The following is the established format for referencing this article:
Córdova, I., P. Escalante-Pliego, P. Laiolo, F. González-Garcia, O. Rojas-Soto, and G. Arnaud. 2026. Geographic variation of the song of the Belding’s Yellowthroat Geothlypis beldingi in the Baja California Peninsula, Mexico. Journal of Field Ornithology 97(2):2.ABSTRACT
Acoustic communication is essential for exchanging information between individuals of the same species. In birds, song plays a vital role in mate attraction and territory defense, and when it varies between populations, it can create barriers to gene flow and promote speciation. The Belding’s Yellowthroat (Geothlypis beldingi) is an endemic bird of the Mexican state of Baja California Sur and is endangered because of declining populations. Its populations are small and highly fragmented, but knowledge of the effects of fragmentation on the ecology and behavior of this species, especially its songs, remains limited. Five allopatric populations along its distribution were studied to assess their acoustic geographic variation. Six acoustic variables from 31 individual songs were analyzed using two supervised classification and multivariate techniques. Our results revealed significant differences between the northern and southern populations, which are separated by approximately 350 km, but the relationship between geographic distance and song variation is limited and could be influenced by different factors. Our results suggest that population-level acoustic differentiation may be associated with the “archipelago-like” distribution of oases. This association with fragmentation should be corroborated by more phenotypic characters and genetic information to determine if there are two significant evolutionary units within this species or lineages at the species level.
RESUMEN
La comunicación acústica es esencial para el intercambio de información entre individuos de la misma especie. En las aves, el canto cumple un papel fundamental en la atracción de pareja y la defensa del territorio, y cuando varía entre poblaciones, puede generar barreras al flujo génico y promover la especiación. Geothlypis beldingi es una especie endémica del estado mexicano de Baja California Sur que se encuentra en peligro debido a la disminución de sus poblaciones. Sus poblaciones son pequeñas y altamente fragmentadas, pero el conocimiento de los efectos de la fragmentación sobre la ecología y el comportamiento de esta especie, especialmente en sus cantos, sigue siendo limitado. Se estudiaron cinco poblaciones alopátricas a lo largo de su distribución para evaluar la variación geográfica acústica. Se analizaron seis variables acústicas a partir de 31 cantos individuales utilizando dos técnicas de clasificación supervisada y análisis multivariados. Nuestros resultados revelaron diferencias significativas entre las poblaciones del norte y del sur, las cuales se encuentran separadas por aproximadamente 350 km; sin embargo, la relación entre la distancia geográfica y la variación del canto es limitada y podría estar influenciada por diferentes factores. Nuestros resultados sugieren que la diferenciación acústica a nivel poblacional podría estar asociada con la distribución de tipo “archipiélago” de los oasis. Esta asociación con la fragmentación debería ser corroborada mediante otros caracteres fenotípicos e información genética para determinar si existen dos unidades evolutivas significativas dentro de esta especie o linajes a nivel de especie.
INTRODUCTION
Acoustic communication is essential for birds, and song plays a fundamental role in both mate attraction and territory defense. Song also conveys information about individual identity and, in some cases, the age and sex of the signaler. In addition, song function and information content may vary between sexes and may vary among populations (Marler and Tamura 1962, Catchpole and Slater 2008). Geographic variation in song can arise from several interacting factors, including differences in habitat structure (Morton 1975), climatic conditions (Slabbekoorn and Smith 2002, Tubaro and Lijtmaer 2006, Patten and Pruett 2009), and geographic isolation among populations (Baker 1996, Lachlan et al. 2018). In the case of learned songs, cultural divergence allows vocalizations to accumulate differences between isolated populations through drift or localized learning traditions (Podos and Warren 2007). Over time, such divergence may hinder recognition between individuals from different populations, generating a reproductive barrier that can eventually lead to speciation (Grant and Grant 1996, Lachlan and Servedio 2004). Thus, song variation not only reflects adaptations to the environment but may also act as a direct driver of evolutionary processes, particularly in species with vocal learning and limited dispersal capacity (Price 2008, Lachlan et al. 2018).
Studying geographic variation in a species’ song provides valuable information about its evolutionary trajectory (Slabbekoorn and Smith 2002, Podos and Warren 2007). Several studies show that geographically isolated populations may develop distinctive acoustic traits, and such changes can influence species recognition, even when genetic differentiation is low (Freeman and Montgomery 2017). For example, there is marked geographic variation in song structure among Hermit Thrush (Catharus guttatus) populations, which is associated with isolation, habitat differences, and cultural drift (Roach and Phillmore 2017). Likewise, evidence suggests that habitat selection and acoustic adaptation can contribute to the evolution of song differences and reproductive isolation when communication is shaped by local selective pressures (Patten et al. 2004). These cases demonstrate that the interplay among fragmentation, limited dispersal, and cultural transmission can have a substantial influence on the evolution of avian vocalizations.
Processes promoting vocal divergence are particularly relevant for species inhabiting naturally fragmented environments, such as the oases of the Baja California Peninsula. These oases function as small ecological islands, characterized by permanent water bodies and dense vegetation embedded within a desert matrix. This strong habitat discontinuity and limited connectivity can promote population differentiation (Grismer and McGuire 1993, Maya et al. 1997). Because dispersal among oases is likely restricted, populations may diverge behaviorally, morphologically, or acoustically in archipelagos or other fragmented systems. Within this context lies the Belding’s Yellowthroat (Geothlypis beldingi), an endemic species classified as endangered (SEMARNAT 2010) and listed as Vulnerable by the IUCN Red List (IUCN 2021). Currently, two subspecies are recognized: G. b. goldmani, in the northern region of Baja California Sur, and G. b. beldingi in the south (Oberholser 1917). The most recent estimates of the species’ total population are tentative. Still, the population estimates range between 650 and 1670 mature individuals (BirdLife International 2021), mainly distributed in San José del Cabo (SJ), San Ignacio (SI), La Purísima (PU), and to a lesser extent Comondú (CO) and San Bartolo (SB; Carmona et al. 2020, 2025). These abbreviations are used throughout the manuscript to refer to each population.
