The following is the established format for referencing this article:
Lindsay, A. M., M. M. Jacobs, H. M. Streby, and A. E. Wiley. 2026. Refining methods to add stable isotope analysis of migratory bird fat to the ornithological toolbox. Journal of Field Ornithology 97(2):4.ABSTRACT
Prior to migration, songbirds increase food consumption to deposit subcutaneous fat that helps fuel migratory flights, and many periodically pause at stopover sites to rest and refuel for the next flight. We conducted a three-phase, proof-of-concept study to move toward a standardized approach to collecting and preparing fat samples from wild, migrating songbirds, and to explore the feasibility of using stable-isotope analysis on fat tissue to estimate previous stopover locations. First, using fat obtained from a store-bought chicken breast, we conducted an experiment to test whether lipid purification method affects percent lipid yield and stable hydrogen (δ2H) and stable carbon (δ13C) isotope values. We found that purified lipids had significantly higher δ2H values and lower δ13C values than whole fat samples indicating the need for consistency in sample preparation. Second, to test for repeatability in δ2H and δ13C values in purified lipids obtained from wild songbirds, we held Swainson’s Thrushes (Catharus ustulatus; n = 9) in temporary captivity provided with the same diet and local water, and found remarkable consistency in both δ2H (SD = 2.6‰) and δ13C (SD = 0.3‰) values between individuals. Third, we sampled fat from seven species (Magnolia Warbler [Setophaga magnolia], Swainson’s Thrush, Hermit Thrush [Catharus guttatus], Wood Thrush [Hylocichla mustelina], Gray Catbird [Dumetella carolinensis], Red-eyed Vireo [Vireo olivaceus], Northern Waterthrush [Parkesia noveboracensis]) of wild, migrating songbirds at Powdermill Nature Reserve, Pennsylvania, USA as a preliminary investigation into the usefulness of stable-isotope analysis in estimating geographic location of fat assimilation. Among five species assumed to have deposited fat at distant latitudes during spring migration, the three thrush species had δ2H values consistent with latitudes farther south relative to Magnolia Warblers and Gray Catbirds. However, in the two species (Red-eyed Vireo and Northern Waterthrush) assumed to have assimilated fat locally, there was significant variation in δ2H values between the species, and δ2H values did not fit into the pattern observed in distant-origin birds. Although geographic assignment of fat assimilation site in migrating songbirds is more complicated than we expected, stable-isotope analysis of fat is a promising method with potential applications in stopover ecology, diet, and habitat use.
RESUMEN
Previo a la migración, las aves paseriformes incrementan su consumo de alimento para depositar grasa subcutánea que utilizan como combustible para los vuelos migratorios, y muchas realizan paradas periódicas en sitios de escala migratoria (stopover) para descansar y reabastecerse para el siguiente vuelo. Realizamos un diseño en tres fases, a modo de prueba de concepto, con el objetivo de avanzar hacia un enfoque estandarizado para la recolección y preparación de muestras de grasa en aves silvestres migratorias, y para explorar la factibilidad del uso del análisis de isótopos estables en tejido adiposo para estimar sitios previos de escala migratoria. Primero, utilizando grasa obtenida de una pechuga de pollo comercial, realizamos un experimento para evaluar si el método de purificación lipídica afecta el porcentaje de rendimiento de lípidos y los valores isotópicos de hidrógeno estable (δ²H) y carbono estable (δ¹³C). Encontramos que los lípidos purificados tuvieron valores significativamente más altos de δ²H y más bajos de δ¹³C que las muestras de grasa completa, lo que indica la necesidad de consistencia en la preparación de muestras. En segundo lugar, para evaluar la repetibilidad de los valores de δ²H y δ¹³C en lípidos purificados provenientes de aves silvestres, mantuvimos individuos de Catharus ustulatus (n = 9) en cautiverio temporal, suministrándoles la misma dieta y agua local. Observamos una notable consistencia entre individuos tanto en δ²H (DE = 2,6%) como en δ¹³C (DE = 0,3%). En tercer lugar, muestreamos grasa de siete especies de aves migratorias silvestres (Setophaga magnolia, Catharus ustulatus, Catharus guttatus, Hylocichla mustelina, Dumetella carolinensis, Vireo olivaceus y Parkesia noveboracensis) en Powdermill Nature Reserve, Pennsylvania, EE. UU., como una evaluación preliminar de la utilidad del análisis isotópico para estimar la ubicación geográfica de la asimilación de grasa. Entre cinco especies que se presume depositaron grasa en latitudes distantes durante la migración primaveral, las tres especies de zorzales (C. ustulatus, C. guttatus e Hylocichla mustelina) mostraron valores de δ²H consistentes con latitudes más australes en comparación con Setophaga magnolia y Dumetella carolinensis. Sin embargo, en las dos especies que se presume asimilaron grasa localmente (Vireo olivaceus y Parkesia noveboracensis), se observó una variación significativa en los valores de δ²H entre especies, y dichos valores no se ajustaron al patrón observado en aves de origen distante. Si bien la asignación geográfica del sitio de asimilación de grasa en aves migratorias resulta más compleja de lo esperado, el análisis de isótopos estables en tejido adiposo se presenta como una metodología prometedora con potenciales aplicaciones en la ecología de sitios de escala migratoria, la dieta y el uso de hábitat.
INTRODUCTION
Every spring and fall, billions of birds migrate through eastern North America. Prior to a migratory flight, songbirds enter a period of hyperphagia wherein they increase their food consumption and deposit fat in the furcular hollow, abdomen, and flanks (Blem 1976, Ralph et al. 1993). The lipid stored in this fat is one of the primary nutrients that provides energy to fuel migration (Guglielmo 2018), which can consist of long (e.g., > 14 hours over the Gulf of Mexico [Deppe et al. 2015], > 72 hours over the Atlantic Ocean [DeLuca et al. 2015, McKinnon et al. 2017]) and relatively short flights (e.g., a few hours between stopover sites [Delmore et al. 2012, Callo et al. 2013, Cooper et al. 2017, Stanley et al. 2021]). Most migratory birds stop periodically during migration to replenish fat stores before continuing migration (Alerstam 2011, Gómez et al. 2017). The distance birds can fly between stopover sites likely varies between species, with some able to travel much greater distances between stopover events than others. For example, a study using radio-tracking technology suggested that some Gray-cheeked Thrushes (Catharus minimus) might travel from Central America to the Great Lakes region in a single flight (Gómez et al. 2017) and a study on physiological condition of spring migrating birds stopping over on the northern Gulf Coast suggest that some songbirds arrive with fat reserves sufficient to continue for considerable distances (Moore and Kerlinger 1987). During fall migration, Blackpoll Warblers (Setophaga striata) and Connecticut Warblers (Oporornis agilis) make non-stop, over-ocean flights from coastal northeast United States to northern South America, a journey lasting up to, and often exceeding, 72 hours (DeLuca et al. 2015, McKinnon et al. 2017, DeLuca et al. 2019). Other species stop more frequently to refuel between flights, with relatively shorter distances between stopover locations. A study using light-level geolocators to determine the migratory pathways of Kirtland’s Warblers (Setophaga kirtlandii) revealed several stopover locations during both spring and fall migration on the species’ route between the breeding ground in Michigan, USA and the wintering ground in central Bahamas (Cooper et al. 2017) and Red-eyed Vireos (Vireo olivaceus) migrating between central South America and northwestern Pennsylvania, USA similarly make frequent stopovers (Callo et al. 2013). Similarly, a study using GPS tags to track Wood Thrushes (Hylocichla mustelina) throughout the annual cycle showed several stopover events during both spring and fall migration seasons (Stanley et al. 2021). The goal of migration is to safely and efficiently move between breeding grounds and stationary non-breeding grounds, and although birds are able to adapt to immediate danger of mortality or future fitness costs (Schmaljohann et al. 2022), there are species-specific differences in how far birds travel between stopover locations.
Stable carbon and hydrogen isotope analysis of bird tissues have enabled tracking movements, feeding origins, diet, and habitat use of migratory songbirds without marking birds with tracking devices or requiring multiple captures of the same individual (Rubenstein and Hobson 2004). The relative coarseness of locations estimated using stable isotopes compared to deployed tracking devices is traded off with the reduced sampling cost and elimination of reliance on return rates and capturing specific individuals (Vander Zanden et al. 2018). These isotopic analyses are possible because isotope ratios vary naturally among habitat types and geographic locations, and because isotope ratios of the local environment are incorporated into growing bird tissue with predictable offsets. Stable carbon isotope ratios of 13C to 12C (reported as δ13C values) vary among primary producers: between C3, C4, and CAM photosynthetic pathways (Marra et al. 1998, Marshall et al. 2007), between terrestrial vs. aquatic habitats (MacDade et al. 2011), and between mesic (lower δ13C values) and xeric (higher δ13C values) habitats (Marra et al. 1998, Marshall et al. 2007). Thus, δ13C values in animal tissue reflect local vegetation types, diet, and habitat quality and usage (Marra et al. 1998, Hobson et al. 2004a, Contina et al. 2022). Stable hydrogen isotope ratios in bird tissues often reflect 2H to 1H ratios (reported as δ2H values) in local precipitation, which varies roughly latitudinally, with more negative δ2H values at increasingly northern latitudes (e.g., Terzer et al. 2013). Geographic origins of bird tissues can be inferred by comparing δ2H values in tissue to the existing isoscape, which is a tissue-specific map of predicted stable-isotope values (Hobson and Wassenaar 1996, Wassenaar and Hobson 2001, Hobson 2002, Kelly et al. 2002, Clegg et al. 2003, Zenzal et al. 2018). Stable carbon isotope values in tissues are derived from diet, whereas stable hydrogen values are assimilated from both drinking water and diet, so hydrogen isotope values in consumer tissue reflect the values of these two sources. Additionally, hydrogen isotope ratios follow a latitudinal and altitudinal gradient but do not follow a longitudinal pattern, so geographic tissue origin assignment is not precise.
