, and T. Drew. 2008. Reducing fish losses to cormorants using artificial fish refuges: an experimental study. Fisheries Management and Ecology 15:189-198. https://doi.org/10.1111/j.1365-2400.2008.00600.x
Schemnitz, S. D. 1996. Capturing and handling wild animals. Pages 106-124 in T. A. Bookhout, editor. Research and management techniques for wildlife and habitats. The Wildlife Society, Bethesda, Maryland, USA.
Schneider, D. C., and D. C. Duffy. 1985. Scale-dependent variability in seabird abundance. Marine Ecology Progress Series 25:211-218. https://doi.org/10.3354/meps025211
Schramm, H. L., Jr., B. French, and M. Ednoff. 1984. Depredation of channel catfish by Florida Double-crested Cormorants. Progressive Fish-Culturist 46:41-43.
Schultz, D. W., A. J. Carlson, S. Mortensen, and D. L. Pereira. 2013. Modeling population dynamics and fish consumption of a managed Double-crested Cormorant colony in Minnesota. North American Journal of Fisheries Management 33:1283-1300. https://doi.org/10.1080/02755947.2013.839972
Stahr, K. J., and M. A. Kaemingk. 2017. An evaluation of emergent macrophytes and use among groups of aquatic taxa. Lake and Reservoir Management 33: 314-323. https://doi.org/10.1080/10402381.2017.1339747
Stahr, K. J., and D. E. Shoup. 2015. American water willow mediates survival and antipredator behavior of juvenile largemouth bass. Transactions of the American Fisheries Society 144:903-910.
Stevenson, M. M., and G. H. Rosenberg. 2004. Arizona region (winter season). North American Birds 58:265-267.
Stickley, Jr., A. R. 1991. Cormorant feeding rates on commercially grown catfish. Aquaculture Magazine 17:89-90.
Stickley, Jr., A. R., G. L. Warrick, and J. F. Glahn. 1992. Impact of Double-crested Cormorant depredations on channel catfish farms. Journal of the World Aquaculture Society 23:192-198. https://doi.org/10.1111/j.1749-7345.1992.tb00768.x
Telfair, II, R. C., and M. L. Morrison. 2020. Neotropic Cormorant (Phalacrocorax brasilianus). Version 2.0. In P. G. Rodewald and B. K. Keeney, editors. Birds of the World. Cornell Lab of Ornithology, Ithaca, New York, USA. https://doi.org/10.2173/bow.neocor.02
U.S. Fish and Wildlife Service. 2003. Final environmental impact statement: Double-crested Cormorant management in the United States. U.S. Department of the Interior, Division of Migratory Bird Management, Arlington, Virginia, USA. https://downloads.regulations.gov/FWS-HQ-MB-2019-0103-1409/content.pdf
U.S. Fish and Wildlife Service. 2013. General provisions. Revised list of migratory birds, final rule. Federal Register 78(212):65844-65864.