Despite its conservation importance, the biology and ecology of this species remain poorly understood, and even less is known about its vocal behavior. A previous study conducted in three oases (SI, CO, and SB) described aspects of the vocal structure of four individuals, focusing on the number and type of notes (Córdova-Rojas 2021). The song of male Belding’s Yellowthroats is a relatively simple structure composed of a short phrase with several clear and modulated notes, which is consistent with the general pattern reported for other Geothlypis species, whose songs are typically brief, repetitive, and characterized by small repertoires. Females only make calls; they do not sing. Variation was observed among the three study populations, with SB showing the least similarity with the other two (SI and CO; Córdova-Rojas 2021). This preliminary evidence showed that the species’ song exhibits detectable acoustic differentiation among populations, although it remains to be tested whether this divergence is due to the fragmented distribution of its habitat, to potentially short-distance cultural transmission, operating mainly within oases or among geographically close male populations.
The objective of this study was to analyze geographic variation in spectro-temporal characteristics of song across five populations. Regarding geographic differentiation and isolation-by-distance in the Belding’s Yellowthroat song, we predict that geographic distance affects song dissimilarities among individuals from different oases. We also expect differences in the temporal and frequency structure of songs among oases, which we tested using both multivariate and univariate techniques at the oasis level and between northern and southern populations.
MATERIALS AND METHODS
Study area
Five oases in Baja California Sur were selected based on the requirements of the Belding’s Yellowthroat, such as the presence of reed grass and cattail vegetation. The oasis habitat is characterized by mesic vegetation associated with permanent bodies of water, where palm groves (Washingtonia robusta and Phoenix dactylifera), reed grass (Phragmites communis), and cattail (Typha dominguensis) predominate, as well as riparian vegetation. Oases vary in area and vegetation composition. SI, PU, and CO oases are in the north of the region, while SB and SJ oases are situated in the south (Fig. 1). In SJ (1.4 km²), SB (0.59 km²), and SI (2.69 km²) sites, dense patches of reed grass predominate with reduced areas of cattail. In contrast, in PU (2.25 km²) and CO (1.49 km²), extensive reed grass patches are found adjacent to fruit orchards. The northern oases are separated by distances ranging from ~30 km (between PU and CO) to 147 km (between SI and PU), while in the southern oases, the distance is ~78 km (between SB and SJ). The separation between regions is considerable, with ~325 km (between CO and SB) and up to ~570 km between the most distant oases (SI and SJ).
Song recording
Digital records of G. beldingi were obtained between April and May 2020, 2023, and 2024. The same person, IBCR, recorded all individuals. A Marantz model PMD620 (44.1 kHz, 16-bit) digital recorder, paired with a Sennheiser ME67 microphone, were used. The recording period was from 04:30 to 05:30 am (approximately 30 minutes to an hour before sunrise), as birds reach their peak vocal activity during this time, which decreases considerably after sunrise.
Recording analysis
For all birds, repertoire size was assessed as the number of distinct song types recorded. Most males produced two song types; however, for the present analysis only the most frequent song type per individual was considered, as the other type was emitted sporadically and in smaller numbers. Of the 155 songs we recorded, 31 belonged to individuals from the five populations (SI = 6, PU = 7, CO = 6, SB = 6, SJ = 6 individuals), with five songs recorded per individual. From all these songs, five from each population, those with the best quality, were selected for analysis. Individual birds were identified by their locations within territories, and persistent territorial song helped distinguish each bird. We selected records from five individuals from each population with the best quality, with minimal urban and ambient noise, and minimal overlap with other songs. Noise reduction was applied to the recordings, followed by low-pass and high-pass filtering to preserve the signal of interest. Finally, the recordings were normalized to -1 dB using Audacity 2.3.3 software. Once processed, the songs were analyzed with the Raven Pro 1.6 Software (Cornell Lab of Ornithology, Ithaca, NY, USA) in two ways: the temporal parameter (song duration) was measured directly in the waveform (Fig. 2b). For this, the visible noise below the song was selected in the spectrogram and removed using the Filter Out Active Selection function (Ríos-Chelén et al. 2016). The threshold method was used for frequency measurements (Podos 1997, 2001, Ríos-Chelén et al. 2016), employing the power spectrum. This method is used to detect signals in a recording by standardizing measurements. A threshold of 15 dB was established, corresponding to a percentage of the maximum amplitude value detected in the signal. This threshold defined the parts of the signal suitable for analysis (Ríos-Chelén et al. 2016, Fernández-Gómez et al. 2020). The threshold value is arbitrary and depends on the quality of the recordings. Therefore, the minimum and maximum frequencies were defined as frequencies corresponding to the intersections with the left and right sides of the power spectrum curve (Fig. 2c), respectively, relative to the peak frequency (Ríos-Chelén et al. 2016). The parameters we measured for each song were the maximum frequency (Hz), minimum frequency (Hz), bandwidth (Hz), peak frequency (Hz), number of notes in the song, and duration of the song(s) (Hann window with 90% overlap, 11.6 ms resolution, and a 512 samples DFT), which represents a good compromise between frequency and duration resolution in a species with songs that include many short, quickly repeated notes that need to be identified.
Statistical analysis
Two supervised classification methods were used to determine whether the six selected acoustic variables could correctly classify G. beldingi populations: linear discriminant analysis (LDA) and random forest (RF) analysis. Additionally, a three-factor nested analysis of variance (ANOVA) was performed. For the LDA, the variables were first reduced to three dimensions using principal component analysis (PCA), with the resulting scores used as input. Locations were used as the dependent variable (explanatory variable), and scores were used as the independent variable. The percentage of correct classifications (TSS) for each group was calculated, and a confusion matrix was constructed. The RF analysis corroborated the population positioning based on acoustic variables. In this analysis, the two regions (north and south) were treated as the dependent variable, and our acoustic parameters were the independent variable. A confusion matrix was constructed, including an average error using the Gini index. For the nested ANOVA, the scores on the first three principal components from the PCA were used as dependent variables (one ANOVA per component, yielding three analyses). Populations were nested within groups (regions), and the individuals were nested within populations as categorical variables. Subsequently, a post-hoc Tukey test was performed. These analyses were conducted using R version 4.3.1 with the Ade4 package version 1.7-22 (Dray and Dufour 2007), MASS version 7.3 (Venables and Ripley 2002), and Random Forest version 4.7 (Liaw and Wiener 2002).