Isotope ratios are assimilated into tissue as it grows and are useful from both metabolically active and inactive tissues. Ratios are fixed in inert tissue such as feathers and claws but are continuously turning over in metabolically active tissue such as blood, muscle, bone, and breath (Marra et al. 1998, MacDade et al. 2011). Isotope ratios from different tissues provide data over distinct time periods, variation that can be exploited to answer ecological questions at different time scales. For example, stable-isotope values of inert feathers reflect the isotope ratio of the area in which they were grown regardless of when they were sampled, whereas isotope values of blood reflects the ratio of the area a bird occupied for a few days to weeks prior to sampling (Barquete et al. 2013). Because fat primarily consists of carbon and hydrogen, and carbon-bound hydrogen does not easily exchange with water vapor or body water (Anparasan and Hobson 2021), fat tissue is relatively inert once it has been deposited (Schimmelmann 1991, Hobson and Wassenaar 2019, Anparasan and Hobson 2021). Therefore, fat tissue should reflect the isotope ratios of the habitat and geographic region in which a bird refueled during a migratory stopover event. Consequently, fat tissue may be useful in estimating distances individuals have flown, and can fly, between stopover sites. This information can be used to estimate averages and ranges of migratory flight distances and help prioritize targeted conservation of migratory stopover habitat, that is, ensuring that there are plenty of stopover locations so that migrating birds can stop frequently enough to support refueling. Additionally, this method could be used to compare species-specific distances between stopover locations, and to study stopover habitat conditions and diet composition.
Few studies have attempted stable-isotope analysis on bird fat tissue, and none has obtained fat from live songbirds in the field, although some have safely sampled fat from live captive (Rocha et al. 2016, Viegas et al. 2017, Araújo et al. 2019, McWilliams et al. 2020, Morrissey and Swekla 2023) and wild (Owen et al. 2010, dos Santos et al. 2023) birds for other research questions. Thus far, stable-isotope studies have relied on fat samples taken from birds sacrificed in the field or after controlled laboratory experiments (Hobson et al. 2004b, Anparasan and Hobson 2021). Additionally, these studies used different methods to pretreat samples for isotope analysis. Hobson et al. (2004b) freeze dried fat samples but Anparasan and Hobson (2021) did not. Both extracted lipids using a 2:1 chloroform:methanol soak. Because each step of the sample preparation process could potentially affect isotopic ratios, consistency in methods between studies is necessary for comparability and replicability. In order for stable-isotope analysis of migratory bird fat to be a useful addition to the ornithological toolbox, it will be necessary to standardize methods of collecting, handling, and analyzing fat samples.
Here, we conducted a three-phase, proof-of-concept study to build upon previous work and move toward a standardized approach to collect, prepare, and analyze stable isotopes of fat samples from free-living birds. First, to determine the minimum amount of fat necessary to yield 0.5mg (for stable carbon isotope analysis) or 0.35mg (for stable hydrogen isotope analysis) of purified lipids, and to investigate how sample preparation method (i.e., freeze-drying, lipid purification) affects lipid yield and hydrogen and carbon isotope values, we performed a suite of sample preparation treatments using fat collected from the breast of a store-bought chicken (Gallus gallus domesticus). We predicted that freeze drying samples would affect δ2H values because the process removes water, and therefore hydrogen, from the samples. Further, we predicted that performing lipid extraction would affect both δ2H and δ13C values because the process separates extraneous tissues (e.g., connective tissue, blood) that could affect isotope values (Post et al. 2007). Second, to assess repeatability of isotope measurements among individuals with identical diet and drinking water, we held nine wild Swainson’s Thrushes (Catharus ustulatus) in short-term captivity in an outdoor aviary with controlled diet. The thrushes lacked visible fat stores upon initial capture and were held until they deposited sufficient fat reserves for sampling. We predicted that isotope values of fat samples from these birds would be very similar. Last, as a first pass at determining if isotope values differ among birds that we expect to have recently migrated to our study site, we sampled fat from seven species of wild migrating songbirds at Powdermill Nature Reserve’s avian research center in Pennsylvania, USA. Our target species assimilated fat both locally (i.e., sampling recaptured birds in fall that had no fat upon first capture) and at more southern latitudes prior to arrival at our site (i.e., sampling birds in spring with low fat reserves caught the morning after a night of strong migratory movement). We predicted that stable hydrogen isotope ratios would vary predictably with presumed fat assimilation latitude, which for this study was simply local latitude vs. any latitude south of our study site.
METHODS
Testing for sample preparation effects
To determine whether lipid yield and isotope ratios are affected by the size of the fat sample or various treatments used to process the sample (Table 1), we conducted a suite of tests using fat collected from a store-bought chicken breast. We do not know where the chicken was raised or what constituted its diet, neither of which was relevant to our within-organism comparative questions about sample preparation. We varied the size of fat samples from 3 to 30 mg to test the minimum size required to isolate an adequate mass of lipids for stable-isotope analysis (i.e., 0.5 mg and 0.35 mg of purified lipids for stable carbon and stable hydrogen analysis, respectively). We collected these samples (n = 28) in one sitting and immediately stored them in small, capped glass vials at -80 °C.
Of the 28 chicken fat samples, we freeze dried 22 overnight in a Labconco Freezone 4.5 L, -84 °C benchtop freeze dryer and stored the remaining six samples (i.e., the non-freeze-dried treatment) in a -80 °C freezer until the lipid purification process. After thawing, we macerated all samples against the side of their individual glass vial using a sterile metal spatula and we added 2–3 mL (i.e., enough to completely submerge the sample) of 2:1 chloroform:methanol solution to each vial. We capped the vials and allowed the samples to soak for 24–48 hours, and vortexed some (n = 4) at the halfway point, to allow the lipids to enter solution.
To remove connective tissue and other impurities, we filtered a subset of lipid solutions (for those receiving this treatment; n = 22; see Table 1) using Whatman GF/F glass filter paper shaped into a cone and placed in the bottom of a glass syringe. We pipetted the solution from each glass vial into the glass syringe and allowed it to filter via gravity into a clean glass vial for evaporation. We then rinsed each vial with ~0.5 mL clean 2:1 chloroform:methanol solution to ensure maximum volume of lipids in the solution. We then evaporated the filtered lipid solution in a fume hood at room temperature for 24 hours. The purified lipids remained as a viscous gel residue in the glass evaporation vials.
We loaded the purified lipid samples into tin (0.5 mg for δ13C) or silver (0.35 mg for δ2H) capsules for stable-isotope analysis at the University of Akron’s Stable Isotope Facility, Akron, Ohio. For both δ13C and δ2H, samples were analyzed on a Vario PYRO Cube elemental analyzer (Elementar Americas) coupled to an Isoprime100 stable isotope ratio mass spectrometer (Isoprime). All lipid samples were calibrated relative to standard reference materials with known δ2H values (USGS70: δ2H = -183.9 ± 1.4‰, and NBS 22: δ2H = -117.2 ± 0.6‰) or δ13C values (USGS40: δ13C = -26.39 ± 0.04‰ and USGS41a: δ13C = 36.55 ± 0.08‰). To minimize instrument memory effect on δ2H values, the first sample analyzed following a set of standards (USGS70 and NBS 22) was run in triplicate and only the third δ2H value was included in our dataset. The analytical precision of δ2H and δ13C values was within ±1.5‰ and 0.1‰, respectively. Isotope values are reported in per mil notation (‰), per the equation
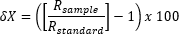
|
(1) |
where X is 13C or 2H, and R is 13C/12C or 2H/1H, respectively. Rstandard is Vienna Peedee Belemnite (V-PDB) for δ13C and Vienna Standard Mean Ocean Water (V-SMOW) for δ2H.
Field tests
Biopsy procedure
We sampled fat from wild birds at Powdermill Nature Reserve in Rector, Pennsylvania, Westmoreland County, USA (40° 9’46” N, 79° 16’11” W; hereafter Powdermill) during the spring and fall 2020 and spring 2021 migration seasons. Powdermill operates an established, long-term bird banding station in an area dominated by transitional, marsh, old field, shrub, and forest edge habitat types. We opened 12 m mist nets (n = 63–67 per day) 30 minutes before sunrise and operated them for six hours, six days per week as part of long-term netting activities. After normal banding procedures, we either placed individuals into temporary captivity (see below) or biopsied focal species for furcular fat tissue using a modification of the method outlined in Rocha et al. (2016). We restrained each bird gently but firmly and placed a small, soft cloth over the head. We cleaned and disinfected the breast using ethyl alcohol then applied a local anesthetic cream (EMLA cream, 2.5% lidocaine and 2.5% prilocaine; Gourdon 2017). We placed each bird in a clean cloth bag for 5–10 minutes, then tested (i.e., tapped gently with forceps on the anesthetized area) each for anesthetic effectiveness prior to the biopsy to confirm lack of apparent pain response by the bird. We made a small incision of about 2 mm on the furcular area of each bird, avoiding capillaries, and pulled a small amount (3–20 mg) of fat tissue through the incision with forceps, snipped the tissue with scissors, and then placed the sample in a cryotube and immediately into a freezer. We sterilized each incision using ethyl alcohol post-biopsy, then closed and sealed each incision with tissue adhesive (New Skin, Benzethonium chloride 0.2%). After the adhesive dried (< 1 minute), we placed the bird into a clean cloth bag for 10–15 minutes to recover and monitored it for signs of discomfort or stress (e.g., lethargy, panting or gaping, fluffed body feathers, closed or partially closed eyes, drooping wings). After the recovery period, we visually confirmed that the incision was closed, and released the bird into the wild.
Captive birds
For validation of consistency in stable hydrogen and stable carbon isotope values within a group of conspecifics fed identical diets, during fall 2020 we held Swainson’s Thrushes (n = 9) in temporary captivity. These birds had no visible fat reserves upon their initial capture during normal banding operations. We placed the thrushes in individual cages in an outdoor aviary at Powdermill and provided them with a constant and identical diet (waxworms grown and distributed by The Bug Company, Ham Lake, MN) and local water for up to 12 days until they deposited fat. Following that period, we biopsied furcular fat and released each thrush following the procedure outlined above. Fat samples from two of the thrushes were large enough to run triplicate stable hydrogen isotope analyses. We calculated the coefficient of variation (SD/mean * 100) for each sample to estimate the precision of stable-isotope analysis.
Wild birds
During the spring migration seasons of 2020 and 2021, we biopsied furcular fat from five species with low fat scores (score of 1–2 on a scale of 0–7) captured on the mornings after nights of heavy migration activity (i.e., banding days with higher capture rates and higher proportions of new versus recaptured individuals, and observations of increased numbers in the general mist net area). Because birds catabolize fat as they migrate and often arrive to stopover locations with low fat stores (e.g., Moore and Kerlinger 1987, DeSimone et al. 2020), we made the assumption that birds with relatively low fat content caught on these days likely arrived to Powdermill that morning and were unlikely to have deposited fat locally, therefore reflecting the isotope values of their previous stopover site. We sampled three species in spring 2020 (Hermit Thrush [Catharus guttatus, n = 2], Swainson’s Thrush [n = 17], and Magnolia Warbler [Setophaga magnolia, n = 22]) and five species in spring 2021 (Swainson’s Thrush [n = 3], Hermit Thrush [n = 2], Wood Thrush [n = 3], Gray Catbird [Dumetella carolinensis, n = 3], and Magnolia Warbler [n = 8]).