U.S. Fish and Wildlife Service. 2017. Environmental assessment for issuing depredation permits for Double-crested Cormorant management. U.S. Fish and Wildlife Service, Division of Migratory Bird Management, Falls Church, Virginia, USA. https://s3.amazonaws.com/bncore/wp-content/uploads/2017/11/CormorantEA.pdf
U.S. Fish and Wildlife Service. 2020a. Final environmental impact statement: management of conflicts associated with Double-crested Cormorants. U.S. Department of the Interior, Division of Migratory Bird Management, Falls Church, Virginia, USA. https://downloads.regulations.gov/FWS-HQ-MB-2019-0103-1409/content.pdf
U.S. Fish and Wildlife Service. 2020b. Title 50 code of federal regulations (CFR), part 21: current and historical CFR for migratory bird permits. U.S. Fish and Wildlife Service, Division of Migratory Bird Management, Falls Church, Virginia, USA. https://www.govinfo.gov/app/collection/cfr/2006/title50
van Eerden, M. R., S. van Rijn, S. Volponi, J.-Y. Paquet, and D. Carss. 2012. Cormorants and the European environment: exploring Cormorant ecology on a continental scale. Final Report. European Cooperation in Science and Technology (COST), Brussels, Belgium. https://ec.europa.eu/environment/nature/cormorants/files/Cormorants_and_Environment_INTERCAFE.pdf
Warton, D. I., M. Lyons, J. Stoklosa, and A. R. Ives. 2016. Three points to consider when choosing a LM or GLM test for count data. Methods in Ecology and Evolution 7:882-890. https://doi.org/10.1111/2041-210X.12552
Weseloh, D. V., P. J. Ewing, J. Struger, P. Mineau, C. A. Bishop, S. Postupalsky, and J. P. Ludwig. 1995. Double-crested Cormorants of the Great Lakes: changes in population size, breeding distribution and reproductive output between 1913 and 1991. Colonial Waterbirds 18 (Special Publication 1):48-59. https://doi.org/10.2307/1521523
Wires, L. R., F. J. Cuthbert, D. R. Trexel, and A. R. Joshi. 2001. Status of the Double-crested Cormorant (Phalacrocorax auritus) in North America. Final report to the U.S. Fish and Wildlife Service, Fort Snelling, Minnesota, USA. https://digitalcommons.unl.edu/usfwspubs/400/
White, G. C., and R. E. Bennetts. 1996. Analysis of frequency count data using the negative binomial distribution. Ecology 77:2549-2557. https://doi.org/10.2307/2265753
Fig. 1
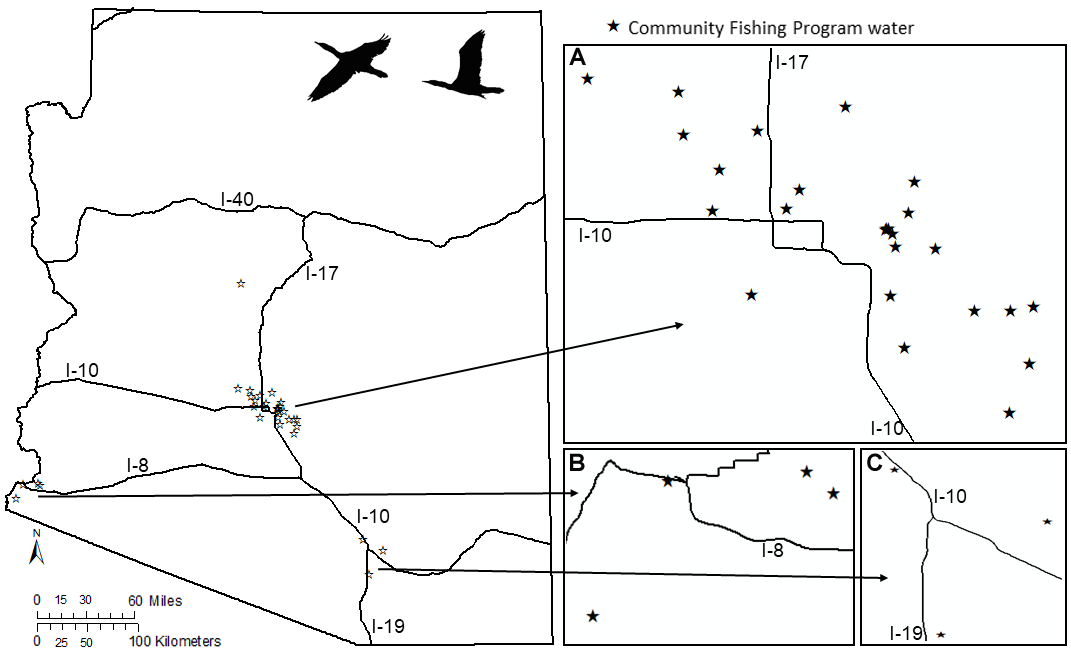
Fig. 1. Community fishing program (CFP) waters in metropolitan centers in Arizona sampled in this study. Major interstates are shown for reference. A) CFP waters around Phoenix metropolitan area, B) CFP waters near Yuma, AZ, and C) CFP waters near Tucson, AZ.

Fig. 2
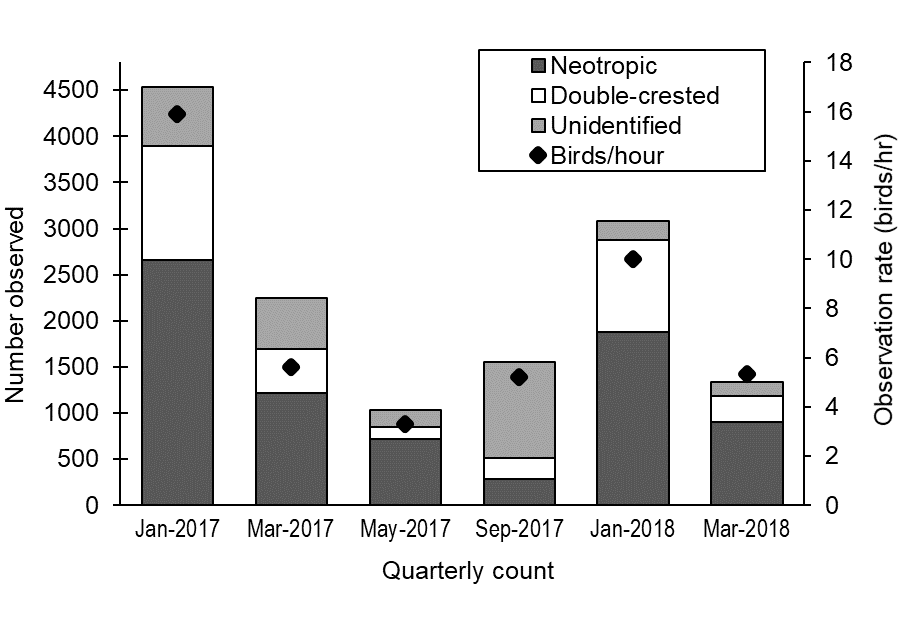
Fig. 2. Number of cormorants observed and observation rate (birds/hour) for seasonal counts in Arizona, USA, January 2017-March 2018.