Using a Mantel test, we examined whether variation in G. beldingi song was influenced by geographic distance between individuals (isolation by distance). Two matrices were generated: one for acoustic distances and one for geographic distances. The first matrix was created by combining the scores of the first three principal components with the Euclidean distances calculated for each element. The second matrix contained geographical distances of the location points between individuals in kilometers. For this analysis, the Vegan package version 2.8-8 of the software in R was used (Oksanen et al. 2024).
To determine the song composition of G. beldingi, the different note types were identified visually by the same researcher (IBCR) and assigned a code for their identification. The composition of note types was subsequently analyzed using a Jaccard dissimilarity matrix constructed from presence/absence data for each note type per individual. To assess whether differences between regions were significant, a permutational analysis of variance (PERMANOVA) was performed. In addition, a principal coordinates analysis (PCoA) was performed to visualize clustering patterns among individuals in the multivariate space defined by the dissimilarity matrix, using the vegan package version 2.8-8 of software R (Oksanen et al. 2024).
RESULTS
The vocal behavior and social interactions of G. beldingi varied across the oases visited. In 2023, the vocal activity of birds in the oases of SI, PU, CO, and SB lasted approximately 30 minutes, while in SJ it lasted longer (60 minutes). However, in 2024, vocal activity differed, reducing the period from 30 to 20 minutes in the four oases mentioned above, while in SJ, the period was 40 minutes. G. beldingi sang within reed grass or cattail vegetation, avoiding open areas. Males are highly territorial and were usually observed accompanied by a female. An initial visual analysis of sonograms suggested that the song structure of populations from the northern and southern regions of Baja California Sur differed. The songs of northern birds (SI, PU, and CO) generally shared the same note types, although they differed in the way these notes were organized and in the number of elements. In SI, a note type we recorded was exclusive to this locality and absent in the other two (Fig. 2, red arrow). Additionally, the SI songs ended with two notes like those that introduce each phrase. In PU, songs were characterized by two similar phrases (Fig. 1). In contrast, in CO, the phrases exhibited a distinct structure compared to the other sites, resulting in shorter phrases with fewer notes (Table 1).
The song of the Belding’s Yellowthroat in the south of the Baja California Sur (SB and SJ) showed divergence from that in the northern region. In these populations, both the phrase structure and the type of notes differed from those in the northern area (Fig. 1). Two shared notes were identified: an ascending note at the beginning of the song and a descending note at the end of the song. Among southern populations, the song with the highest number of notes and the longest duration was that of the SJ population (Table 1). Despite this, birds from these two latter localities produced more notes per song than northern populations, which sang fewer notes and had longer intervals between them. Generally, the birds with the lowest minimum frequency were in the northern group, and those with the lowest maximum frequency were in the southern group (Table 1).
A total of 18 distinct note types were identified, with note type richness per individual being significantly higher in southern populations compared to northern ones. On average, northern individuals presented 4.89 ± 0.809 note types, while in the south, it was 6.08 ± 1.44 note types (see Fig. A1.1). PERMANOVA analysis revealed significant differences in note type composition between populations (R² = 0.61, F = 46.56, p < 0.001). These differences explained more than 60% of the total variation in repertoire structure between individuals. PCoA revealed a distinct separation across the northern and southern regions (see Fig. A1.2).
PCA’s first three principal components explained 81.39% of the total song variance, indicating a continuum of variation (see Table A1.1) with some separation between northern and southern populations, particularly in the first component (see Figure A1.3). The variation in PCA across latitude and longitude indicates a particular geographic pattern (higher PC1 in both southern and northern populations; see Fig. A1.3). The LDA revealed a difference between the northern and southern regions, correctly classifying 76.77% of songs (see Table A1.2). Similarly, the RF model showed a 78.26% correct classification rate (see Table A1.3), indicating good predictive performance in distinguishing between the northern and southern regions. However, greater confusion was observed when classifying observations in the southern group, which could suggest that the acoustic variables analyzed exhibit greater similarity or overlap in this region (see Table A1.3). The most important variable for this classification was the minimum frequency, followed by the number of notes, maximum frequency, and peak frequency (see Fig. A1.4).
When testing for differences among regions, populations, and individuals using nested ANOVAs, we found significant variation in the first component, primarily driven by differences between regions, followed by differences among populations within regions and individuals within populations. This indicates distinct acoustic patterns in minimum and maximum frequencies, bandwidth, and number of notes among areas, populations, and individuals, as also identified by Random Forest analyses (see Tables A1.1 and A1.4). The second component was strongly correlated with song duration. Although no significant differences were found between regions in this component, significant differences were observed among populations within regions and among individuals within populations. Finally, there were substantial differences between regions and among individuals within populations for the third component; however, no differences were observed among populations within regions, suggesting a variation related to the peak frequency (see Tables A1.1 and A1.4). With an isolation-by-distance analysis, the Mantel test showed a positive, albeit low, correlation between the acoustic characteristics of G. beldingi songs and the geographic distance between populations (r = 0.19, p = 0.001), suggesting that this relationship is limited.
DISCUSSION
Analysis of Geothlypis beldingi song composition showed marked differences between northern and southern populations, particularly in the diversity of note types. The clearest pattern supported by analyses is the geographic structuring of song; we found significant differences in note-type occurrence, minimum and maximum frequencies, and the number of notes, resulting in a consistent separation between the northern and southern regions. These results suggest that geographic distance and the fragmented “archipelago-like” distribution of oases reduce connectivity among populations and, as a consequence, may promote divergence in song characteristics independently of potential differences in population size, for which we lack information (see also Laiolo and Tella 2005, Benedict and Bowie 2009, Hamao et al. 2018, Yandell et al. 2018).
The PCA results support the geographic pattern of song divergence; the first component, linked to frequency parameters, showed significant differences between the northern and southern regions. Because these regions correspond to two recognized subspecies, G. b. goldmani in the north (larger bodied) and G. b. beldingi in the south (smaller bodied), part of the frequency divergence probably reflects morphological differences, as body size constrains the range of frequencies that a bird can produce (Shizuka et al. 2016, Roach and Phillmore 2017). The second component, associated with song duration, showed significant differences primarily at the local and individual levels, suggesting that temporal traits are more sensitive to microenvironmental factors or to learning between individuals within the same population. This pattern may also reflect that song duration is not under strong stabilizing selection, leading to greater flexibility and higher individual-level variability. In contrast, the third component, related to peak frequency, exhibited both regional and individual differences, indicating the combined influence of broad geographic divergence processes and intraindividual variation. Finally, Mantel analysis revealed a relatively low (r = 0.19), yet significantly positive, correlation between geographic distance and acoustic dissimilarity. This suggests that although distance contributes to song divergence, a significant portion of the variation remains unexplained.