For local validation during fall 2020, we biopsied fat from birds that had no fat at their initial capture but that had gained fat when they were recaptured 4 to 46 days later with the assumption that newly deposited fat would reflect local isotope values. We sampled two species in fall 2020: Red-eyed Vireo (Vireo olivaceus, n = 10) and Northern Waterthrush (Parkesia noveboracensis, n = 4).
We stored fat samples from the captive and wild birds in a -80 °C freezer prior to stable-isotope analysis. We freeze dried the samples then isolated the lipids from the fat tissue using the process of soaking for 48 hours in a 2:1 chloroform:methanol solution and vortexing at the midpoint of the soak. We collected the purified lipids by filtering the solution as outlined in the chicken fat experiment methods and allowed the chloroform:methanol to evaporate. We loaded the lipids into tin (δ13C, 0.5mg; captive Swainson’s Thrushes only) or silver (δ2H, 0.35mg; all captive and wild birds) capsules for stable-isotope analysis at the University of Akron’s Stable Isotope Facility, Akron, Ohio following the procedure outlined above in the chicken fat experiments.
Statistical analysis
We used R version 4.3.2 (R Core Team 2023) for all statistical tests. We calculated percent yield from the chicken fat samples using the following equation
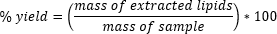
|
(2) |
and used a one-way ANOVA to test whether lipid extraction processes affected the volume of lipid extracted from the samples, and a Tukey HSD test to conduct post-hoc comparisons.
To test for effects of sample preparation methods on stable-isotope values, we used t-tests to compare isotope values of lipids from chicken fat samples that were freeze dried versus not freeze dried (excluding the unpurified samples) and to compare isotope values of purified lipids versus whole samples. We used a one-way ANOVA to test whether variations of sample preparation (i.e., length of soak in chloroform:methanol solution and whether samples were vortexed during soak) affected stable-isotope values (Table 1: Treatments C, D, and E). We conducted Tukey HSD tests to identify pairwise differences in isotope values between sample preparation methods.
To provide evidence that fat biopsy in live, wild birds is a safe method, we compared the recapture rate for biopsied (spring 2020, fall 2020, and spring 2021) versus non-biopsied birds using Fisher’s exact test for each of the seven focal species (Table 2). We used the recapture rate of unsampled birds in spring 2019 as a proxy for spring 2020 because the COVID-19 pandemic prevented normal banding operations in spring 2020. We performed separate tests for each species because stopover biology and therefore recapture rate differs among focal species (e.g., Northern Waterthrushes frequently stopover at Powdermill for two or more weeks and are recaptured at a high rate, whereas Swainson’s Thrushes are rarely recaptured).
For presumed distant-origin birds, we used t-tests to compare stable hydrogen and stable carbon isotope values between years (i.e., 2020 and 2021) for each of three species (Magnolia Warbler, Swainson’s Thrush, Hermit Thrush). We used one-way ANOVAs to test for differences in stable hydrogen and stable carbon isotope values between these three species and performed a post-hoc Tukey Honest Significant Differences test to compare stable hydrogen isotope values between the species. We sampled two additional species in spring 2021 (Gray Catbird and Wood Thrush) and used one-way ANOVAs to test for differences in stable hydrogen and stable carbon isotope values between these five species. We performed a post-hoc Tukey Honest Significant Differences test to compare stable hydrogen isotope values between the species.
We used t-tests to compare stable hydrogen and stable carbon isotope values between the two species with presumed local-origin fat (Northern Waterthrush, Red-eyed Vireo) to test whether two species that presumably foraged and assimilated fat in the same location showed comparable stable-isotope values.
RESULTS
Sample preparation effects
The yield of extracted lipids from samples of store-bought chicken fat tissue (n = 28) ranged between 35.6 and 64.3% per sample, and lipid yield in large (23.2–30.8 mg; mean = 52.3%) and small (12.9–15.7mg; mean = 54.2%) fat samples was similar. There was no statistical difference in yield between sample preparation methods (Table 1; F(3,18) = 0.041, p = 0.989).
Sample preparation methods affected stable hydrogen and carbon isotope values (Fig. 1). Samples that were freeze dried had significantly less negative δ2H values than those that were not freeze dried (t = 2.2554, df = 9.2376, p = 0.0498) but there was no significant difference in δ13C values in freeze dried versus non-freeze dried samples (t = -0.7457, df = 18.551, p = 0.465). Lipids had significantly higher δ2H values (t = -2.8242, df = 5.1072, p = 0.03606) and significantly lower δ13C values (t = 5.169, df = 15.04, p < 0.001) than whole fat samples. Lipid stable hydrogen and carbon isotope values did not differ between lipid purification processes (i.e., treatments that varied in soak length and whether samples were vortexed during soak; δ2H: F(2,13) = 1.888, p = 0.191; δ13C: F(2,12) = 1.155, p = 0.348).
Field tests
Biopsy procedure
There were no statistical differences in recapture rate between biopsied and unbiopsied birds for any species sampled (all p > 0.05, Table 2). All birds were released within 10–15 minutes of the biopsy procedure. The veterinary adhesive set in < 1 minute, effectively closing the incision sites and remaining closed throughout the 10–15 minute recovery and observation period. All birds were alert and none showed signs of stress upon release, and all flew strongly.
Captive birds
Fat sampled from captive Swainson’s Thrushes with known identical diet and drinking water showed consistency in both δ2H (-269.8 ± 2.6‰) and δ13C values (-20.9 ± 0.3‰). Additionally, samples from two thrushes were separated into three subsamples each to run triplicate stable hydrogen isotope analyses. The coefficient of variation for these birds was -0.73 (SD = 1.96‰) and -0.33 (SD = 0.9‰). Variation among replicates and among individuals both fell within typical analytical error for δ2H and δ13C measurements. These samples showed that the method (i.e., freeze drying samples then purifying lipids in a 48-hour chloroform:methanol soak with a vortex at midpoint) is repeatable and consistent within individuals.
Wild birds
In spring 2020 and spring 2021, we sampled wild, migrating birds that we assumed to have distant-origin fat, that is, fat that was assimilated at a stopover location somewhere south of Powdermill. In 2021, the majority of birds were sampled on days following strong migration nights over Westmoreland County as estimated on BirdCast.info’s migration dashboard (Cornell Lab of Ornithology 2026; data from spring 2020 is not available). For all distant-origin species combined, lipid δ2H values ranged from -274.1 to -168.7‰ and lipid δ13C values ranged from -34.9 to -27.3‰. A subset of species were sampled both years (Swainson’s Thrush, Hermit Thrush, Magnolia Warbler). Stable hydrogen isotope values were not significantly different between years for Swainson’s Thrush (t = -0.89, df = 2.23, p = 0.46) or Hermit Thrush (t = -4.62, df = 1.49, p = 0.07), but Magnolia Warblers had less negative δ2H values in spring 2020 than in spring 2021 (t = 3.45, df = 9.25, p = 0.007). Stable carbon isotope values were not significantly different between years for Swainson’s Thrush (t = -2.1, df = 6.67, p = 0.08) and Magnolia Warbler (t = -0.09, df = 10.2, p = 0.93), and sample size was not sufficient to test this for Hermit Thrush.
Between species comparisons of the presumed distant-origin species sampled in the spring for both years pooled show significant differences in lipid δ2H values (F(2,51) = 27.61, p < 0.001). Tukey post-hoc comparisons showed that lipid δ2H values Magnolia Warblers were significantly more negative than Swainson’s Thrush (p < 0.001) and Hermit Thrush (p = 0.002), indicating that Magnolia Warblers likely assimilated fat further north (i.e., closer to our study site) than either species of thrush. Because δ2H values vary with latitude (increasingly negative values at northern latitudes), the among-species differences in δ2H values we found are consistent with the expectation that Swainson’s Thrushes may migrate farther between stopover events than Magnolia Warblers. Post-hoc comparisons showed no significant difference in δ2H values between Swainson’s Thrush and Hermit Thrush (p = 0.96). Sample sizes were lower for stable carbon isotope analysis for all three species. There was no significant difference (F(2,30) = 0.11, p = 0.9) in δ13C values between Magnolia Warblers (n = 18), Swainson’s Thrushes (n = 12), and Hermit Thrushes (n = 3), which suggests there were no major among-species differences in diet, vegetation types, and habitat quality and usage.
In spring 2021, we sampled fat from two additional presumed distant-origin species (Gray Catbird and Wood Thrush) and found differences between lipid δ2H values among species (F(4,56) = 15.41, p < 0.001; Fig. 2), which provides further evidence that species migrate varying distances between stopover sites. Tukey post-hoc comparisons show that Gray Catbirds had δ2H values (-248.4 ± 19.0‰, n = 3) similar to Magnolia Warblers (-241.9 ± 15.2‰, n = 30; p = 0.96), and Wood Thrushes (-207.8 ± 6.3‰, n = 3) had similar values to Hermit Thrushes (-211.6 ± 6.7‰, n = 4; p = 0.99) and Swainson’s Thrushes (-209.2 ± 17.8‰, n = 20; p = 0.99). However, Magnolia Warblers and Gray Catbirds had significantly more negative lipid δ2H values than the three thrush species (all p < 0.05). All five species had similar δ13C values (F(4,35) = 0.72, p = 0.58): Gray Catbird (-28.7 ± 0.8‰, n = 3), Magnolia Warbler (-30.1 ± 1.8‰, n = 18), Hermit Thrush (-29.7 ± 0.8‰, n = 3), Wood Thrush (-29.3 ± 0.7‰, n = 3), and Swainson’s Thrush (-30.2 ± 1.7‰, n = 12). However, sample size for these species in 2021 is low and we take these data as little more than evidence that our methods show strong potential.
Based on visual inspection of furcular fat reserves at initial capture and then upon recapture, we assumed that the birds sampled in fall 2020 had assimilated fat locally. Northern Waterthrushes (-228.35 ± 3.5‰, n = 4) had significantly more negative δ2H values than Red-eyed Vireos (-196.76 ± 5.7‰, n = 10; t = -12.6, df = 9.2, p < 0.05; Fig. 3) but δ13C did not differ significantly (t = -0.42, df = 3.5, p = 0.699) between the two species (Northern Waterthrush: -29.66 ± 1.1‰, n = 4; Red-eyed Vireo: -29.42 ± 0.49‰, n = 10).
