The following is the established format for referencing this article:
Lappin, O. A., K. O. Evans, R. B. Iglay, and M. D. McConnell. 2023. Northern Bobwhite (Colinus virginianus) breeding season roost site selection in a working agricultural landscape in Clay County, Mississippi. Journal of Field Ornithology 94(4):4.ABSTRACT
Appropriate habitat management may be one of the most important factors contributing to Northern Bobwhite (Colinus virginianus) population persistence, but biologists lack information on how individual bobwhite select roost sites during the breeding season. Therefore, we examined breeding season third-order roost site selection on B. Bryan Farms, Mississippi, from 2021 to 2022. We observed a quadratic relationship with average vegetation height, where roost site selection increased with increasing vegetation height to a point and then slightly decreased (β1 = 0.14084, 95% CI = 0.05, 0.24; β12 = -0.01005, 95% CI = -0.06, 0.04). However, uncertainty in the quadratic term was notable. Similarly, we observed a quadratic relationship with litter (β1 = 0.25479, 95% CI = 0.12, 0.39; β12 = -0.09606, 95% CI = -0.16, -0.04). We also found selection decreased linearly with increasing bare ground (β1 =-0.20938, 95% CI = -0.31, -0.11). Individual birds may require taller vegetation, greater visual obstruction, greater litter coverage, and lesser bare ground coverage for better concealment from nocturnal predators when they are roosting individually during the breeding season or are constrained by limited mobility (i.e., brooding). Understanding the vegetative composition, structure, and location of roost sites during the breeding season may provide land managers with a better understanding of the vegetative characteristics necessary during all phases of bobwhite life history. Our results provide the first information on bobwhite breeding season roost site selection, which will help to develop a more complete understanding of bobwhite habitat requirements and increase the effectiveness of habitat management and conservation efforts for this species of conservation concern.
RESUMEN
El manejo de hábitat apropiado puede ser uno de los factores más importantes que contribuyen a la persistencia de las poblaciones de Colinus virginainus, pero a los biólogos les hace falta información sobre como los individuos seleccionan los dormideros durante la temporada reproductiva. Por esta razón, examinamos la selección de tercer orden de los dormideros durante la temporada reproductiva en B. Bryan Farms, Mississippi, desde 2021 hasta 2022. Observamos una relación cuadrática con la altura promedio de la vegetación, donde la selección del dormidero incrementó con la altura de la vegetación hasta un punto en el que disminuyó levemente (β1 = 0.14084, 95% CI = 0.05, 0.24; β12 = -0.01005, 95% CI = -0.06, 0.04). Sin embargo, la incertidumbre en el término cuadrático fue notoria. Similarmente, observamos una relación cuadrática con los desechos vegetales (β1 = 0.25479, 95% CI = 0.12, 0.39; β12 = -0.09606, 95% CI = -0.16, -0.04). También encontramos que la selección disminuyo linealmente con el incremento del suelo desnudo (β1 =-0.20938, 95% CI = -0.31, -0.11). Aves individuales pueden requerir una vegetación más alta, mayor obstrucción visual, una mayor cobertura de desechos vegetales y una menor cobertura de suelo desnudo para ocultarse mejor de depredadores nocturnos cuando están durmiendo individualmente durante la temporada de reproducción o están restringidos por una movilidad limitada (i.e., incubando). Comprender la composición y estructura de la vegetación y la ubicación de los dormideros durante la temporada reproductiva puede proveer a los administradores de las tierras con un mejor entendimiento de las características de la vegetación necesarias durante todas las fases de la historia de vida de Colinus virginianus. Nuestros resultados proporcionan la primera información sobre la selección de los dormideros durante la temporada reproductiva para esta especie, lo cual ayudará a desarrollar un entendimiento más completo de los requerimientos de hábitat de Colinus virginianus e incrementar la efectividad en el manejo del hábitat y esfuerzos de conservación para esta especie sobre la cual existe preocupación por su conservación.
INTRODUCTION
Northern Bobwhite (Colinus virginianus; hereafter bobwhite) are a diurnal species that tend to select nocturnal ground roost sites to minimize predation and optimize thermoregulatory costs (Rosene 1969). Throughout their annual cycle, bobwhite experience changes in their social behavior. During the fall-winter bobwhite congregate in social groups, called coveys. Bobwhite coveys tend to roost in disk-like formations (i.e., with heads pointing toward the outside of the disk) to maximize vigilance and conserve heat, thereby increasing chances of overwinter survival (Rosene 1969). Coveys include individual bobwhite from either the same or different family groups and are most often composed of 10–15 individuals (Rosene 1969). Groups will join other nearby coveys in situations where covey numbers are too small and insufficient to form a large enough disk for adequate heat conservation and vigilance (Williams et al. 2003).
Although a myriad of studies have described roosting areas of bobwhite, most only quantified roost site selection of coveys during fall-winter (Stoddard 1931, Rosene 1969, Klimstra and Ziccardi 1963, Wiseman and Lewis 1981, Chamberlain et al. 2002, Hiller and Guthery 2005, Perkins et al. 2014). Klimstra and Ziccardi (1963), Hiller and Guthery (2005) and Perkins et al. (2014) observed covey roosting sites were most often in sparsely vegetated areas consistent with both Stoddard’s (1931) and Rosene’s (1969) roost site observations. Alternatively, Chamberlain et al. (2002) found that coveys in Missouri selected for roost sites with greater litter, vegetation height, and visual obstruction (VOS) compared to surrounding vegetation types. Although woody cover is important for predation evasion, roosting in more open areas allows disturbed coveys to quickly escape nocturnal predators (The National Bobwhite Technical Committee 2011, Perkins et al. 2014). Tillman (2009) studied nocturnal roosting behavior of Gray Partridge (Perdix perdix) coveys during the winter (November–February) and speculated that roosting in an exposed area was a type of predator avoidance as it allowed for unobstructed clear flight paths for escape if disturbed by a predator. Perkins et al. (2014) reasoned that this may be similar for bobwhite.