A key ecological trait of G. beldingi is its limited dispersal capability, as it is considered a highly sedentary species (Rodríguez-Estrella et al. 1999). The species is closely associated with oases, where it inhabits reed grass (Phragmites communis) and cattail (Typha dominguensis) vegetation. These habitats are naturally discontinuous along the peninsula, with an approximate 350 km separation between the northern and southern regions. This spatial configuration restricts individual movements and significantly reduces opportunities for cultural exchange between populations. In this sense, oases function as small ecological islands in an arid environment, originating after the last glaciation (Ruiz-Campos et al. 2014). The discontinuity of these ecosystems reflects local processes that could be linked to broader dynamics of incipient differentiation.
Southern populations showed a greater variety of note types in Belding’s Yellowthroat songs than northern ones. This pattern is consistent with expectations for fragmented systems, where restricted dispersal and local cultural transmission can promote divergence in song structure among populations. In other bird species, larger or less isolated populations have been shown to exhibit greater acoustic diversity, likely because of increased opportunities for cultural exchange (Laiolo and Tella 2005). However, not all populations face the same degree of isolation. Differences in oasis size, spatial isolation, and habitat continuity may influence both effective population size and the dynamics of cultural transmission. In smaller or more isolated populations, restricted dispersal can accentuate song divergence by limiting cultural flow among individuals. This isolation also influences population size and, consequently, the dynamics of cultural transmission, thereby favoring processes such as cultural drift (Slabbekoorn and Smith 2002, Lachlan and Slater 2003, Podos and Warren 2007, Williams et al. 2013). Under these conditions, song divergence and the loss or differentiation of repertoire elements are more likely to occur (Marler and Tamura 1962, Chilton et al. 2002, Päckert 2018, Hensel et al. 2022). Although demographic data are not currently available for each oasis, preventing a direct assessment of the relationship between population size, oasis area, and note type diversity, the observed pattern is consistent with theoretical and empirical expectations for culturally transmitted traits in fragmented landscapes.
A similar pattern may be observed in Comondú (northern), one of the smallest and possibly most isolated populations of G. beldingi, whose songs are characterized by fewer notes than neighboring populations (SI and PU). This pattern is consistent with reports in other species, in which small populations experience a “cultural bottleneck” in song transmission, leading to reduced variety and complexity (Laiolo and Tella 2007). Song divergence patterns associated with small populations and geographic isolation have been documented in other birds’ species. In Darwin’s finches of the Galápagos Islands, cultural differences in song have contributed to the emergence of reproductive barriers and to allopatric speciation processes (Grant and Grant 1996). In the White-crowned Sparrow (Zonotrichia leucophrys), the formation of regional dialects has been linked to population isolation. It is considered a mechanism that reinforces separation between populations (Lipshutz et al. 2017, Toews 2017). Similarly, the Olive Sparrow (Arremonops rufivirgatus) exhibits song divergence in allopatric populations, reinforcing that spatial isolation can have significant effects on acoustic divergence in the absence of geographic connectivity (Fernández-Gómez et al. 2020). Finally, studies of the Common Yellowthroat (Geothlypis trichas) have shown marked geographic variation in song related to geographic isolation (Bolus 2014). These examples highlight that limited dispersal not only influences genetic diversity but also cultural divergence in song, a process that may be occurring in G. beldingi.
Although the classification models (LDA and RF) showed low accuracy, likely because of the limited sample size, our findings suggest that an early stage of acoustic differentiation between populations is present. Multiple factors, including morphological traits, ecological conditions, and anthropogenic pressures, could influence this divergence. For example, bird body size has been documented to influence song variation; larger birds tend to produce lower frequencies (Martin et al. 2011, Bolus 2014, Ríos-Chelén et al. 2017), which is consistent with the described morphological divergence between northern and southern populations of G. beldingi (Oberholser 1917). Furthermore, habitat structure can modify song propagation, as high frequencies are attenuated more rapidly in denser environments (Morton 1975). Finally, anthropogenic pressure is a key factor for southern populations, such as San José del Cabo (Rodríguez-Estrella et al. 1999, Navarro et al. 2019), where urban noise can interfere with bird communication (León et al. 2014, Ríos-Chelén et al. 2017), generating adjustments in song frequencies to achieve effective communication (Francis et al. 2011, Derryberry et al. 2016, Phillips et al. 2020). In summary, this evidence suggests that the divergence of G. belding’s song could be modulated by morphological, environmental, and anthropogenic factors. Beyond the description of acoustic patterns, our results underscore the importance of considering habitat connectivity as a crucial element for the conservation of this Mexican endemic species. The loss or degradation of these environments not only threatens the viability of their populations but also provides a natural scenario to understand how limited dispersal in fragmented environments can drive processes of population differentiation and evolution.
CONCLUSIONS
Significant differences were found in the song frequencies of G. beldingi between the northern and southern regions of Baja California Sur. Geographic distance influenced the observed variation, but it may not be the only factor explaining it. Morphological and acoustic divergence between northern and southern populations suggests these groups may represent early stages of population differentiation but additional genetic and ecological evidence is needed to determine whether they constitute distinct evolutionary units. Genetic analyses would help clarify the evolutionary processes underlying the patterns observed here. This study represents the first step in understanding the geographic variation in the song of one of the most endangered endemic species in Baja California Sur. Conducting this research posed significant challenges because of limited knowledge about the species and its behavior. Despite these difficulties in the field, our findings lay the groundwork for future research to explore potential factors influencing variation in this species’ song, such as environmental, reproductive, or anthropogenic factors.
RESPONSES TO THIS ARTICLE
Responses to this article are invited. If accepted for publication, your response will be hyperlinked to the article. To submit a response, follow this link. To read responses already accepted, follow this link.