DISCUSSION
Using fat tissue sampled from a store-bought chicken breast, we found that lipid extraction yields were near or greater than 50% of the sample mass. Because less than 1 mg of lipids are needed for stable hydrogen (0.35 mg) and stable carbon (0.5 mg) isotope analysis, small samples are adequate for these analyses, and sufficiently sized fat samples can be biopsied from small songbirds in the wild (e.g., Magnolia Warbler; ≈ 8–10g; Mulvihill et al. 2004, Dunning 2007).
Sample preparation effects and recommendations
We experimented with fat from the same individual chicken to test whether δ2H and δ13C values varied with different methods of sample preparation. We found that freeze drying had a small but significant impact on δ2H values but not δ13C values. As expected, water in lipid samples likely had different δ2H values than those contained in bird fat, and therefore residual water would alter hydrogen isotope ratios. Other studies involving soft animal tissues freeze dry samples prior to stable hydrogen isotope analysis (e.g., Wassenaar and Hobson 2000a, Hobson et al. 2004b, Keenan and DeBruyn 2019), stable carbon isotope analysis (Podlesak and McWilliams 2007, Génier 2025), and fatty acid analysis of lipids (e.g., Pierce and McWilliams 2005, McCue et al. 2009), and is a recommended preservation method to prevent sample decomposition (Elliot et al. 2023). We therefore recommend freeze drying fat tissue samples prior to lipid purification until there is strong evidence that it is not necessary.
We found that isolating lipids from biopsied fat tissue impacts both stable hydrogen and stable carbon isotope values. We predicted that extraneous materials (e.g., connective tissue, blood) in biopsied fat samples would affect both hydrogen and carbon isotope ratios: in δ13C because lipids tend to be depleted in 13C relative to other tissue types (Post et al. 2007) and in δ2H because hydrogen isotope values vary with diet source and physiology of fatty acid synthesis (Pilecky et al. 2021, Pilecky et al. 2023) and are depleted in 2H compared to other tissues (Hobson et al. 1999). That is, whole fat samples (i.e., samples from which lipids were not isolated) have isotope values derived from multiple tissues whereas purified lipid samples should have δ2H and δ13C values from lipids only. As expected, both stable hydrogen and carbon isotope ratios were significantly different in whole samples versus purified lipid samples, suggesting that the small amount of connective tissue and other tissues contained in the excised fat samples likely affect isotope ratios. For example, blood is metabolically active and because of its relatively fast turnover rate, it can reflect local isotope ratios in migrating birds (Podlesak et al. 2005, McKinnon et al. 2012). These findings are consistent with other studies with the inverse goal of investigating isotope ratios of other tissue types (e.g., muscle) and report the importance of removing lipids to obtain accurate stable-isotope data for those tissues (Newsome et al. 2010, Watt and Ferguson 2015, Elliot and Elliot 2016, Pilecky et al. 2021, Pilecky et al. 2023).
Statistical analysis suggests that δ2H values were similar between our lipid purification processes, which differed in length of soak in the chloroform:methanol solution and vortexing. However, δ2H values were the least variable when samples were freeze dried, soaked for 48 hours in chloroform:methanol solution, and vortexed at the midpoint of the soak. The sample size (n = 3) was not sufficient to quantify variation in δ13C values for this treatment. We used this process to pretreat our wild bird samples prior to isotope analysis and recommend it for similar, future studies. Notably, lipids are extracted for stable-isotope analysis of proteinaceous tissue in non-avian taxa using similar chloroform:methanol extraction techniques (e.g., narwhals, Watt and Ferguson 2015; sharks, McMeans et al. 2009, McMeans et al. 2013).
Overall, we found that stable hydrogen and carbon isotope analysis of fat is consistent and repeatable using a standardized method of freeze drying and lipid purification. In our study, the similarity in stable hydrogen and stable carbon values between the treatments in the chicken fat experiments and the small variation in values within each treatment indicates that the method we used shows promise.
We emphasize the importance of consistency among studies in sample preparation methods and reporting methods in detail for stable-isotope analysis of fat samples if results are to be comparable. We recognize that our study is an early step in the development of isotope analysis from wild bird fat, and we encourage further testing and refinements by others. From our assessments, we make the following recommendations for sample preparation in future studies:
- Biopsy at least 10–15 mg of fat tissue. Less than 1 mg of material is needed for stable hydrogen and stable carbon isotope analysis, so a sample of this size will ensure enough purified lipids considering percent yield from the lipid extraction process and loss of material due to transfer between vials.
- Freeze dry the fat tissue. This is important for stable hydrogen isotope analysis because freeze drying removes excess water that can affect δ2H values. However, freeze drying is potentially unnecessary for stable carbon isotope analysis because water does not affect δ13C values.
- Isolate lipids from the fat samples to remove extraneous tissue (e.g., blood, connective tissue) that can affect both δ2H values and δ13C values. We recommend dissolving the fat in a 2:1 chloroform:methanol solution for 48 hours and vortexing the vial at the midpoint of the soak, the treatment method that showed the least δ2H variation in our study.
Field tests
Biopsy procedure
Rocha et al. (2016) described a safe method to biopsy fat from the furcular area of living birds within a laboratory setting, with the suggestion that the method would be safe to use in the field on live birds. Using recapture rate of biopsied and unbiopsied birds, and visual inspection of the incision site on recaptured sampled birds, we provide evidence that fat biopsies in wild, migrating songbirds is a non-lethal method of obtaining fat tissue.
Although short-term recapture rates (i.e., recaptures within a migration season) did not suggest negative effects of our biopsy method on immediate condition or survival, we were unable to quantify potential effects biopsy might have had on the remainder of migration or on other periods during the birds’ annual cycle. We might expect some inherent risk (e.g., delayed migration and related carry-over effects) from sampling fat tissue because it is surgical and therefore more invasive than other tissue sampling (e.g., blood, feathers, claws). However, like humans who can regain fat after liposuction through increased lipid storage within remaining adipocytes (i.e., hypertrophy; Beidas and Gusenoff 2021), birds also employ hypertrophy (Everaert et al. 2022). Additionally, unlike in humans where fat cells are finite in adulthood (Beidas and Gusenoff 2021), birds can generate new fat cells (i.e., hyperplasia) in adulthood (Robey et al. 1988). Therefore, hyperplasia and hypertrophy during migratory fat deposition might allow birds to replace biopsied fat tissue as readily as they replace sampled feathers. Regardless, when collecting subcutaneous fat samples from wild birds, caution should be taken to biopsy the minimum volume necessary for analysis to reduce the energetic demands of recovery. In large seabirds, Owen et al. (2010) found no evidence of negative longer-term effects of fat sampling, including no greater weight loss, no reduced breeding success during the biopsy year, and no reduction in recapture rates in biopsied birds compared to unbiopsied birds. Similarly, Morrissey and Swekla (2023) found fat biopsy in captive Killdeer (Charadrius vociferus) to be safe. As with any method of handling, marking, or collecting samples from wildlife, studies should be designed, when possible, to test for potential long-term lethal and non-lethal effects of fat biopsy on wild migrating birds to ensure the safety of the procedure. We therefore encourage future studies to test for effect of biopsy on condition, survival, and long-term risks of the procedure. This could be addressed, for example, by deploying radio transmitters on non-biopsied and biopsied individuals to compare migration pathways, migration timing, and apparent survival between the groups using transmitter detections via the Motus Wildlife Tracking Network.
To increase sample size and expand number of species sampled, we suggest that fat be sampled from fresh, recently dead birds as part of the process of study skin preparation, as one avenue for eliminating potential risks from fat biopsy to migratory birds. Fat collection from salvaged specimens (e.g., birds collected during window collision monitoring) could provide large sample sizes from broad geographic regions using tissue that would likely otherwise be discarded. Although postmortem breakdown of tissues may affect isotope values, these changes do not occur within several hours. Using decomposing beaver carcasses, Keenan and DeBruyn (2019) found that during the active decomposition phase, fat tissue showed significant 15N enrichment compared to fresh tissue, but that there were no significant changes to 13C values throughout decomposition. Although the effect of decomposition on δ2H is unknown, there likely is minimal concern that isotope values will be affected by decomposition if samples are obtained shortly after death.
Captive birds
To our knowledge, the only studies that have attempted to trace the provenance of stable hydrogen and stable carbon isotopes in bird fat tissue (Hobson et al. 2004b, Anparasan and Hobson 2021) used tissue from sacrificed birds, some of which were held in strict laboratory conditions. We explored the feasibility of obtaining meaningful lipid isotope data from wild, migrating birds by quantifying δ2H and δ13C variation within a group of conspecifics (Swainson’s Thrushes) held in temporary captivity in an outdoor aviary with known diet and water source. The remarkable consistency in hydrogen (SD = 2.6‰) and carbon (SD = 0.3‰) isotope values in lipids from these individuals, and the small intra-individual variation we found in triplicates from larger samples from two individuals (SD = 0.9‰ and 1.9‰), showed similar, or lower, variation compared to other stable-isotope studies in bird tissues. Standard deviations in feather δ2H can range between 4–12 ‰ in wild birds depending on species and groupings within species (e.g., Wassenaar and Hobson 2000b, Langin et al. 2007, Reese et al. 2018) and in lipid δ2H are 3–4‰ in wild birds (Hobson et al. 2004b) and 5.5–14.5‰ in controlled experiments with captive birds (Anparasan and Hobson 2021). Other studies have found standard deviations in feather δ13C that are 0.6–5.3‰ (Wassenaar and Hobson 2000b), in breath 0.6–5.7‰ (MacDade et al. 2011), in plasma and red blood cells 0.1–0.3‰ (Norris et al. 2005), and in lipids 0.6–1.2‰ (Hobson et al. 2004b), all in wild birds. Standard deviation in lipid δ13C in captive birds in a controlled experiment were 0.1–0.6‰ (Anparasan and Hobson 2021). Our results from our temporarily captive Swainson’s Thrushes indicate that stable-isotope analysis of lipids is repeatable.
We found consistent δ13C values in fat sampled from captive Swainson’s Thrushes and from wild, migrating birds. However, because δ13C values do not give specific information about geographic origin, and because assessing diet and habitat use is beyond the scope of this paper, we used stable carbon isotope analysis in fat only to test sample preparation methods.