Research focusing on covey roost site selection is important considering the influence of overwinter survival on bobwhite population growth rates (Sandercock et al. 2008). Similar focus on individual roost site selection during the breeding season could also prove important considering the duration of the bobwhite breeding season (~6 months). Sandercock et al. (2008) also demonstrated that summer survival of adults had the second greatest contribution to variation in population growth rates. Individual bobwhite must optimize predator avoidance, proximity to mating opportunities, and thermal and energetic costs and benefits when selecting a roost site (Walsberg 1986, Burger et al. 2017). As a result, roost site selection likely has implications for near-term survival and long-term fitness of individual bobwhite. Understanding bobwhite habitat requirements throughout their annual cycle and at multiple spatial and temporal scales is essential to aid in bobwhite conservation efforts as individual roost site selection could influence bobwhite mortality during the breeding season. To date no published research has investigated individual bobwhite roost site selection during the breeding season.
We sought to identify and characterize, for the first time for this species, third-order roost site selection of bobwhite during the breeding season. Third-order selection can be described as use of habitat components within an individual’s home range (Johnson 1980). Examination of third-order, micro-habitat selection can increase understanding of bobwhite habitat requirements to inform management. We hypothesized that at the third-order of selection, bobwhite would optimize trade-offs between concealment and unobstructed flight paths to escape predators. Previous research on coveys indicates that bobwhite make context-specific evasive maneuvers when disturbed by predators (McGrath et al. 2018). McGrath et al (2018) also demonstrated that visual obstruction of vegetation influenced the type of evasive maneuvers used. Therefore, we expected bobwhites to select open grassland areas with lower visual height obstruction (height at which 100% obstruction stops) for nocturnal roosting throughout the breeding season. We also predicted individual bobwhite roost site locations would be inversely related to percentage of woody stems, non-native/invasive grass, and canopy closure. Hence, individual roost site locations should have a parabolic relationship with visual height obstruction, vegetation height, percentage native grass, forbs, litter, and bare ground.
METHODS
Study area
The study was conducted at B. Bryan Farms, a 1902.02-hectare privately owned property located near West Point, Mississippi (33°38′53″N, 88°34′43″W), elevation 64.01 m. Located in the Blackland Prairie soil region of Mississippi, B. Bryan Farms has a history of agricultural production (> 50 years) consisting of row crop (rotational soybean, corn), pastures, and livestock production. The property consists of 570.20 hectares (30%) of exotic pastures managed for livestock and forage production), 467.01 hectares (25%) of hardwood forests, 395.38 hectares (21%) managed for row crop production, and 364.62 hectares (19%) of managed native warm season grass (NWSG)/forb plant communities. The remaining 5% of the property comprises open pine forest, shrub rows, open water, and roads and yard. Average monthly precipitation for Starkville, MS (approximately 32 kilometers from West Point, MS) during the months of April–October was 148.749 mm in 2021 (NCEI 2023a) and 123.711 mm in 2022 (NCEI 2023b). Average monthly average temperature from April–October was 17.9 °C in 2021 (NCEI 2023a) and 22.1 °C in 2022 (NCEI 2023b).
Capture techniques
We captured male and female bobwhite from February to March 2021 and 2022 using baited funnel traps (Stoddard 1931). We baited traps with milo (Sorghum bicolor) and checked them every evening until traps were removed. We raked trap sites and cleared them of brush. We covered each trap with branches to conceal captured bobwhite from predators. We opportunistically placed traps at known bobwhite locations from the 2019–2021 breeding season counts and fall covey call surveys. We assumed that all birds in areas where bobwhites had been detected had an equal opportunity to be trapped and included in our study. We recorded trap ID, UTM coordinates, date deployed, and date removed. Bobwhite extracted from funnel traps were kept in pillowcases until every captured bird was processed. We recorded age and mass (g) of each bird captured (Rosene 1969). We banded all birds with an aluminum leg band stamped with a unique identification number. We used the presence of buffy/notched tips on the upper primary coverts to determine age (juvenile or adult), and we used plumage color and pattern to determine sex (Rosene 1969). We fitted both sexes with 6 g pendant-style mortality-sensing radio transmitters (Crystal-controlled, two-stage design pulsed by CMOS; American Wildlife Enterprises, Monticello, Florida, USA). We released birds simultaneously from pillowcases at the trap site after all data was recorded. Transmitters were located immediately after detecting a mortality signal and inferred cause of mortality was recorded. All capture and handling of bobwhite was conducted under MSU IACUC Protocol #: IACUC-21-017.