AUTHOR CONTRIBUTIONS
IC: conceptualization, methodology, data curation, formal analysis, writing-original draft. PE: conceptualization, writing-review & editing. PL: resource, conceptualization, writing-review & editing. FG: conceptualization, methodology, writing-review & editing. OR: conceptualization, writing-review & editing. GA: Resource, funding acquisition, writing-review & editing.
ACKNOWLEDGMENTS
To CONAHCyT for the scholarship granted (No. 922194) to conduct this research. To the project "Endemic, NOM and ecologically relevant vertebrate species of the Baja California peninsula and Gulf of California islands" from CIBNOR, for funding for the fieldwork. To Fauna del Noroeste, A.C., for the scholarship provided for the partial realization of this work. To technicians José Abelino Cota Castro and Fany Reyes Bolaños for their valuable help on the field. To Doña Lupita, Don Bruno, Mrs. Paty, and Chava, who welcomed us into their homes during the field trips. To Gerardo Marrón, thank you for providing us with an invaluable photograph in this work. This work is dedicated to the memory of Dr. Ricardo Rodríguez-Estrella, who devoted his life to researching and conserving birds in northwestern Mexico, and with whom we started this project and whose support was fundamental to begin this work.
DATA AVAILABILITY
All of the data collected are included in the document.
LITERATURE CITED
Baker, M. C. 1996. Depauperate meme pool of vocal signals in an island population of singing honeyeaters. Animal Behaviour 51:853-858. https://doi.org/10.1006/anbe.1996.0089
Benedict, L., and R. C. Bowie. 2009. Macrogeographical variation in the song of a widely distributed African warbler. Biology Letters 5(4):484-487. https://doi.org/10.1098/rsbl.2009.0244
BirdLife International. 2021. Species factsheet: Belding’s Yellowthroat Geothlypis beldingi. https://datazone.birdlife.org/species/factsheet/beldings-yellowthroat-geothlypis-beldingi
Bolus, R. T. 2014. Geographic variation in songs of the Common Yellowthroat. Auk 131(2):175-185. https://doi.org/10.1642/AUK-12-187.1
Carmona, R., G. Marrón, S. Águila, I. Mata, S. Pérez, S. Alcalá, S. Ávalos, A. Mendoza, E. Flores, D. González, and S. Flores-Ramírez. 2025. Update on the breeding distribution of Belding’s Yellowthroat (Geothlypis beldingi). Western Birds 56(2). https://doi.org/10.21199/WB56.2.1
Carmona, R., G. Marrón, S. Águila, A. Rivas, S. Flores-Ramírez, and H. Reyes-Bonilla. 2020. Population assessment of Belding’s Yellowthroat (Geothlypis beldingi) at San Ignacio oasis, Baja California Sur, Mexico. Western Birds 51(1):38-46. https://doi.org/10.21199/WB51.1.3
Catchpole, C. K., and P. J. B. Slater. 2008. Bird song: biological themes and variations. Second edition. Cambridge University Press, Cambridge, UK. https://doi.org/10.1017/CBO9780511754791
Córdova-Rojas, I. B. 2021. Ecología del endémico y diversidad de cantos de la mascarita peninsular Geothlypis beldingi en oasis de Baja California Sur. Tesis de maestria. Centro de Investigaciones Biológicas del Noroeste, La Paz, Baja California Sur, México.
Chilton, G., M. O. Wiebe, and P. Handford. 2002. Large-scale geographic variation in songs of Gambel's White-crowned Sparrows. Condor 104(2):378-386. https://doi.org/10.1093/condor/104.2.378
Derryberry, E. P., R. M. Danner, J. E. Danner, G. E. Derryberry, J. N. Phillips, S. E. Lipshutz, K. Gentry, and D. A. Luther. 2016. Patterns of song across natural and anthropogenic soundscapes suggest that White-crowned Sparrows minimize acoustic masking and maximize signal content. PLoS ONE 11(4):e0154456. https://doi.org/10.1371/journal.pone.0154456
Dray, S., and A.-B. Dufour. 2007. The ade4 package: implementing the duality diagram for ecologists. Journal of Statistical Software 22(4):1-20. https://doi.org/10.18637/jss.v022.i04
Fernández Gómez, R. A., J. E. Morales-Mávil, and J. R. Sosa-López. 2020. Geographic variation and divergence of songs in the Olive Sparrow species complex. Journal of Field Ornithology 91(1):77-91. https://doi.org/10.1111/jofo.12320
Francis, C. D., C. P. Ortega, and A. Cruz. 2011. Noise pollution filters bird communities based on vocal frequency. PLoS ONE 6(11):e27052. https://doi.org/10.1371/journal.pone.0027052
Freeman, B. G., and G. A Montgomery. 2017. Using song playback experiments to measure species recognition between geographically isolated populations: a comparison with acoustic trait analyses. Auk 134(4):857-870. https://doi.org/10.1642/AUK-17-63.1
Grant, B. R., and P. R. Grant. 1996. Cultural inheritance of song and its role in the evolution of Darwin’s finches. Evolution 50(6):2471-2487. https://doi.org/10.1111/j.1558-5646.1996.tb03633.x
Grismer, L. L., and J. A. McGuire. 1993. The oases of central Baja California, Mexico. Part I. A preliminary account of the relict mesophilic herpetofauna and the status of the oases. Bulletin of the Southern California Academy of Sciences 92:2-24.