From their experiments, Anparasan (2019) and Anparasan and Hobson (2021) estimate that starch, carbohydrates, and oil in food contribute 44–46% and dietary water contributes 17.8–26.5% of the hydrogen in lipids extracted from fat in laboratory-held birds. These results indicate that about a quarter of the hydrogen in lipids derive from some other source likely contained within other dietary macromolecules or in residual fat. Hydrogen isotope variation in our study could have arisen from captive Swainson’s Thrushes ingesting other insects that may have entered the open-air aviary, variation in isotope values of waxworms, and isotope values from residual fat that was assimilated at a location away from Powdermill. Although stable-isotope analysis is repeatable in a controlled environment, free-living birds introduce movement and foraging choice as variables that, in combination with relative diet and drinking water contribution to hydrogen in lipids, makes geographic assignment of fat origin difficult at best.
Wild birds
Lipids in fat sampled from presumed distant-origin songbirds during spring migration show variability in δ2H values. Although we did not seek to assign specific geographic origins to fat in this study, interesting species-level patterns are discernable among focal species (Fig. 2). Magnolia Warblers had more negative δ2H values in both years of the study than Swainson’s Thrushes, indicating that they likely assimilated fat at more northerly latitudes and therefore the distance between their previous stopover site and Powdermill was shorter than in Swainson’s Thrushes. Some songbirds are capable of extreme feats of migration, flying hundreds of kilometers without fully depleting fat reserves (e.g., Moore and Kerlinger 1987, Gómez et al. 2017) or thousands of kilometers without stopping (e.g., Blackpoll Warbler; Nisbet 1970, DeLuca et al. 2015), whereas others stopover more frequently to rest and refuel (Stutchbury et al. 2011, Callo et al. 2013). Additional samples from three other species, albeit with low sample sizes, supported our expectations that some species are capable of longer flights and therefore longer distances between stopover locations. Hermit Thrushes and Wood Thrushes had δ2H values similar to Swainson’s Thrushes whereas Gray Catbirds had δ2H values similar to Magnolia Warblers (Fig. 2), suggesting that thrushes seem to be arriving to Powdermill from considerably farther south and therefore may be migrating longer distances between stopover locations than other species (e.g., Gómez et al. 2017). The δ2H variation among individuals of the same species arises because all individuals are not migrating together, and are likely stopping over at slightly different latitudes and spending variable amounts of time at those sites.
Although we made informed assumptions about which birds were likely to have arrived each morning, some variation in lipid δ2H also may arise from uncertainty about each individual’s arrival date. Because it is challenging to distinguish newly arrived birds from lean birds that may have been at a site for several days, especially at stopover sites that are not located along a migration barrier, we recommend that future work should strive to sample from birds with known arrival dates. Additionally, physiological sources of δ2H variation could result from diet composition in heterogeneous habitats (Bortolotti et al. 2013, Magozzi et al. 2020) and the proportion of water obtained from drinking water versus diet (Magozzi et al. 2020), differences in metabolic rate (Storm-Suke et al. 2012), and evaporative water loss (McKechnie et al. 2004).
Because hydrogen isotope values of animal tissues reflect the hydrogen in both diet and drinking water, the natural history, including seasonal diet switching and habitat use, of each species must be taken into account when attempting to assign geographic origin to fat. For example, although the sample sizes for Northern Waterthrush and Red-eyed Vireo were relatively small in this study, these species differed considerably in average δ2H despite the assumption that their fat was deposited locally. These species forage in distinct habitats and consume different food items, breed across different geographic ranges, and spend different amounts of time during stopover events. These dissimilarities, especially foraging level and habitat, may account for the offset in lipid δ2H values between these two species as they do in claw and feather tissues (Fraser et al. 2011). Alternatively, the offset in lipid δ2H between Red-eyed Vireo and Northern Waterthrush could reflect a true difference in the location of fat assimilation. Red-eyed Vireos breed locally to Powdermill Nature Reserve and many of the individual vireos in our study were known local breeders (based on recaptures). In contrast, Northern Waterthrushes sampled in this study migrated from more northerly latitudes. It is possible that waterthrushes had residual fat assimilated from those northern breeding grounds, in addition to locally assimilated fat, leading to their relatively low δ2H values. Although these two species and the captive Swainson’s Thrushes consumed the same local water, the δ2H contribution from diet differed (local food sources versus waxworms obtained from a company in Minnesota that were fed an unknown diet) and we therefore expected dissimilarity in δ2H values in fat. Both Red-eyed Vireos and Northern Waterthrushes have significantly less negative δ2H values than the captive Swainson’s Thrushes, and these differences could be further explained by δ2H in residual fat that was assimilated near the breeding grounds or more northern stopover location prior to Swainson’s Thrushes’ arrival to Powdermill. However, the δ2H values in both local species are less negative than in many of the distant-origin birds. Following the logic of relative geographic location, these values suggest that the birds local to Powdermill would have assimilated fat further south than the distant-origin birds, which were assumed to have fat origins south of Powdermill. The validity of this method, therefore, loses promise with between-species comparisons.
The diet of migrating songbirds changes seasonally (see McWilliams and Karasov 2001). Many birds rely heavily on insects to fuel spring migration whereas their diet may include a wider range of food items (e.g., arthropods, fruit, seeds) during fall migration. In particular, many songbirds incorporate fruit into their diets during migratory fueling as it is a high-energy source of food that helps build fat stores (Parrish 2000, Levey and Martinez del Rio 2001). Birds with dietary flexibility are able to utilize the most abundant food sources in a variety of habitats as they fuel for migration (Parrish 2000). Differences in diet between species, seasonal changes in diet within a species or individual, dietary trophic level (Birchall et al. 2005, Peters et al. 2012, van Wijk et al. 2021), and the composition of macronutrients in diet (Podlesak and McWilliams 2006), can affect isotopic values during tissue synthesis. The variation in δ2H values that arises from seasonal diet switching likely creates a barrier to between-season comparisons.
The next step toward geographic assignment of lipid δ2H values obtained from fat biopsied from migrating songbirds is to develop a calibration curve for lipids versus local precipitation δ2H as exists for feathers and other tissues. Such a calibration would require a geographically broad sampling of birds. Although our attempt to assign relative geographic location of distant-origin fat during spring migration showed promise, the significant offset in δ2H between species with locally deposited fat, and in δ2H between these local species and presumed distant-origin species, our study serves as a caution that lipid δ2H values are likely not useful in determining geographic location of fat assimilation. However, for those wishing to proceed, species-specific calibration curves relating compound-specific δ2H of individual fatty acids to precipitation will likely be most reliable for geographic assignment. Lipids are biochemically complex mixtures of fatty acids that have unique δ2H values due to the physiology and metabolic routing of macromolecules during tissue synthesis (Pilecky et al. 2023). Study is needed in consumer physiology to understand how macromolecule routing during tissue synthesis affects isotope values in lipids, however promising work using δ2H values in fatty acids allowed for tracing dietary fatty acids from producers to consumers (Pilecky et al. 2021). Because isotope values can vary significantly with diet and metabolic routing during tissue synthesis (Whiteman et al. 2019, Pilecky et al. 2021, Pilecky et al. 2023), the potential to gain meaningful information about origins of fat tissue using this method merits further investigation.
CONCLUSION
In this proof-of-concept study, we built on previous lab-based work to provide evidence that fat tissue can be safely biopsied from wild, migrating birds using a method that does not require sacrifice of study subjects. Further, we showed that our method of obtaining isotope values from purified lipids is repeatable within the same bird and between individuals of the same species. We tested whether stable-isotope analysis of purified lipids yields meaningful information about the location of fat assimilation and therefore relative distance between stopover locations. Although the spring season species-level patterns in birds assumed to have distant-origin fat showed promise, between-season and -species differences in δ2H values indicated that geographic assignment is much more complicated than we initially expected. Many variables need to be considered including how much δ2H values in residual lipids or other tissues contribute to newly assimilated fat, foraging habits and habitat, seasonal diet and contribution of animal versus plant and aquatic versus terrestrial food origin, and other interspecific differences that at present likely make interpretation of lipid δ2H values impossible. However, the small variation in isotope values we observed in fat sampled from captive birds supports the usefulness of stable-isotope analysis of lipids in research that does not involve identifying geographic origin of fat tissue. Future work could instead focus on information gained from stable carbon isotope analysis to assess habitat quality, diet and prey origins, and potentially cross-species comparisons of habitat use during stopover events.
RESPONSES TO THIS ARTICLE
Responses to this article are invited. If accepted for publication, your response will be hyperlinked to the article. To submit a response, follow this link. To read responses already accepted, follow this link.
ACKNOWLEDGMENTS
We thank Powdermill Nature Reserve, environmental field research station of the Carnegie Museum of Natural History for allowing us to conduct the field portions of this study on site. We express gratitude to Powdermill’s staff, seasonal bird banding technicians, and dedicated volunteers for their assistance with constant-effort mist netting during the field seasons, especially T. Crary for his help in 2020. Funding for laboratory work was provided by a student research grant from the Pennsylvania Society for Ornithology. Funding for Powdermill’s long-term bird monitoring program is generously provided by the Colcom Foundation, Laurel Foundation, and numerous private donors who have supported the program since its inception.
Bird banding was conducted under Powdermill Nature Reserve’s federal bird banding permit #08231 and Pennsylvania state bird banding permit #47050. Fat sampling and the short-term captivity portions of the study were conducted under state permit #48387 and U.S. Fish and Wildlife Service Scientific Collecting Permit #MB09838B-0. The study was approved by the University of Toledo’s IACUC (#108881).
Thank you to S. Morris and J. Refsnider for constructive comments on earlier drafts of this manuscript.
DATA AVAILABILITY
Data and code are currently available via a temporary, private-for-review link on Figshare (https://figshare.com/s/c0bfd7ba122cf216f62b). Upon publication, all data and code will be deposited in a permanent, publicly accessible repository.
LITERATURE CITED
Alerstam, T. 2011. Optimal bird migration revisited. Journal of Ornithology 152:5-23. https://doi.org/10.1007/s10336-011-0694-1
Anparasan, L. 2019. Tracing nutrient sources to lipid production in birds and insects using stable isotope (δ13C, δ2H) tracers: implications for nutritional physiology of migratory species. Thesis. Western University, London, Ontario, Canada.