Fig. 3
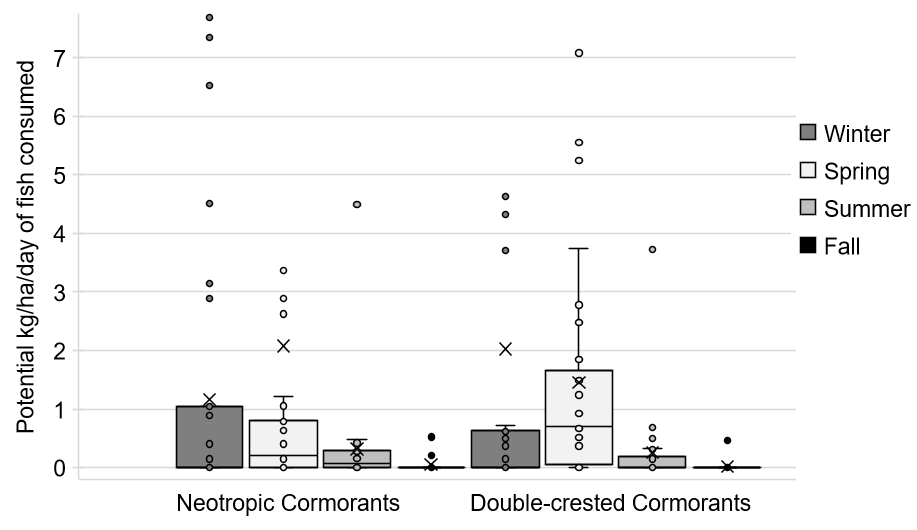
Fig. 3. Estimated potential fish consumed (kg/ha/day) by cormorants in Arizona Community Fishing Program waters during each sampling season, January 2017-March 2018. For outliers above 10 kg/ha/day, see Appendix 1, Table A1.1. Estimates calculated using maximum number of birds observed on a single day during each quarterly count at each surveyed community fishing water and published feeding rates for each cormorant species. ‘x’ denotes a mean value each season in the estimates for potential fish losses.

Table 1
Table 1. Count of fish in size and shape classes successfully swallowed (attempted, but not successfully swallowed) by adult, juvenile, and unknown age cormorants at all observed waters in Arizona, USA, 2017-2018 (n = 52 observations of Double-crested Cormorants, Nannopterum auritum, n = 1970 observations of Neotropic Cormorants, N. brasilianum). Fish size was estimated relative to the bill length of the cormorant holding it.
| Adult Cormorants | Juvenile Cormorants | Unknown Age Cormorants | ||||||||
| Fish Size | Elongate | Round | Unknown | Elongate | Round | Unknown | Elongate | Round | Unknown | Total |
| Double-crested Cormorants | ||||||||||
| ≤ 10 cm | 0 (2) | 21 | 2 | 1 (1) | 1 | 8 | 1 | 0 | 0 | 34 (3) |
| 15 cm | 0 | 0 | 1 | 0 | 0 | 2 | 0 | 0 | 0 | 3 |
| 20 cm | 2 (1) | 2 | 0 | 0 (1) | 2 | 0 | 0 | 0 | 0 | 6 (2) |
| 25 cm | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| ≥ 30 cm |
0 (1) | 1 | 1 | 0 | 0 (1) | 0 | 0 | 0 | 0 | 2 (2) |
| Neotropic Cormorants | ||||||||||
| ≤ 8 cm | 219 | 855 (1) | 336 (2) | 67 | 78 | 117 (4) | 18 | 24 | 96 (1) | 1810 (8) |
| 12 cm | 2 | 35 (1) | 5 | 1 | 0 | 1 | 0 | 2 (2) | 1 | 47 (3) |
| 16 cm | 7 | 51 (3) | 9 | 4 | 2 | 3 | 1 | 2 (4) | 2 | 81 (7) |
| 20 cm | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 |
| ≥ 24 cm | 1 (1) | 6 (2) | 0 | 0 (1) | 0 | 0 | 0 | 0 | 0 (1) | 7 (5) |
Table 2
Table 2. Average fish length in cm (sample size) of fish removed from stomachs of lethally sampled Double-crested Cormorants (Nannopterum auritum) in large rivers or reservoirs in Arizona, USA, January-March 2019. Fish were classified as being ‘elongate’ in body shape; some measurements (~ half) represent a minimum length if fish were partial but could still be estimated in length and identified. Note: BWNWR = Bill Williams National Wildlife Refuge (NWR), COR = Colorado River.