Hamao, S., H. M. Komatsu, and M. Shinohara. 2018. Geographic variation in Yellow Bunting songs. Ornithological Science 17(2):159-164. https://doi.org/10.2326/osj.17.159
Hensel, A. L. J., S. L. Dobney, S. M. Doucet, D. R. Norris, A. E. M. Newman, H. Williams, and D. J. Mennill. 2022. Microgeographical variation in birdsong: Savannah Sparrows exhibit microdialects in an island population. Animal Behaviour 188:119-131. https://doi.org/10.1016/j.anbehav.2022.04.008
International Union for Conservation of Nature (IUCN). 2021. Belding's Yellowthroat Geothlypis beldingi. The IUCN Red List of Threatened Species 2021:e.T22721839A182929291. https://dx.doi.org/10.2305/IUCN.UK.2021-3.RLTS.T22721839A182929291.en
Lachlan, R. F., O. Ratmann, and S. Nowicki. 2018. Cultural conformity generates extremely stable traditions in bird song. Nature Communications 9(1):2417. https://doi.org/10.1038/s41467-018-04728-1
Lachlan, R. F., and M. R. Servedio. 2004. Song learning accelerates allopatric speciation. Evolution 58(9):2049-2063. https://doi.org/10.1111/j.0014-3820.2004.tb00489.x
Lachlan, R. F., and P. J. B. Slater. 2003. Song learning by chaffinches: how accurate, and from where? Animal Behaviour 65(5):957-969. https://doi.org/10.1006/anbe.2003.2091
Laiolo, P., and J. L. Tella. 2005. Habitat fragmentation affects culture transmission: patterns of song matching in Dupont’s lark. Journal of Applied Ecology 42(6):1183-1193. https://doi.org/10.1111/j.1365-2664.2005.01093.x
Laiolo, P., and J. L. Tella. 2007. Erosion of animal cultures in fragmented landscapes. Frontiers in Ecology and the Environment 5(2):68-72. https://doi.org/10.1890/1540-9295(2007)5[68:EOACIF]2.0.CO;2
León, E., A. Beltzer, and M. Quiroga. 2014. El jilguero dorado (Sicalis flaveola) modifica la estructura de sus vocalizaciones para adaptarse a hábitats urbanos. Revista Mexicana de Biodiversidad 85(2):546-552. https://doi.org/10.7550/rmb.32123
Liaw, A., and M. Wiener. 2002. Classification and regression by randomForest. R News 2(3):18-22.
Lipshutz, S. E., I. A. Overcast, M. J. Hickerson, R. T. Brumfield, and E. P. Derryberry. 2017. Behavioural response to song and genetic divergence in two subspecies of White-crowned Sparrows (Zonotrichia leucophrys). Molecular Ecology 26(11):3011-3027. https://doi.org/10.1111/mec.14002
Marler, P., and M. Tamura. 1962. Song “dialects” in three populations of White-crowned Sparrows. Condor 64(5):368-377. https://doi.org/10.2307/1365545
Martin, J. P., S. M. Doucet, R. C. Knox, and D. J. Mennill. 2011. Body size correlates negatively with the frequency of distress calls and songs of Neotropical birds. Journal of Field Ornithology 82(3):259-268. https://doi.org/10.1111/j.1557-9263.2011.00329.x
Maya, Y., R. Coria, and R. Domínguez. 1997. Caracterización de los oasis. Pages 5-26 in L. Arriaga y R. Rodríguez-Estrella, editors. Los oasis de la península de Baja California. Centro de Investigaciones Biológicas del Noroeste, La Paz, Baja California Sur, México.
Morton, E. S. 1975. Ecological sources of selection on avian sounds. American Naturalist 109(965):17-34. https://doi.org/10.1086/282971
Navarro, J. J. P., R. Rodríguez-Estrella, and A. González-Abraham. 2019. Human activity and geographical factors influence vegetation and plant richness in the vanishing oases of Baja California peninsula. Natural Areas Journal 39(4):409-419. https://doi.org/10.3375/043.039.0404
Oberholser, H. C. 1917. A new subspecies of Geothlypis beldingi. Condor 19(6):182-184. https://doi.org/10.2307/1362768
Oksanen, J., R. Kindt, P. Legendre, B. O’Hara, and M. H. H. Stevens. 2024. The vegan package. Community ecology package 10:631-637.
Päckert, M. 2018. Song: the learned language of three major bird clades. Pages 75-94 in D. T. Tietze, editor. Birds species: how they arise, modify and vanish. Springer Nature, Cham, Switzerland. https://doi.org/10.1007/978-3-319-91689-7_5
Patten, M. A., and C. L. Pruett. 2009. The Song Sparrow, Melospiza melodia, as a ring species: patterns of geographic variation, a revision of subspecies, and implications for speciation. Systematics and Biodiversity 7(1):33-62. https://doi.org/10.1017/S1477200008002867
Patten, M. A., J. T. Rotenberry, and M. Zuk. 2004. Habitat selection, acoustic adaptation, and the evolution of reproductive isolation. Evolution 58(10):2144-2155. https://doi.org/10.1111/j.0014-3820.2004.tb01593.x
Phillips, J. N., C. Rochefort, S. Lipshutz, G. E. Derryberry, D. Luther, and E. P. Derryberry. 2020. Increased attenuation and reverberation are associated with lower maximum frequencies and narrow bandwidth of bird songs in cities. Journal of Ornithology 161:593-608. https://doi.org/10.1007/s10336-020-01751-2
Podos, J. 1997. A performance constraint on the evolution of trilled vocalizations in a songbird family (Passeriformes: Emberizidae). Evolution 51(2):537-551. https://doi.org/10.1111/j.1558-5646.1997.tb02441.x
Podos, J. 2001. Correlated evolution of morphology and vocal signal structure in Darwin’s finches. Nature 409:185-188. https://doi.org/10.1038/35051570
Podos, J., and P. S. Warren. 2007. The evolution of geographic variation in birdsong. Advances in the Study of Behavior 37:403-458. https://doi.org/10.1016/S0065-3454(07)37009-5
Price, T. 2008. Speciation in birds. Roberts and Company, Colorado, USA.
Ríos-Chelén, A. A., G. C. Lee, and G. L. Patricelli. 2016. A comparison between two ways to measure minimum frequency and an experimental test of vocal plasticity in Red-winged Blackbirds in response to noise. Behaviour 153(12):1445-1472. https://doi.org/10.1163/1568539X-00003390
Ríos-Chelén, A. A., A. N. McDonald, A. Berger, A. C. Perry, A. H. Krakauer, and G. L. Patricelli. 2017. Do birds vocalize at higher pitch in noise, or is it a matter of measurement? Behavioral Ecology and Sociobiology 71:29. https://doi.org/10.1007/s00265-016-2243-7
Roach, S. P., and L. S. Phillmore. 2017. Geographic variation in song structure in the Hermit Thrush (Catharus guttatus). Auk 134(3):612-626. https://doi.org/10.1642/AUK-16-222.1
Rodríguez-Estrella, R., L. R. Delgado, E. P. D. de Bonilla, and G. Blanco. 1999. Belding’s Yellowthroat: current status, habitat preferences and threats in oases of Baja California, Mexico. Animal Conservation 2(2):77-84. https://doi.org/10.1111/j.1469-1795.1999.tb00052.x
Ruiz-Campos, G., A. Andreu-Soler, M. R. Vidal-Abarca Gutiérrez, J. Delgadillo-Rodríguez, M. L. Suárez-Alonso, C. González-Abraham, and V. H. Luja. 2014. Catálogo de humedales dulceacuícolas de Baja California Sur, México. Secretaría de Medio Ambiente y Recursos Naturales, Instituto Nacional de Ecología y Cambio Climático, Cuidad de México, México.