We supplemented our capture effort using small, unoccupied aircraft systems (UAS) with infrared (thermal) camera technology. We used UAS and homing techniques with a handheld receiver and H-series antenna to help locate and capture coveys containing a previously radio collared bird from baited-funnel trapping. We located coveys on their nocturnal roost sites following similar capture techniques used by Shirley and Janke (2021). We conducted all flights using an FAA part 107 certified UAS remote pilot and following part 107 regulations. We used three different UAS models: (1) a DJI Zenmuse XT2 with a 25 mm thermal camera mounted on a multirotor DJI Matrice 200 V2 (SZ DJI Technology Co., Ltd., Shenzen, China), (2) a DJI Zenmuse H20T with a 13.5 mm thermal camera mounted on a multirotor DJI Matrice 300 RTK, and (3) an Autel Evo II Dual with a 13mm, 640 X 512 pixel thermal camera (Autel Robotics, Bothell, Washington, USA). We conducted flights using both manual flights with a DJI Cendence remote controller or Autel Robotics V2 Smart Controller and the DJI Pilot or Autel Explorer app on Android software with a Samsung T500 tablet. We conducted flights in optimal conditions consisting of low wind (< 24 kph), low temperatures, cloudy skies, and no precipitation (Shirley and Janke 2021). One capture team member would walk 25–50 m of the covey using homing techniques with a handheld receiver and H-series antenna determined by telemetry signal strength. Pilots would then fly the drone over the estimated covey location and visual observers watched the tablet screen during flights for the presence of a covey, and primarily the ring-shaped thermal signature. Flights varied in altitude, but typically started at approximately 60 m above ground level (AGL) and descended to 15–30 m AGL if/when a possible covey was found. The UAS hovered overhead to help guide the capture team to the covey’s location if the covey was deemed probable of capture. The capture team, consisting of two or more individuals, walked toward the covey with handheld nets or weighted cast nets and spotlights. We used telemetry equipment to estimate distance to the covey before turning on spotlights. The capture team turned on spotlights when they were within an estimated 5 m of the covey determined by telemetry signal strength and UAS imagery. The team would set handheld nets or a weighted cast net over the covey as quickly as possible after the covey was located with spotlights. The team located birds that were not captured after the initial flush with the UAS and attempted to capture them with spotlights and handheld nets. A UAS capture attempt was defined as any time a UAS was deployed to search for a covey. We recorded the general location of UAS deployment, date of capture effort, time of UAS deployment and removal, number of bobwhites captured, and number of bobwhites collared. We continued to use UAS systems opportunistically throughout the 2022 non-breeding season (February–March). Birds were marked in the same way as above.
In addition to the use of UAS systems and baited funnel traps, we implemented targeted mist-netting techniques following methods described by Wiley et al. (2012) once coveys broke up and males began to whistle (around May). Targeted mist-netting was only used to capture males. We used 61 mm nylon mesh, 4-shelf mist nets (12 m x 2.6 m) suspended between two 3.05 m aluminum conduit poles (1.91 cm diameter). We used up to 4 nets depending on the vegetation structure and difficulty of capture locations where flight direction was less predictable (Wiley et al. 2012). We identified the apparent location for individual males using auditory and visual cues (Wiley et al. 2012). We assembled nets near the anticipated bird location and positioned to align with the predicted flight path of flushed individuals. The most likely flight path was assumed from nearby cover and vegetation arrangement (Wiley et al. 2012).
Mist-netting was opportunistic and non-random because we relied on sensory cues. We used a Bluetooth speaker to play a variety of bobwhite calls (mostly female calls) to attract males to the net. Birds that did not successfully enter the net on their own were flushed by the observer(s). A flushing attempt occurred once a bird was < 25 m from the net. The observer(s) walked around behind the bird attempting to flush it directly into the net. A netting attempt was defined as any time a net was deployed and playback was used. A successful attempt was when ≥ 1 bobwhite was captured. We recorded the general location of nets, date of capture effort, time of net deployment and removal, number of bobwhites captured, and number of bobwhites collared. We continued to mist-net for birds opportunistically throughout the breeding season (15 April–15 August 2021–2022) to increase our sample size. Bobwhite capture and surveys occurred in most weather conditions except during periods of rain and high wind speed (≥ 33 kph; Seiler et al. 2002). Birds were marked in the same way as above.
Roost site selection
Comparing used resources to available resources is the most common method for evaluating bobwhite resource selection (i.e., resource selection function; Boyce and McDonald 1999, Williams et al. 2000, Janke and Gates 2013). Comparing characteristics (i.e., vegetation structure, environmental covariates) at both used and available points helps to better understand which characteristics are most important to bobwhite when selecting a roost location during the breeding season.
We determined roost site locations of radio-collared birds 4–7 times weekly after sunset on rotation using homing techniques with a handheld receiver and H-series antenna. We did not locate birds on days with rain or high wind speeds (≥ 33 kph; Seiler et al. 2002). We flagged approximate roost site locations at observation points 25–50 m from the individual to avoid flushing or disturbing birds (Wellendorf et al. 2004). We marked where we estimated the individual to be roosting in Avenza Maps 3.14.1 (154.2; https://store.avenza.com/) on a mobile device. We recorded our best estimates of the birds’ locations, but some location error was unavoidable (Kroeger et al. 2020). We collected roost site vegetation data the following day. We collected vegetation data during a different time or later date if a bird had not moved further than 200 m from the roost location to avoid flushing using a 140 m buffer distance. We used a 140 m radius from individual roost locations for random point selection based on the average daily core area used by bobwhite during the breeding season (Cohen et al. 2020). We continued this tracking rotation throughout the breeding season weekly (15 April–15 August 2021–2022). We were focused on roost site selection of individuals and did not collect roosting data on birds once they entered reproductive activities (i.e., nesting, brooding). Further, we did not collect data on individuals who were found dead in the same location or moved by a predator the following day.
To account for telemetry error among observers, we placed a radio collar in a randomly selected location at B. Bryan Farms and used homing techniques via an uninformed telemetry operator to locate the collar from a point at which they felt would not flush the bird (approximately 25–50 m). They walked to where they would have marked the bird’s location on a map and then flagged the location. We then measured the distance (m) from the “marked” location to the actual position of the radio collar and recorded the distance. This process was completed four times for each observer. We averaged error distances for each individual observer and overall to determine an error radius. We implemented this error radius (14m ± 3.2 m) as a buffer for “used” resources.