Location |
CENT† |
LEMA‡ |
LEPO§ |
MISA| |
ICPU¶ |
PIPR# |
DOCE†† |
DOPE‡‡ |
TILA§§ |
Unk|| Bass |
Unk Shad | Unk Fish |
| Apache Lake | 10.5(1) | 5.4(6) | 5.8(16) | |||||||||
| BWNWR | 17.0(1) | 16.0(1) | ||||||||||
| Catfish Paradise | 15.3(2) | 17.5(1) | 6.6(1) | 4.5(1) | ||||||||
| COR south | 32.5(2) | |||||||||||
| Havasu NWR | 13.0(1) | 15.0(1) | 19.0(1) | 29.0(1) | 19.0(1) | 11.5(1) | ||||||
| Hidden Lake | 8.6(3) | 7.5(20) | 7.9(26) | 6.0(3) | ||||||||
| Martinez Lake | 17.0(1) | 14.5(2) | 14.0(1) | 21(1) | 6.0(1) | 8.3(2) | ||||||
| Patagonia Lake | 27.0(1) | |||||||||||
| Robbins Butte | 10.2(3) | 9.8(3) | 12.5(1) | 8.9(20) | 4.0(1) | |||||||
| Roosevelt Lake | 18.0(1) | 12.3(2) | 27.0(1) | 14.0(1) | 8.0(1) | |||||||
| Mean fish length | 15.5(2) | 16.0(2) | 14.3(7) | 27.4(5) | 17.9(4) | 17.5(1) | 9.4(6) | 7.3(29) | 16.8(2) | 13.2(3) | 7.6(63) | 6.9(9) |
| †CENT: Centrarchid species, ‡LEMA: Lepomis macrochirus (bluegill), §LEPO: Lepomis species, |MISA: Micropterus salmoides (largemouth bass), ¶ICPU: Ictalurus punctatus (channel catfish), #PIPR: Pimephales promelas (fathead minnow), ††DOCE: Dorosoma cepedianum (gizzard shad), ‡‡DOPE: D. petenense (threadfin shad), §§TILA: Tilapia species, ||Unk: unknown | ||||||||||||
Table 3
Table 3. Average fish length in cm (sample size) of fish removed from stomachs of lethally sampled Neotropic Cormorants (Nannopterum brasilianum) in large rivers or reservoirs in Arizona, USA, January-March 2019. Fish were classified as being ‘elongate’ in body shape; no fish specimens collected from Neotropic Cormorants were ‘rounded.’ Some measurements represent minimum length if fish were partial but could still be estimated in length and identified.
| Location | CENT† | LEPO‡ | DOCE§ | DOPE| | TILA¶ | Unk shad# |
| Apache Lake | 12.0(1) | |||||
| Hidden Lake | 11.0(3) | 5.0(2) | 12.0(1) | 7.2(30) | ||
| Patagonia Lake | 13.0(1) | |||||
| Robbins Butte | 8.6(7) | 9.2(3) | 9.0(20) | |||
| Mean fish length | 8.6(7) | 12.0(1) | 11.0(3) | 7.5(5) | 12.0(1) | 8.0(51) |
| †CENT: Centrarchid species, ‡LEPO: Lepomis species, §DOCE: Dorosoma cepedianum (gizzard shad), |DOPE: D. petenense (threadfin shad), ¶TILA: Tilapia species, #Unk: unknown | ||||||
Table 4
Table 4. Top five generalized linear models estimating cormorant numbers as a function of environmental covariates found at community fishing ponds in Arizona, USA, 2017-2018. Note: df = degrees of freedom.
| Model | df | AICc | ΔAIC | Weight |
| Artificial features + trees > 2 m | 4 | 254.24 | 0.00 | 0.35 |
| Artificial features + natural structures + trees > 2 m | 5 | 256.10 | 1.86 | 0.14 |
| Artificial features + trees > 2 m + distance to water | 5 | 256.49 | 2.25 | 0.11 |
| Artificial features + time since last stocking + trees > 2 m | 5 | 256.90 | 2.66 | 0.09 |
| Artificial features + depth + trees > 2 m | 5 | 256.93 | 2.68 | 0.09 |