Anparasan, L., and K. A. Hobson. 2021. Tracing sources of carbon and hydrogen to stored lipids in migratory passerines using stable isotope (δ13C, δ2H) measurements. Oecologia 195:37-49. https://doi.org/10.1007/s00442-020-04827-1
Araújo, P. M., I. Viegas, A. D. Rocha, A. Villegas, J. G. Jones, L. Mendonça, J. A. Ramos, J. A. Masero, and J. A. Alves. 2019. Understanding how birds rebuild fat stores during migration: insights from an experimental study. Scientific Reports 9:10065. https://doi.org/10.1038/s41598-019-46487-z
Barquete, V., V. Strauss, and P. G. Ryan. 2013. Stable isotope turnover in blood and claws: a case study in captive African Penguins. Journal of Experimental Marine Biology and Ecology 448:121-127. https://doi.org/10.1016/j.jembe.2013.06.021
Beidas, O. E., and J. A. Gusenoff. 2021. Update on liposuction: what all plastic surgeons should know. Plastic and Reconstructive Surgery 147:658-668. https://doi.org/10.1097/PRS.0000000000007419
Birchall, J., T. C. O’Connell, T. H. E. Heaton, and R. E. M. Hedges. 2005. Hydrogen isotope ratios in animal body protein reflect trophic level. Journal of Animal Ecology 74:877-881. https://doi.org/10.1111/j.1365-2656.2005.00979.x
Blem, C. R. 1976. Patterns of lipid storage and utilization in birds. American Zoologist 16(4):671-684. https://doi.org/10.1093/icb/16.4.671
Bortolotti, L. E., R. G. Clark, and L. I. Wassenaar. 2013. Hydrogen isotope variability in prairie wetland systems: implications for studies of migratory connectivity. Ecological Applications 23(1):110-121. https://doi.org/10.1890/12-0232.1
Callo, P. A., E. S. Morton, and B. J. M. Stutchbury. 2013. Prolonged spring migration in the Red-eyed Vireo (Vireo olivaceus). Auk 130(2):240-246. https://doi.org/10.1525/auk.2013.12213
Clegg, S. M., J. F. Kelly, M. Kimura, and T. B. Smith. 2003. Combining genetic markers and stable isotopes to reveal population connectivity and migration patterns in a Neotropical migrant, Wilson’s Warbler (Wilsonia pusilla). Molecular Ecology 12:819-830. https://doi.org/10.1046/j.1365-294X.2003.01757.x
Contina, A., A. K. Pierce, S. W. Yanco, E. S. Bridge, J. F. Kelly, and M. B. Wunder. 2022. Application of stable isotopes to study movement ecology and diet variation in a migratory songbird. Journal of Field Ornithology 93(4):6. https://doi.org/10.5751/JFO-00179-930406
Cooper, N. W., M. T. Hallworth, and P. P. Marra. 2017. Light-level geolocation reveals wintering distribution, migration routes, and primary stopover locations of an endangered long-distance migratory songbird. Journal of Avian Biology 48:209-219. https://doi.org/10.1111/jav.01096
Cornell Lab of Ornithology. 2026. BirdCast, Migration Dashboard, Westmoreland County, Pennsylvania, spring 2021. Cornell Lab of Ornithology, Ithaca, New York, USA. https://dashboard.birdcast.org/region/US-PA-129
Delmore, K. E., J. W. Fox, and D. E. Irwin. 2012. Dramatic intraspecific differences in migratory routes, stopover sites and wintering areas, revealed using light-level geolocators. Proceedings of the Royal Society B 279:4582-4589. https://doi.org/10.1098/rspb.2012.1229
DeLuca, W. V., B. K. Woodworth, S. A. Mackenzie, A. E. M. Newman, H. A. Cooke, L. M. Phillips, N. E. Freeman, A. O. Sutton, L. Tauzer, C. McIntyre, I. J. Setnhouse, S. Weidensaul, P. D. Taylor, and D. R. Norris. 2019. A boreal songbird’s 20,000 km migration across North America and the Atlantic Ocean. Ecology 100(5):e02651. https://doi.org/10.1002/ecy.2651
DeLuca, W. V., B. K. Woodworth, C. C. Rimmer, P. P. Marra, P. D. Taylor, K. P. McFarland, S. A. Mackenzie, and R. D. Norris. 2015. Transoceanic migration by a 12 g songbird. Biology Letters 11(4):20141045. https://doi.org/10.1098/rsbl.2014.1045
Deppe, J. L., M. P. Ward, R. T. Bolus, R. H Diehl, A. Celis-Murillo, T. J. Zenzal Jr., F. R. Moore, T. J. Benson, J. A. Smolinsky, L. N. Schofield, D. A. Enstrom, E. H. Paxton, G. Bohrer, T. A. Beveroth, A. Raim, R. L. Obringer, D. Delaney, and W. W. Cochran. 2015. Fat, weather, and date affect migratory songbirds’ departure decisions, routes, and time it takes to cross the Gulf of Mexico. Proceedings of the National Academy of Sciences 112(46):E6331-E6338. https://doi.org/10.1073/pnas.1503381112
DeSimone, J. G., M. G. Ramierez, C. R. Elowe, M. S. Griego, C. W. Breuner, and A. R. Gerson. 2020. Developing a stopover-CORT hypothesis: corticosterone predicts body composition and refueling rate in Gray Catbirds during migratory stopover. Hormones and Behavior 124:104776. https://doi.org/10.1016/j.yhbeh.2020.104776
dos Santos, I., A. M. M. Gonçalves, A. R. Carreiro, B. Martins, C. P. Rocha, C. Vieira, D. M. Matos, I. B. Gutiérrez, I. Rodrigues, N. Almeida, J. A. Ramos, V. H. Paiva, and P. M. Araújo. 2023. Similar breeding performance despite inter-annual differences in diet composition of seabirds inhabiting a tropical environment. Marine Ecology Progress Series 725:95-119. https://doi.org/10.3354/meps14463
Dunning, J. B. 2007. CRC handbook of avian body masses. Second edition. CRC, Boca Raton, Florida, USA https://doi.org/10.1201/9781420064452
Elliot, K. H., and J. E. Elliot. 2016. Lipid extraction techniques for stable isotope analysis of bird eggs: chloroform-methanol leads to more enriched 13C values than extraction via petroleum ether. Journal of Experimental Marine Biology and Ecology 474:54-57. https://doi.org/10.1016/j.jembe.2015.09.017
Elliot, K. H., J. D. Roth, K. Crook, and D. Yurkowski. 2023. Lipid extraction and sample preservation techniques for stable isotope analysis and ecological assays. Pages 241-257 in S. K. Bhattacharya, editor. Lipidomics: methods and protocols. Methods in Molecular Biology series vol. 2625. Springer Nature, New York, New York, USA. https://doi.org/10.1007/978-1-0716-2966-6_21
Everaert, N., E. Decuypere, and J. Buyse. 2022. Adipose tissue and lipid metabolism. Pages 647-660 in C. G. Scanes and S. Dridi, editors. Sturkie’s avian physiology. Academic, Cambridge, Massachusetts, USA. https://doi.org/10.1016/B978-0-12-819770-7.00015-3
Fraser, K. C., E. A. McKinnon, A. W. Diamond, and L. Chavarría. 2011. The influence of microhabitat, moisture, and diet on stable-hydrogen isotope variation in a Neotropical avian food web. Journal of Tropical Ecology 27:563-572. https://doi.org/10.1017/S0266467411000332
Génier, C. S. V. 2025. Combining fatty acid and stable isotope techniques to race lipids in birds. Dissertation. Western University, London, Ontario, Canada.
Gómez, C., N. J. Bayly, D. R. Norris, S. A. Mackenzie, K. V. Rosenberg, P. D. Taylor, K. A. Hobson, and C. D. Cadena. 2017. Fuel loads acquired at a stopover site influence the pace of intercontinental migration in a boreal songbird. Scientific Reports 7:3405. https://doi.org/10.1038/s41598-017-03503-4
Gourdon, J. 2017. Standard operating procedure #107: bird analgesia. McGill University, Comparative Medicine & Animal Resources Center, Montréal, Québec, Canada.