We considered roost selection at the third-order (microhabitat) scale as use within an individual’s average daily core area during the breeding season (6.3 ha) based on Cohen et al. (2020). We surveyed vegetation at all individual bobwhite roost site locations and four random points per location (1:4 ratio) to measure availability. We defined third-order availability as the proportion of each vegetation type within the average daily core area of an individual and use (i.e., selection) as the proportion of individual roost locations within each vegetation type (Chamberlain et al. 2000, Terhune et al. 2006). We took random point measurements to determine availability on the same day as selected roost site measurements by dropping a 1 m² Daubenmire frame in each cardinal direction from the estimated roost site center point at a random distance between 14 m (based on observer error radius) and 140 m (Cohen et al. 2020). We then randomly tossed a 1 m² Daubenmire frame three times within 14 m of the estimated used roost site center point to determine selection. We used Daubenmire classes to determine the average predominant vegetation cover for the selected and available roost sites (Stromberg 1990). We recorded Daubenmire classes as (0 = 0 | 1 = 0–5 | 2 = 5–25 | 3 = 25–50 | 4 = 50–75 | 5 = 75–95 | 6 = 95–100%) coverage in each frame such that the components of each frame sum to 100% (Taylor et al. 1999). Substrate classes were listed as: bare ground, litter cover (i.e., dead plant material), forbs, native grasses, non-native grasses, and shrubs/woody stems (Taylor et al 1999; Table 1). Average vegetation height (excluding woody plants > 2 m tall) at each point was recorded by averaging the height (cm) of three randomly selected plants within the Daubenmire frame (Masters et al. 2009). We used a spherical densiometer to quantify percent canopy closure when necessary, and a Robel pole (Robel et al. 1970) to measure the VOS (i.e., height at which 100% visual obstruction stop) at each point. We took an observation at each roost/available site at 4 m from the Robel pole in each cardinal direction and recorded the mean of the four observations at each site as the VOS for the point (Robel et al. 1970, Taylor et al. 1999, Martin 2010).
Statistical analysis
We modeled breeding season roost site selection in a logistic regression framework where selected and available locations represented response variables (used = 1, available = 0). We constructed a generalized linear mixed model (GLMM) in package glmmTMB in program R (Boyce et al. 2002, Brooks et al. 2017, R Core Team 2019) to model the effects of vegetative covariates on third-order breeding season roost site selection. We treated individuals as a random effect to account for repeated measures of individuals and variation among individuals (Gillies et al. 2006). We first scaled all continuous explanatory covariates because measurement units varied among covariates. We then tested for collinearity among predictor covariates using Pearson’s correlation coefficient and set the maximum threshold to 0.6 and minimum to -0.6. We removed the covariate from the model that would be most difficult to alter through vegetation management if there was a strong correlation between two variables shown by an exceeding threshold value (i.e., VOS; Shrestha 2020, Rosche et al. 2021). Following tests of collinearity, we tested a set of a priori candidate models that incorporated a combination of covariates to explain bobwhite third-order breeding season roost site selection (see Appendix 1). We used Akaike’s Information Criterion corrected for small sample sizes (AICc) to rank competing models (Burnham and Anderson 2002). We considered models with a ΔAICc ≤ 2 most competitive in explaining third-order breeding season roost site selection (Burnham and Anderson 2002).
RESULTS
We captured a total of 61 birds between 2021 and 2022, collaring 28 birds (14 female, 14 male) in 2021 and 30 birds (13 female, 17 male) in 2022 (N = 58 radio-collared birds). We did not collar 3 of the 61 birds because they had either a broken wing or a significant loss of head or neck feathers. Some radio-collared birds (i.e., 7 birds per year) were not surveyed because of radio-collar failure or inability to locate the bird prior to the breeding season survey period (N = 34). We recorded 1355 locations in 2021 and 595 locations in 2022. Average number of locations per bird in 2021 was 62 ± 7.4 (SE) and 28 ± 5.4 (SE) in 2022. Average time on air for birds in 2021 was 87.4 ± 14.8 (SE) days and 81.8 ± 23.8 (SE) in 2022.
We had a total of seven observers between 2021 and 2022 collecting third-order roost site selection vegetation data. A total of 163 roost sites were measured in 2021 for 20 birds, and 159 roost sites were measured in 2022 for 14 birds. The average number of roost locations per bird in 2021 was 8.15 ± 0.95 (SE) and 12 ± 2.1(SE) in 2022. Results of Pearson correlation analysis indicated that native grass, non-native grass, and average vegetation height exceeded the maximum threshold (0.6, -0.6), which prevented us from comparing those covariates in the same models. We evaluated a set of 43 a priori (see Appendix 1) candidate models of which only two models were considered competitive in explaining bobwhite third-order breeding season roost site selection. The most competitive model (Avg_Height + Bare_Ground + Litter + Litter^2, ΔAIC=0.00, AICcWt = 0.67), indicated that the probability of an individual selecting a roost site is expected to increase linearly with average vegetation height (Avg_Height, β = 0.13377, 95% CI = 0.05, 0.22; Fig. 1) and quadratically with litter coverage (Litter, β = 0.25479, 95% CI = 0.12, 0.39; Litter^2, β = -0.09606, 95% CI = -0.16, -0.04; Fig. 2). Selection was also expected to decrease linearly with bare ground (B_Ground, β = -0.20938, 95% CI = -0.31, -0.11; Fig. 3). The second-best approximating model (Avg_Height + Avg_Height^2 + Bare_Ground + Litter + Litter^2, ΔAIC = 1.88, AICcWt = 0.26) retained all the previous covariates but added the quadratic term for average vegetation height (Avg_Height^2, β = -0.01005, 95% CI = -0.06, 0.04), suggesting support for a parabolic relationship between average vegetation height and probability of selection. However, the precision of the quadratic estimate for Avg_Height^2 was non-informative. The top two models explained 94% of the cumulative weight to predict the observed data. We did not find sufficient evidence to support relationships among selection and forb, native grass, and non-native grass coverages based on model selection results.