Secretaria de Medio Ambiente y Recursos Naturales. 2010. Norma Oficial Mexicana NOM-059-SEMARNAT-2010. Diario Oficial de la Federación (DOF), Ciudad de México, México.
Shizuka, D., M. R. Lein, and G. Chilton. 2016. Range-wide patterns of geographic variation in songs of Golden-crowned Sparrows (Zonotrichia atricapilla). Auk 133(3):520-529. https://doi.org/10.1642/AUK-16-27.1
Slabbekoorn, H., and T. B. Smith. 2002. Bird song, ecology and speciation. Philosophical Transactions of the Royal Society of London B: Biological Sciences 357(1420):493-503. https://doi.org/10.1098/rstb.2001.1056
Toews, D. P. 2017. From song dialects to speciation in White‐crowned Sparrows. Molecular Ecology 26:2842-2844. https://doi.org/10.1111/mec.14104
Tubaro, P. L., and D. A. Lijtmaer. 2006. Environmental correlates of song structure in forest grosbeaks and saltators. Condor 108(1):120-129. https://doi.org/10.1093/condor/108.1.120
Venables, W. N., and B. D. Ripley. 2002. Modern applied statistics with S. Fourth edition. Springer, New York, New York, USA. https://doi.org/10.1007/978-0-387-21706-2
Williams, H., I. I. Levin, D. R. Norris, A. E. Newman, and N. T. Wheelwright. 2013. Three decades of cultural evolution in Savannah Sparrow songs. Animal Behaviour 85(1):213-223. https://doi.org/10.1016/j.anbehav.2012.10.028
Yandell, D. D., W. M. Hochachka, S. Pruett-Jones, M. S. Webster, and E. I. Greig. 2018. Geographic patterns of song variation in four species of Malurus fairy-wrens. Journal of Avian Biology 49(2):e01446. https://doi.org/10.1111/jav.01446
Fig. 1
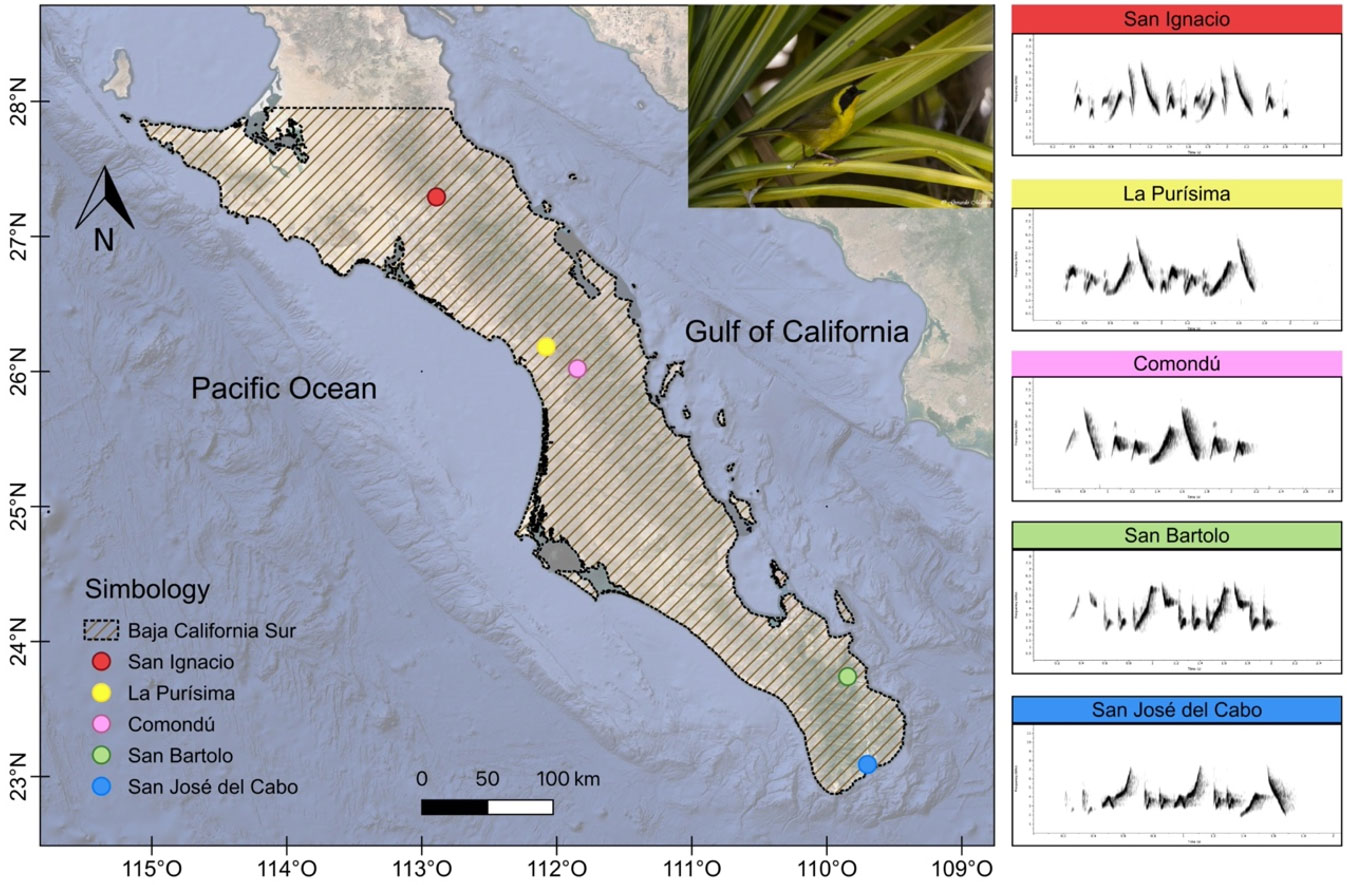
Fig. 1. Sampling sites of Belding’s Yellowthroat (Geothlypis beldingi) in Baja California Sur, Mexico, grouped into northern (San Ignacio, La Purísima, Comondú) and southern (San Bartolo, San José del Cabo) regions. Representative song spectrograms for each locality are shown (Hanning window, 90% overlap, 512 samples DFT).