Guglielmo, C. G. 2018. Obese super athletes: fat-fueled migration in birds and bats. Journal of Experimental Biology 221(Suppl_1):jeb165753. https://doi.org/10.1242/jeb.165753
Hobson, K. A. 2002. Incredible journeys. Science 295:981-983. https://doi.org/10.1126/science.1069824
Hobson, K. A., L. Atwell, and L. I. Wassenaar. 1999. Influence of drinking water and diet on the stable-hydrogen isotope ratios of animal tissues. Proceedings of the National Academy of Sciences 96(14):8003-8006. https://doi.org/10.1073/pnas.96.14.8003
Hobson, K. A., L. Atwell, L. I. Wassenaar, and T. Yerkes. 2004b. Estimating endogenous nutrient allocations to reproduction in Redhead Ducks: a dual isotope approach using δD and δ13C measurements of female and egg tissues. Functional Ecology 18:737-745. https://doi.org/10.1111/j.0269-8463.2004.00890.x
Hobson, K. A., and L. I. Wassenaar. 1996. Linking breeding and wintering grounds of neotropical migrant songbirds using stable hydrogen isotopic analysis of feathers. Oecologia 109:142-148. https://doi.org/10.1007/s004420050068
Hobson, K. A., and L. I. Wassenaar. 2019. Tracking animal migration with stable isotopes. Second edition. Academic, London, UK. https://doi.org/10.1016/C2017-0-01125-4
Hobson, K. A., L. I. Wassenaar, and E. Bayne. 2004a. Using isotopic variance to detect long-distance dispersal and philopatry in birds: an example with Ovenbirds and American Redstarts. Condor 106:732-743. https://doi.org/10.1093/condor/106.4.732
Keenan, S. W., and J. M. DeBruyn. 2019. Changes to vertebrate tissue stable isotope (δ15N) composition during decomposition. Scientific Reports 9:9929. https://doi.org/10.1038/s41598-019-46368-5
Kelly, J. R., V. Atudorei, Z. D. Sharp, and D. M. Finch. 2002. Insights into Wilson’s Warbler migration from analyses of hydrogen stable-isotope ratios. Oecologia 130:216-221. https://doi.org/10.1007/s004420100789
Langin, K. M., M. W. Reudink, P. P. Marra, D. R. Norris, T. K. Kyser, and L. M. Ratcliffe. 2007. Hydrogen isotopic variation in migratory bird tissues of known origin: implications for geographic assignment. Oecologia 152:449-457. https://doi.org/10.1007/s00442-007-0669-3
Levey, D. J., and C. Martinez del Rio. 2001. It takes guts (and more) to eat fruit: lessons from avian nutritional ecology. Auk 118:819-831. https://doi.org/10.1093/auk/118.4.819
MacDade, L. S., P. G. Rodewald, and K. A. Hatch. 2011. Contribution of emergent aquatic insects to refueling in spring migrant songbirds. Auk 128:127-137. https://doi.org/10.1525/auk.2011.10186
Magozzi, S., H. B. Vander Zanden, M. B. Wunder, C. N. Trueman, K. Pinney, D. Peers, P. E. Dennison, J. J. Horns, Ç. H. Şekercioğlu, and G. J. Bowen. 2020. Combining models of environment, behavior, and physiology to predict tissue hydrogen and oxygen isotope variance among individual terrestrial animals. Frontiers in Ecology and Evolution 8:536109. https://doi.org/10.3389/fevo.2020.536109
Marra, P. P., K. A. Hobson, and R. T. Holmes. 1998. Linking winter and summer events in a migratory bird using stable-carbon isotopes. Science 282:1884-1886. https://doi.org/10.1126/science.282.5395.1884
Marshall, J. D., J. R. Brooks, and K. Lajtha. 2007. Sources of variation in the stable isotopic composition of plants. Pages 22-60 in R. Michener and K. Lajtha, editors. Stable isotopes in ecology and environmental science. Blackwell, Malden, Massachusetts, USA. https://doi.org/10.1002/9780470691854.ch2
McCue, M. D., O. Amitai, I. Khozin-Goldbery, S. R. McWilliams, and B. Pinshow. 2009. Effect of dietary fatty acid composition on fatty acid profiles of polar and neutral lipid tissue in Zebra Finches, Taeniopygia guttata. Comparative Biochemistry and Physiology Part A 154:165-172. https://doi.org/10.1016/j.cbpa.2009.06.002
McKechnie, A. E., B. O. Wolf, and C. Martínez del Rio. 2004. Deuterium stable isotope ratios as tracers of water resource use: an experimental test with rock doves. Oecologia 140(2):191-200. https://doi.org/10.1007/s00442-004-1564-9
McKinnon, E. A., C. Artuso, and O. P. Love. 2017. The mystery of the missing warbler. Ecology 98(7):1970-1972. https://doi.org/10.1002/ecy.1844
McKinnon, E. A., K. C. Fraser, A. W. Diamond, C. C. Rimmer, and J. M. Townsend. 2012. Stable-hydrogen isotope turnover in red blood cells of two migratory thrushes: application to studies of connectivity and carry-over effects. Journal of Field Ornithology 83(3):306-314. https://doi.org/10.1111/j.1557-9263.2012.00380.x
McMeans, B. C., M. T. Arts, C. Lydersen, K. M. Kovacs, H. Hop, S. Falk-Petersen, and A. T. Fisk. 2013. The role of Greenland sharks (Somniosus microcephalus) in an Arctic ecosystem: assessed via stable isotopes and fatty acids. Marine Biology 160:1223-1238. https://doi.org/10.1007/s00227-013-2174-z
McMeans, B. C., J. A. Olin, and G. W. Benz. 2009. Stable-isotope comparisons between embryos and mothers of a placenatrophic shark species. Journal of Fish Biology 75:2464-2474. https://doi.org/10.1111/j.1095-8649.2009.02402.x
McWilliams, S. R., and W. H. Karasov. 2001. Phenotypic flexibility in digestive system structure and function in migratory birds and its ecological significance. Comparative Biochemistry and Physiology Part A: Molecular & Integrative Physiology 128(3):557-591. https://doi.org/10.1016/S1095-6433(00)00336-6
McWilliams, S., B. Pierce, A. Wittenzellner, L. Langlois, S. Engel, J. R. Speakman, O. Fatica, K. DeMoranville, W. Goymann, L. Trost, A. Bryla, M. Dzialo, E. Sadowska, and U. Bauchinger. 2020. The energy savings-oxidative cost trade-off for migratory birds during endurance flight. eLife 9:e60626. https://doi.org/10.7554/eLife.60626
Moore, F. R., and P. Kerlinger. 1987. Stopover and fat deposition by North American Wood-Warblers (Parulinae) following spring migration over the Gulf of Mexico. Oecologia 74:47-54. https://doi.org/10.1007/BF00377344
Morrissey, C., and K. Swekla. 2023. Refinements in adipose tissue biopsy collection in shorebirds: effect on pain, wound healing, and mass gain. Journal of Field Ornithology 94(1):10. https://doi.org/10.5751/JFO-00219-940110
Mulvihill, R. S., R. C. Leberman, and A. J. Leppold. 2004. Relationships among body mass, fat, wing length, age and sex for 170 species of birds banded at Powdermill Nature Reserve. Eastern Bird Banding Association Monograph No. 1.
Newsome, S. D., M. T. Clementz, and P. L. Koch. 2010. Using stable isotope biogeochemistry to study marine mammal ecology. Marine Mammal Science 26:509-572. https://doi.org/10.1111/j.1748-7692.2009.00354.x
Nisbet, I. C. 1970. Autumn migration of the Blackpoll Warbler: evidence for long flight provided by regional survey. Bird Banding 41:207-240. https://doi.org/10.2307/4511673
Norris, D. R., P. P. Marra, T. K. Kyser, and L. M. Ratcliffe. 2005. Tracking habitat use of a long-distance migratory bird, the American Redstart Setophaga ruticilla, using stable-carbon isotopes in cellular blood. Journal of Avian Biology 36:164-170. https://doi.org/10.1111/j.0908-8857.2005.03398.x
Owen, E., F. Daunt, and S. Wanless. 2010. Sampling avian adipose tissue: assessing a nondestructive biopsy technique. Journal of Field Ornithology 81(1):92-98. https://doi.org/10.1111/j.1557-9263.2009.00265.x
Parrish, J. D. 2000. Behavioral, energetic, and conservation implications of foraging plasticity during migration. Studies in Avian Biology 20:53-70.
Peters, J. M., N. Wolf, C. A. Stricker, T. R. Collier, and C. Martínez del Rio. 2012. Effects of trophic level and metamorphosis on discrimination of hydrogen isotopes in a plant-herbivore system. PLoS ONE 7(3):e32744. https://doi.org/10.1371/journal.pone.0032744
Pierce, B. J., and S. R. McWilliams. 2005. Seasonal changes in composition of lipid stores in migratory birds: causes and consequences. Condor 107:269-279. https://doi.org/10.1650/7809
Pilecky, M., L. I. Wassenaar, S. Taipale, and M. J. Kainz. 2023. Protocols for sample preparation and compound-specific stable-isotope analyses (δ2H, δ13C) of fatty acids in biological and environmental samples. MethodsX 11:102283. https://doi.org/10.1016/j.mex.2023.102283
Pilecky, M., K. Winter, L. I. Wassenaar, and M. J. Kainz. 2021. Compound-specific stable hydrogen isotope (δ2H) analyses of fatty acids: a new method and perspectives for trophic and movement ecology. Rapid Communications in Mass Spectrometry 35(16):e9135. https://doi.org/10.1002/rcm.9135
Podlesak, D. W., and S. R. McWilliams. 2006. Metabolic routing of dietary nutrients in birds: effects of diet quality and macronutrient composition revealed using stable isotopes. Physiological and Biochemical Zoology 79(3):534-549. https://doi.org/10.1086/502813
Podlesak, D. W., and S. R. McWilliams. 2007. Metabolic routing of dietary nutrients in birds: effects of dietary lipid concentration on δ13C of depot fat and its ecological implications. Auk 124(3):916-925. https://doi.org/10.1093/auk/124.3.916