DISCUSSION
We predicted that bobwhite would show a parabolic relationship with VOS and average vegetation height to optimize the trade-off between concealment and unobstructed flight paths for escape from nocturnal predators (Tillman 2009, Perkins et al. 2014) but had high uncertainty in our supporting model because of poor precision. We removed VOS because of collinearity with average vegetation height but found that bobwhite selected for greater average vegetation height at roost sites. Our second-best model showed support for a parabolic relationship between probability of selection and average vegetation height, which did meet our prediction. We predicted that bobwhite would show a parabolic relationship with bare ground, but our models indicated that probability of selection decreased with increasing bare ground. Our prediction that bobwhite would show a parabolic relationship with litter cover was met.
Selection for a roost site with greater average vegetation height may be a survival strategy of bobwhite that roost individually during the breeding season. Although, we did have some radio-collared males and females roost together periodically. Alternatively, we reason that coveys may be able to roost in shorter vegetation with lower VOS (i.e., sparsely vegetated), during the non-breeding season because of increased vigilance provided by multiple individuals roosting together in a covey (Klimstra and Ziccardi 1963, Tillman 2009, Perkins et al. 2014). However, individuals may require taller vegetation, greater VOS, greater litter coverage, and lesser bare ground for better concealment from nocturnal predators when they are roosting individually during the breeding season or are constrained by limited mobility (i.e., brooding; Kubečka et al. 2021). We encourage future researchers to investigate the presence of a parabolic relationship between vegetation height and breeding season roost site selection to better understand what range of vegetation height is most selected for by roosting bobwhite during the breeding season. Additionally, it would be beneficial for managers to understand how the height of vegetation at a roost site may influence survival during the breeding season. A better understanding of the scope of this relationship may help managers create a landscape with greater interspersion of differing vegetation height required by bobwhite at different life stages. For example, early spring prescribed burns facilitate foraging efficiency for broods and increase arthropod abundance but may also remove vegetation required for concealment from predators during roosting (Hurst 1972, Burke et al. 2008, Kubečka et al. 2021). Thus, providing a mosaic of vegetational components, in terms of both configuration and dispersion, is pertinent for population persistence as it allows individuals to consider trade-offs among cover options for brooding, nesting, or roosting during the breeding season.
Despite being one of the most studied avian species in the United States, research on bobwhite roosting ecology during the breeding season has yet to be evaluated. Failure to fully understand the ecology of a species throughout its annual cycle could pose challenges to long-term viability (Marra et al. 2015). Unlike Kubečka et al. (2021) who investigated roost site selection of broods during the breeding season, we were focused on roost site selection of individuals and did not collect data on birds once they entered reproductive activities (i.e., nesting, brooding). We acknowledge our sample size (N = 34) and a single study area limits our inferential power but suggest that our results represent a novel contribution to bobwhite ecology that hopefully encourages new research questions.
Our study had similar results to Chamberlain et al. (2002) who investigated roost site selection of coveys in Missouri and found that average vegetation height, VOS, and litter coverage was greater or higher at selected roost sites. Alternatively, Perkins et al. (2014), observed coveys selecting roost sites with lower VOS and a greater angle of obstruction (angle to nearest non-grass vegetation). Klimstra and Ziccardi (1963) found 60% of covey roosts on bare ground and 31% of roosts on duff (litter). Nearly all studies that investigated roost site selection by coveys found that selection was greatest for sparsely vegetated (low stem density), early successional vegetation (Stoddard 1931, Klimstra and Ziccardi 1963, Rosene 1969, Wiseman and Lewis 1981, Hiller and Guthery 2005, Perkins et al. 2014). Tillman (2009) studied nocturnal roosting behavior of Gray Partridge (Perdix perdix) coveys during the winter (November–February) and speculated that roosting in an exposed area was a type of predator avoidance as it allowed for unobstructed clear flight paths for escape if disturbed by a predator. Perkins et al. (2014) reasoned that this may be similar for bobwhite. Kubečka et al. (2021) found that brood roost sites consisted of more woody cover and visual obscurity. They also found that roosts had less grass and bare ground compared to other available areas, but broods were more likely to use sparser cover at older ages.
Our study is the first to investigate individual bobwhite breeding season roost site selection and although we reported some interesting findings, it is important to note that roost site selection likely varies spatially. Our findings may not be applicable among differing vegetation communities within the bobwhite range. Birds on our study area selected for greater vegetation height, greater litter coverage, and less bare ground, but birds may select for different vegetational components in other areas across the bobwhite range. For example, bobwhite in a more arid, rangeland landscape in Texas may have to consider different trade-offs in selecting a roost site during the breeding season. As well, bobwhite in areas with less naturalized grass species (Paspalum spp., Festuca arundinacea, Setaria spp.) may have more bare ground available.