Fig. 2
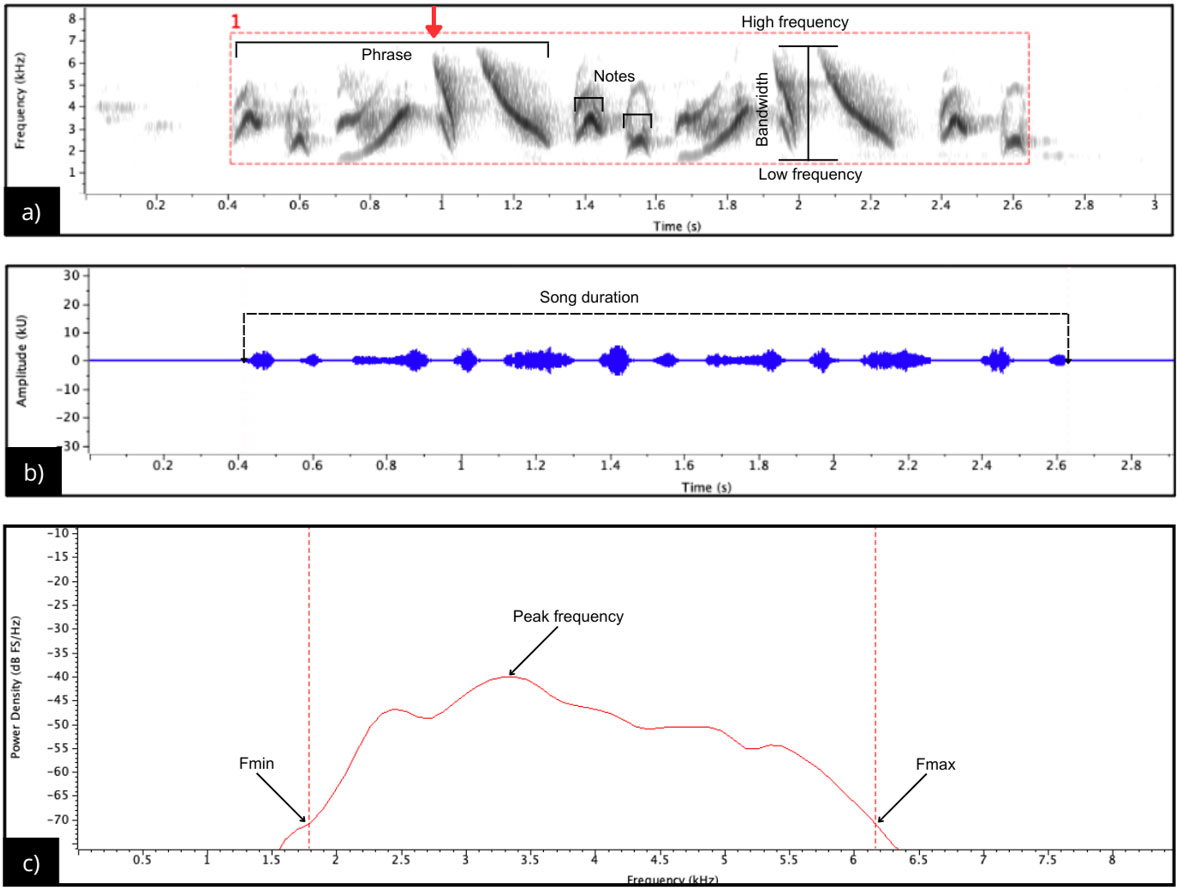
Fig. 2. Acoustic structure of Geothlypis beldingi song from San Ignacio: (a) spectrogram illustrating the temporal and frequency components used to measure song parameters (Hanning window, 90% overlap, 512 samples DFT). The song consists of two phrases of five notes each, followed by two terminal notes similar to introductory ones. A red arrow marks a note type observed only in songs from San Ignacio; (b) oscillogram showing how song duration measured; and (c) power spectrum indicating how minimum and maximum frequencies were determined using the threshold method.

Table 1
Table 1. Mean values (± SE) of acoustic variables measured from songs of Belding’s Yellowthroat (Geothlypis beldingi) across five localities in the Baja California Sur: San Ignacio (SI), La Purísima (PU), Comondú (CO), San Bartolo (SB), and San José del Cabo (SJ). Variables include minimum frequency (Fmin, kHz), maximum frequency (Fmax, kHz), bandwidth (AB, kHz), peak frequency (FP, kHz), number of notes (NN) and song duration (s). “Bird number” refers to the number of individual males from which songs were recorded in each locality.
| Variables | SI | PU | CO | SB | SJ | ||||
| Bird number | 6 | 7 | 6 | 6 | 6 | ||||
| Fmin | 2059 ± 37.8 | 2058 ± 28.3 | 2115 ± 51.6 | 2427 ± 36.3 | 2489 ± 50.5 | ||||
| Fmax | 5619 ± 66.5 | 5165 ± 59.4 | 5388 ± 111 | 5240 ± 80.2 | 5039 ± 145 | ||||
| AB | 2263 ± 57.2 | 1712 ± 37.1 | 1884 ± 63.9 | 1981 ± 66.3 | 1772 ± 86.2 | ||||
| FP | 3359 ± 58.6 | 3490 ± 39.6 | 3434 ± 52.3 | 3581 ± 100 | 3525 ± 64 | ||||
| NN | 10.7 ± 0.43 | 11 ± 0.40 | 9.5 ± 0.51 | 13.5 ± 0.54 | 14.1 ± 0.39 | ||||
| Duration | 1.90 ± 0.061 | 1.94 ± 0.067 | 1.62 ± 0.075 | 1.74 ± 0.056 | 1.91 ± 0.063 | ||||