Podlesak, D. W., S. R. McWilliams, and K. A. Hatch. 2005. Stable isotopes in breath, blood, feces, and feathers can indicate intra-individual changes in the diet of migratory songbirds. Oecologia 142:501-510. https://doi.org/10.1007/s00442-004-1737-6
Post, D. M., C. A. Layman, D. A. Arrington, G. Takimoto, J. Quattrochi, and C. G. Montaña. 2007. Getting to the fat of the matter: models, methods and assumptions for dealing with lipids in stable isotope analysis. Oecologia 152:179-189. https://doi.org/10.1007/s00442-006-0630-x
R Core Team. 2023. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://doi.org/10.32614/R.manuals
Ralph, C. J., G. R. Geupel, P. Pyle, T. E. Martin, and D. F. DeSante. 1993. Handbook of field methods for monitoring landbirds. General Technical Report PSW-GTR-144. U.S. Forest Service, Pacific Southwest Research Station, Albany, California, USA. https://doi.org/10.2737/PSW-GTR-144
Reese, J. A., C. Tonra, C. Viverette, P. P. Marra, and L. P. Bulluck. 2018. Variation in stable hydrogen isotope values in a wetland-associated songbird. Waterbirds 41(3):247-256. https://doi.org/10.1675/063.041.0304
Robey, W. W., J. A. Cherry, P. B. Siegel, and H. P. van Krey. 1988. Hyperplastic response of adipose tissue to caloric overconsumption in sexually mature chickens. Poultry Science 67:800-808. https://doi.org/10.3382/ps.0670800
Rocha, A. D., P. M. Araújo, F. R. Martinho, J. A. Ramos, and J. A. Masero. 2016. A non-lethal biopsy technique for sampling subcutaneous adipose tissue of small and medium-sized birds. Journal of Field Ornithology 87:213-221. https://doi.org/10.1111/jofo.12145
Rubenstein, D. R., and K. A. Hobson. 2004. From birds to butterflies: animal movement patterns and stable isotopes. Trends in Ecology and Evolution 19(5):256-263. https://doi.org/10.1016/j.tree.2004.03.017
Schimmelmann, A. 1991. Determination of the concentration and stable isotopic composition of nonexchangeable hydrogen in organic matter. Analytical Chemistry 63:2456-2459. https://doi.org/10.1021/ac00021a013
Schmaljohann, H., C. Eikenaar, and N. Sapir. 2022. Understanding the ecological and evolutionary function of stopover in migrating birds. Biological Reviews 97(4):1231-1252. https://doi.org/10.1111/brv.12839
Stanley, C. Q., M. R. Dudash, T. B. Ryder, W. G. Shriver, K. Serno, S. Adalsteinsson, and P. P. Marra. 2021. Seasonal variation in habitat selection for a Neotropical migratory songbird using high-resolution GPS tracking. Ecosphere 12(3):e03421. https://doi.org/10.1002/ecs2.3421
Storm-Suke, A., L. I. Wassenaar, E. Nol, and D. R. Norris. 2012. The influence of metabolic rate on the contribution of stable-hydrogen and oxygen isotopes in drinking water to quail blood plasma and feathers. Functional Ecology 26(5): 1111-1119. https://doi.org/10.1111/j.1365-2435.2012.02014.x
Stutchbury, B. J. M., E. A. Gow, T. Done, M. MacPherson, J. W. Fox, and V. Afanasyev. 2011. Effects of post-breeding moult and energetic condition on timing of songbird migration into the tropics. Proceedings of the Royal Society B: Biological Sciences 278(1702):131-137. https://doi.org/10.1098/rspb.2010.1220
Terzer, S., L. I. Wassenaar, L. J. Araguás-Araguás, and P. K. Aggarwal. 2013. Global isoscapes for δ18O and δ2H in precipitation: improved prediction using regionalized climatic regression models. Hydrology and Earth Systems Sciences 17:4713-4728. https://doi.org/10.5194/hess-17-4713-2013
van Wijk, R. E., Y. Barshep, and K. A. Hobson. 2021. On the use of stable hydrogen isotope measurements (δ2H) to discern trophic level in avian terrestrial food webs. Diversity 13:202. https://doi.org/10.3390/d13050202
Vander Zanden, H. B., D. M. Nelson, M. B. Wunder, T. J. Conkling, and T. Katzner. 2018. Application of isoscapes to determine geographic origin of terrestrial wildlife for conservation and management. Biological Conservation 228:268-280. https://doi.org/10.1016/j.biocon.2018.10.019
Viegas, I., P. M. Araújo, A. D. Rocha, A. Villegas, J. G. Jones, J. A. Ramos, J. A. Masero, and J. A. Alves. 2017. Metabolic plasticity for subcutaneous fat accumulation in a long-distance migratory bird traced by 2H2O. Journal of Experimental Biology 220:1072-1078. https://doi.org/10.1242/jeb.150490
Wassenaar, L. I., and K. A. Hobson. 2000a. Improved method for determining the stable-hydrogen isotopic composition (δD) of complex organic materials of environmental interest. Environmental Science and Technology 34:2354-2360. https://doi.org/10.1021/es990804i
Wassenaar, L. I., and K. A. Hobson. 2000b. Stable-carbon and hydrogen isotope ratios reveal breeding origins of Red-winged Blackbirds. Ecological Applications 10:911-916. https://doi.org/10.1890/1051-0761(2000)010[0911:SCAHIR]2.0.CO;2
Wassenaar, L. I., and K. A. Hobson. 2001. A stable-isotope approach to delineate geographical catchment areas of avian migration monitoring stations in North America. Environmental Science and Technology 35:1845-1850. https://doi.org/10.1021/es0010204
Watt, C. A., and S. H. Ferguson. 2015. Fatty acids and stable isotopes (δ13C and δ15N) reveal temporal changes in narwhal (Monodon monoceros) diet linked to migration patterns. Marine Mammal Science 31(1):21-44. https://doi.org/10.1111/mms.12131
Whiteman, J. P., E. A. Elliott-Smith, A. C. Besser, and S. D. Newsome. 2019. A guide to using compound-specific stable isotope analysis to study the fates of molecules in organisms and ecosystems. Diversity 11:8. https://doi.org/10.3390/d11010008
Zenzal, Jr., T. J., A. J. Contina, J. F. Kelly, and F. R. Moore. 2018. Temporal migration patterns between natal locations of Ruby-throated Hummingbirds (Archilochus colubris) and their Gulf Coast stopover site. Movement Ecology 6:2. https://doi.org/10.1186/s40462-017-0120-2
Fig. 1
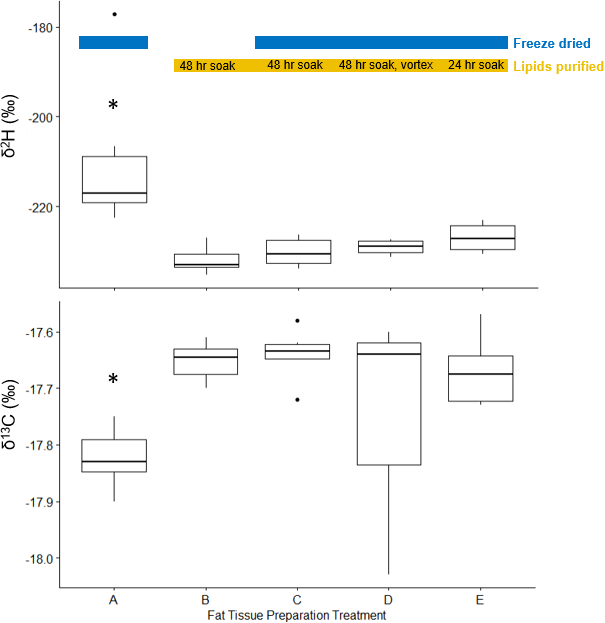
Fig. 1. Comparison of δ2H and δ13C values (reported in per mil notation) of fat sampled from a store-bought chicken breast for five different sample preparation treatments: (A) samples were freeze dried but lipids were not purified, (B) samples were not freeze dried but lipids were purified using a 48 hour soak, (C) samples were freeze dried and lipids were purified using a 48 hour soak, (D) samples were freeze dried and lipids were purified using a 48 hour soak and vortexed at the midpoint of the soak, and (E) samples were freeze dried and lipids were purified using a 24 hour soak. Whole fat samples had significantly less negative δ2H values and significantly more negative δ13C values than purified lipids (indicated with an asterisk).

Fig. 2
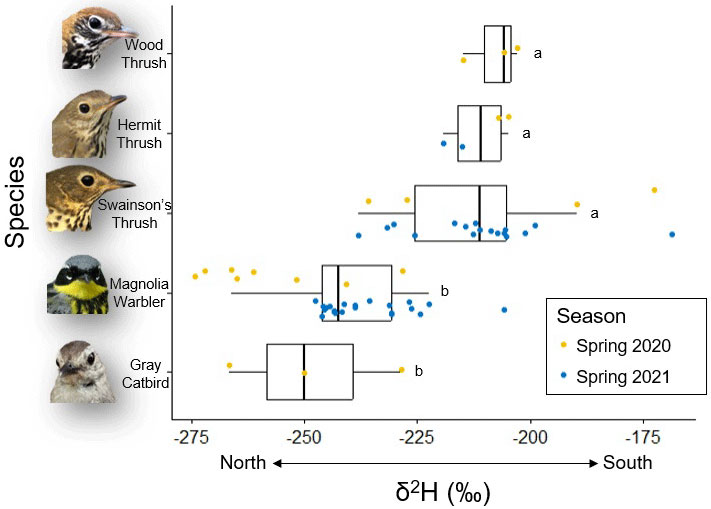
Fig. 2. The δ2H values (reported in per mil notation) of fat sampled from five species of presumed distant-origin migrating songbirds at Powdermill Nature Reserve in southwest Pennsylvania, USA during the spring 2020 and spring 2021 migration seasons. Different lowercase letters next to each species indicate statistical differences between δ2H values and same lowercase letters indicate no statistical differences between δ2H values.

Fig. 3
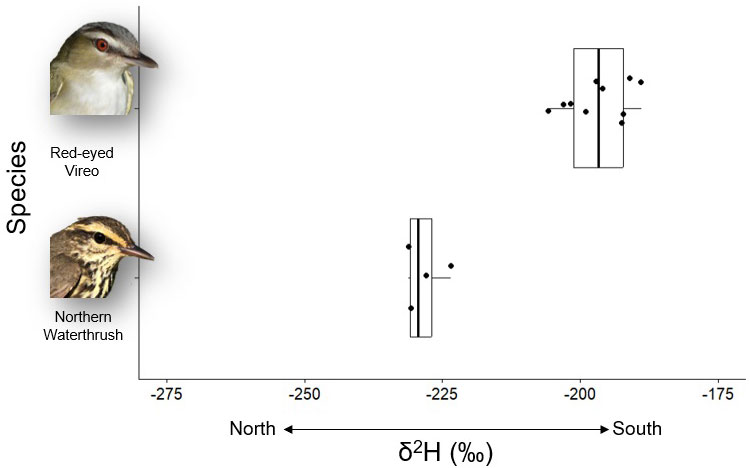
Fig. 3. The δ2H values (reported in per mil notation) of fat sampled from two species of migrating songbirds with presumed local-origin fat at Powdermill Nature Reserve in southwest Pennsylvania, USA during the fall 2020 migration season.

Table 1
Table 1. Sample preparation methods and lipid extraction processes used with chicken fat samples and mean (± standard deviation) for percent yield of lipids, lipid δ2H values, and lipid δ13C values for each treatment group.
| Treatment ID | Sample size | Freeze dry | Lipid purification | Vortex | Soak length | Lipid yield | δ2H | δ13C | |
| A | 6 | Yes | No | No | n/a | n/a | -210.0 ± 16.9‰ | -17.8 ± 0.05‰ | |
| B | 6 | No | Yes | No | 48 hours | 52.7 ± 10.3% | -232.0 ± 3.0‰ | -17.7 ± 0.03‰ | |
| C | 6 | Yes | Yes | No | 48 hours | 52.8 ± 7.1% | -230.2 ± 3.1‰ | -17.6 ± 0.05‰ | |
| D | 4 | Yes | Yes | Yes | 48 hours | 54.4 ± 8.1% | -229.1 ± 1.9‰ | n/a | |
| E | 6 | Yes | Yes | No | 24 hours | 53.1 ± 5.9% | -226.9 ± 3.2‰ | -17.7 ± 0.06‰ | |
Table 2
Table 2. Fisher’s exact tests comparing the recapture rate of biopsied and non-biopsied birds for each species sampled in this study.
| Biopsied | Non-biopsied | ||||||||
| Species | New | Recap | New | Recap | p-value | ||||
| Gray Catbird (Dumetella carolinensis) |
3 | 2 | 883 | 389 | 0.6449 | ||||
| Hermit Thrush (Catharus guttatus) |
4 | 1 | 161 | 31 | 0.5919 | ||||
| Magnolia Warbler (Setophaga magnolia) |
31 | 9 | 624 | 99 | 0.1569 | ||||
| Northern Waterthrush (Parkesia noveboracensis) |
6 | 2 | 116 | 60 | 0.7193 | ||||
| Red-eyed Vireo (Vireo olivaceus) |
12 | 1 | 979 | 150 | 1 | ||||
| Swainson's Thrush (Catharus ustulatus) |
42 | 1 | 1175 | 35 | 1 | ||||
| Wood Thrush (Hylocichla mustelina) |
3 | 0 | 170 | 10 | 1 | ||||