CONCLUSION
Management implications
Managers already focus on improving important habitat requirements for bobwhite (i.e., nesting cover, brood rearing cover, shrub cover, etc.), but few consider breeding season roost cover, which accounts for approximately 5 months of the bobwhite annual cycle. Bobwhite managers must consider all conditions of the environment and habitat requirements among all seasons to understand population persistence, including breeding season roost cover. Managers should focus not only on resource requirements for nesting, brood rearing, and roosting during the fall-winter, but also incorporate efforts on providing components necessary for roosting in the spring and summer when an individual bird is not incubating a nest and may be vulnerable to predation. Gaining a better understanding on bobwhite roost site selection during the breeding season will help managers determine how to provide the best arrangement of vegetation components for bobwhite to be successful throughout their life cycle. We hesitate to make any habitat management recommendations beyond what is consistently suggested for bobwhite (i.e., improving early successional vegetation composition through use of prescribed fire, disking, controlled grazing etc.) because this study was more of an initial exploratory analysis on habitat components and because of our high uncertainty associated with the strength of our estimates due to poor precision. Future studies should continue to investigate roost site selection across the bobwhite range to further understand vegetational components and potential thermal benefits of roost sites selected by individual bobwhite during the breeding season. Furthermore, breeding season roost site selection likely has implications for both the near-term survival and long-term fitness of bobwhite, but we have yet to examine this potential relationship. We will enhance our ability to manage and conserve bobwhite populations with continued research focused on exploring vegetational components required during all life stages, in terms of both configuration and dispersion, across the bobwhite range.
RESPONSES TO THIS ARTICLE
Responses to this article are invited. If accepted for publication, your response will be hyperlinked to the article. To submit a response, follow this link. To read responses already accepted, follow this link.
ACKNOWLEDGMENTS
This research was supported by the Bryan Endowment for Bobwhite Habitat Restoration, the James C. Kennedy Endowment for Upland Gamebird Ecology, and the Mississippi Agriculture and Forestry Experiment Station. We also thank the Mississippi State University system, the College of Forest Resources, and the Department of Wildlife, Fisheries, and Aquaculture. We are extremely grateful to Mr. Jimmy Bryan for his willingness to provide land access and assistance for our fieldwork. We also acknowledge Bennie Atkinson, Corey Cantrell, and Cameron Cantrell for project assistance in the field. We also thank the undergraduate and graduate students who helped with data collection. Lastly, we thank the associated editor and reviewers for their contribution and suggestions to improve this manuscript.
DATA AVAILABILITY
The data/code that supports the findings of this study are openly available upon request from the authors.
LITERATURE CITED
Boyce, M. S., and L. L. McDonald. 1999. Relating populations to habitats using resource selection functions. Trends in Ecology and Evolution 14:268-272. https://doi.org/10.1016/S0169-5347(99)01593-1
Boyce, M. S., P. R. Vernier, S. E. Nielsen, and F. K. A. Schmiegelow. 2002. Evaluating resource selection functions. Ecological Modelling 157:281-300. https://doi.org/10.1016/S0304-3800(02)00200-4
Brooks, M. E., K. Kristensen, K. J. van Benthem, A. Magnusson, C. W. Berg, A. Nielsen, H. J. Skaug, M. Mächler, and B. M. Bolker. 2017. glmmTMB balances speed and flexibility among packages for zero‐ inflated generalized linear mixed modeling. R Journal 9(2):378-400. https://doi.org/10.32614/RJ-2017-066
Burger, L. W., T. V. Dailey, M. R. Ryan, and E. Kurzejeski. 2017. Effect of temperature and wind on metabolism of Northern Bobwhite in winter. Proceedings of the National Quail Symposium 8:80. https://doi.org/10.7290/nqsp08xolv
Burke, J. D., M. J. Chamberlain, and J. P. Geaghan. 2008. Effects of understory vegetation management on brood habitat for Northern Bobwhites. Journal of Wildlife Management 72:1361-1368. https://doi.org/10.2193/2007-290
Burnham, K. P., and D. R. Anderson. 2002. Model selection and multimodal inference. Springer, New York, New York, USA.
Chamberlain, E., R. D. Drobney, and T. V. Dailey. 2002. Winter macro- and microhabitat use of winter roost sites in central Missouri. Proceedings of the National Quail Symposium 5:25. https://doi.org/10.7290/nqsp05zshb
Chamberlain, M. J., B. D. Leopold, and L. W. Burger. 2000. Characteristics of roost sites of adult Wild Turkey females. Journal of Wildlife Management 64:1025-1032. https://doi.org/10.2307/3803213
Cohen, B. S., D. D. Marquardt, N. W. Bakner, R. M. Perez, and B. A. Collier. 2020. Daily movements, space use, and habitat selection of GPS‐tagged Northern Bobwhite in Texas. Wildlife Society Bulletin 44:790-797. https://doi.org/10.1002/wsb.1137
Gillies, C. S., M. Hebblewhite, S. E. Nielsen, M. A. Krawchuk, C. L. Aldridge, J. L. Friar, D. J. Saher, C. E. Stevens, and C. L. Jerde. 2006. Application of random effects to the study of resource selection by animals. Journal of Animal Ecology 75:887-898. https://doi.org/10.1111/j.1365-2656.2006.01106.x
Hiller, T. L., and F. S. Guthery. 2005. Microclimate versus predation risk in roost and covert selection by bobwhites. Journal of Wildlife Management 69:140-149. https://doi.org/10.2193/0022-541X(2005)069<0140:MVPRIR>2.0.CO;2
Hurst, G. A. 1972. Insects and bobwhite quail brood habitat management. Proceedings of the National Quail Symposium 1:13. https://doi.org/10.7290/nqsp012hxa
Janke, A. K., and R. J. Gates. 2013. Home range and habitat selection of Northern Bobwhite coveys in an agricultural landscape. Journal of Wildlife Management 77:405-413. https://doi.org/10.1002/jwmg.461
Johnson, D. H. 1980. The comparison of usage and availability measurements for evaluating resource preference. Ecology 61:65-71. https://doi.org/10.2307/1937156
Klimstra, W. D., and V. C. Ziccardi. 1963. Night-roosting habitat of bobwhites. Journal of Wildlife Management 27:202-214. https://doi.org/10.2307/3798399
Kroeger, A. J., C. S. Deperno, C. A. Harper, S. B. Rosche, and C. E. Moorman. 2020. Northern Bobwhite non‐breeding habitat selection in a longleaf pine woodland. Journal of Wildlife Management 84:1348-1360. https://doi.org/10.1002/jwmg.21925
Kubečka, B. W., T. M. Terhune, and J. A. Martin. 2021. Temporal and scalar variations affect resource use of Northern Bobwhite broods. Ecology and Evolution 11:14758-14774. https://doi.org/10.1002/ece3.8161
Marra, P. P., E. B. Cohen, S. R. Loss, J. E. Rutter, and C. M. Tonra. 2015. A call for full annual cycle research in animal ecology. Biology Letters 11:20150552. https://doi.org/10.1098/rsbl.2015.0552
Martin, J. A. 2010. Population and landscape ecology of Northern Bobwhites on Florida rangelands. Dissertation. University of Georgia, Athens, Georgia, USA.
Masters, R. E., F. S. Guthery, W. R. Walsh, D. S. Cram, and W. G. Montague. 2009. Usable space versus habitat quality in forest management for bobwhites. Proceedings of the National Quail Symposium 6:21. https://doi.org/10.7290/nqsp06iq8k
McGrath, D. J., T. M. Terhune II, and J. A. Martin. 2018. Vegetation and predator interactions affect Northern Bobwhite behavior. Journal of Wildlife Management 82:1026-1038. https://doi.org/10.1002/jwmg.21441
National Centers for Environmental Information (NCEI). 2023a. Global summary of the month for 2021. NCEI, Washington, D.C., USA. https://www.ncei.noaa.gov/maps/monthly/
National Centers for Environmental Information (NCEI). 2023b. Global summary of the month for 2022. NCEI, Washington, D.C., USA. https://www.ncei.noaa.gov/maps/monthly/
Perkins, R., C. Boal, D. Rollins, and R. M. Perez. 2014. Northern Bobwhite predator avoidance behavior in response to varying types of threat. Journal of Wildlife Management 78:1272-1281. https://doi.org/10.1002/jwmg.766
R Core Team. 2019. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria.
Robel, R. J., J. N. Briggs, A. D. Dayton, and L. C. Hulbert. 1970. Relationships between visual obstruction measurements and weight of grassland vegetation. Journal of Range Management 23:295-297. https://doi.org/10.2307/3896225
Rosche, S. B., C. E. Moorman, A. J. Kroeger, K. Pacifici, J. G. Jones, and C. S. Deperno. 2021. Effects of prescribed fire on Northern Bobwhite nesting ecology. Wildlife Society Bulletin 45:249-257. https://doi.org/10.1002/wsb.1183
Rosene, W. 1969. The bobwhite quail: its life and management. Rutgers University Press, New Brunswick, New Jersey, USA.
Sandercock, B. K., W. E. Jensen, C. K. Williams, and R. D. Applegate. 2008. Demographic sensitivity of population change in Northern Bobwhite. Journal of Wildlife Management 72:970–982. https://doi.org/10.2193/2007-124
Seiler, T. P., R. D. Drobney, and T. V. Dailey. 2002. Use of weather variables for predicting fall covey calling rates of Northern Bobwhites. Proceedings of the National Quail Symposium 5:17. https://doi.org/10.7290/nqsp051e6d
Shirley, T. R., and A. K. Janke. 2021. Use of small, unmanned aircraft systems and mist nets to capture Ring-necked Pheasants. Wildlife Society Bulletin 45:725-730. https://doi.org/10.1002/wsb.1230
Shrestha, N. 2020. Detecting multicollinearity in regression analysis. American Journal of Applied Mathematics and Statistics 8:39-42. https://doi.org/10.12691/ajams-8-2-1
Stoddard, H. L. 1931. The bobwhite quail: its habits, preservation and increase. Charles Scribner and Sons, New York, New York, USA.
Stromberg, M. R. 1990. Habitat, movements, and roost characteristics of Montezuma Quail in southeastern Arizona. Condor 92:229-236. https://doi.org/10.2307/1368404
Taylor, J. S., K. E. Church, and D. H. Rusch. 1999. Microhabitat selection by nesting and brood-rearing Northern Bobwhite in Kansas. Journal of Wildlife Management 63:686-694. https://doi.org/10.2307/3802658
Terhune, T. M., D. C. Sisson, H. L. Stribling, and J. P. Carroll. 2006. Home range, movement, and site fidelity of translocated Northern Bobwhite (Colinus virginianus) in southwest Georgia, USA. European Journal of Wildlife Research 52:119-124. https://doi.org/10.1007/s10344-005-0015-1
The National Bobwhite Technical Committee. 2011. The National Bobwhite Conservation Initiative: The range-wide plan for recovering bobwhites. W. E. Palmer, T. M. Terhune, and D. F. McKenzie, editors. National Bobwhite Technical Committee Publication, ver. 2.0, Knoxville, Tennessee, USA.
Tillman, J. E. 2009. Fear of the dark: night-time roosting and anti-predation behaviour in the Grey Partridge (Perdix perdix L.). Behaviour 146:999-1023. https://doi.org/10.1163/156853908X398924
Walsberg, G. E. 1986. Thermal consequences of roost-site selection: the relative importance of three modes of heat conservation. Auk 103:1-7. https://doi.org/10.1093/auk/103.1.1
Wellendorf, S. D., W. E. Palmer, and P. T. Bromley. 2004. Estimating calling rates of Northern Bobwhite coveys and measuring abundance. Journal of Wildlife Management 68:672-682. https://doi.org/10.2193/0022-541X(2004)068[0672:ECRONB]2.0.CO;2
Wiley, M. J., A. K. Janke, and R. J. Gates. 2012. Efficacy of targeted mist-netting to capture Northern Bobwhites during the non-breeding season in Ohio. Proceedings of the National Quail Symposium 7:94. https://doi.org/10.7290/nqsp07xzo4
Williams, C. K., R. S. Lutz, and R. D. Applegate. 2003. Optimal group size and Northern Bobwhite coveys. Animal Behaviour 66:377-387. https://doi.org/10.1006/anbe.2003.2215
Williams, C. K., R. S. Lutz, R. D. Applegate, and D. H. Rusch. 2000. Habitat use and survival of Northern Bobwhite (Colinus virginianus) in cropland and rangeland ecosystems during the hunting season. Canadian Journal of Zoology 78:1562-1566. https://doi.org/10.1139/z00-081
Wiseman, D. S., and J. C. Lewis. 1981. Bobwhite use of habitat in tallgrass rangeland. Wildlife Society Bulletin 9:248-255.
Fig. 1
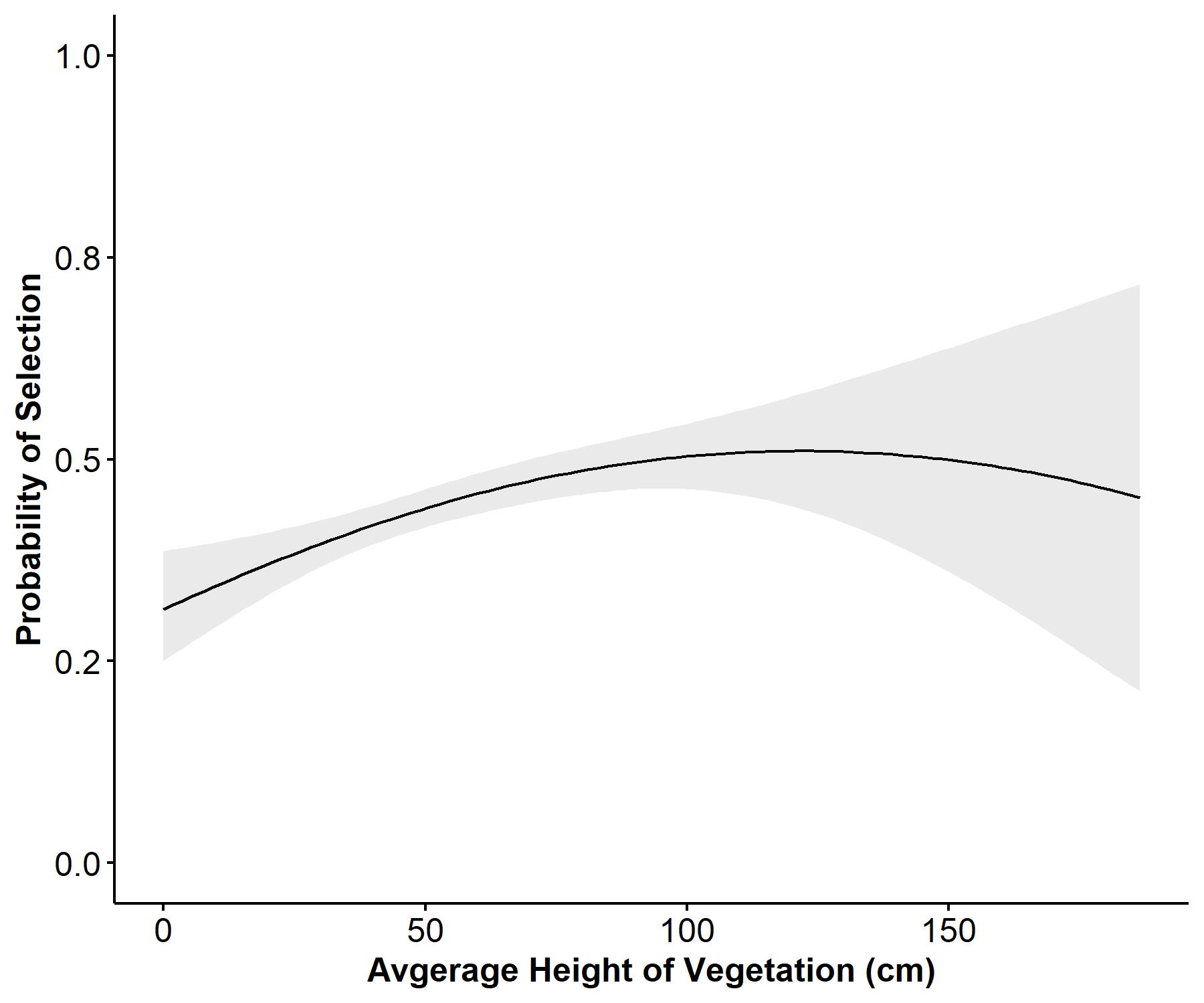
Fig. 1. Predicted probability of a breeding season (April–August) roost site being selected by a radio-collared Northern Bobwhite (Colinus virginianus) as a function of the average vegetation height (cm) at B. Bryan Farms, Clay County, Mississippi, USA, 2021–2022.

Fig. 2
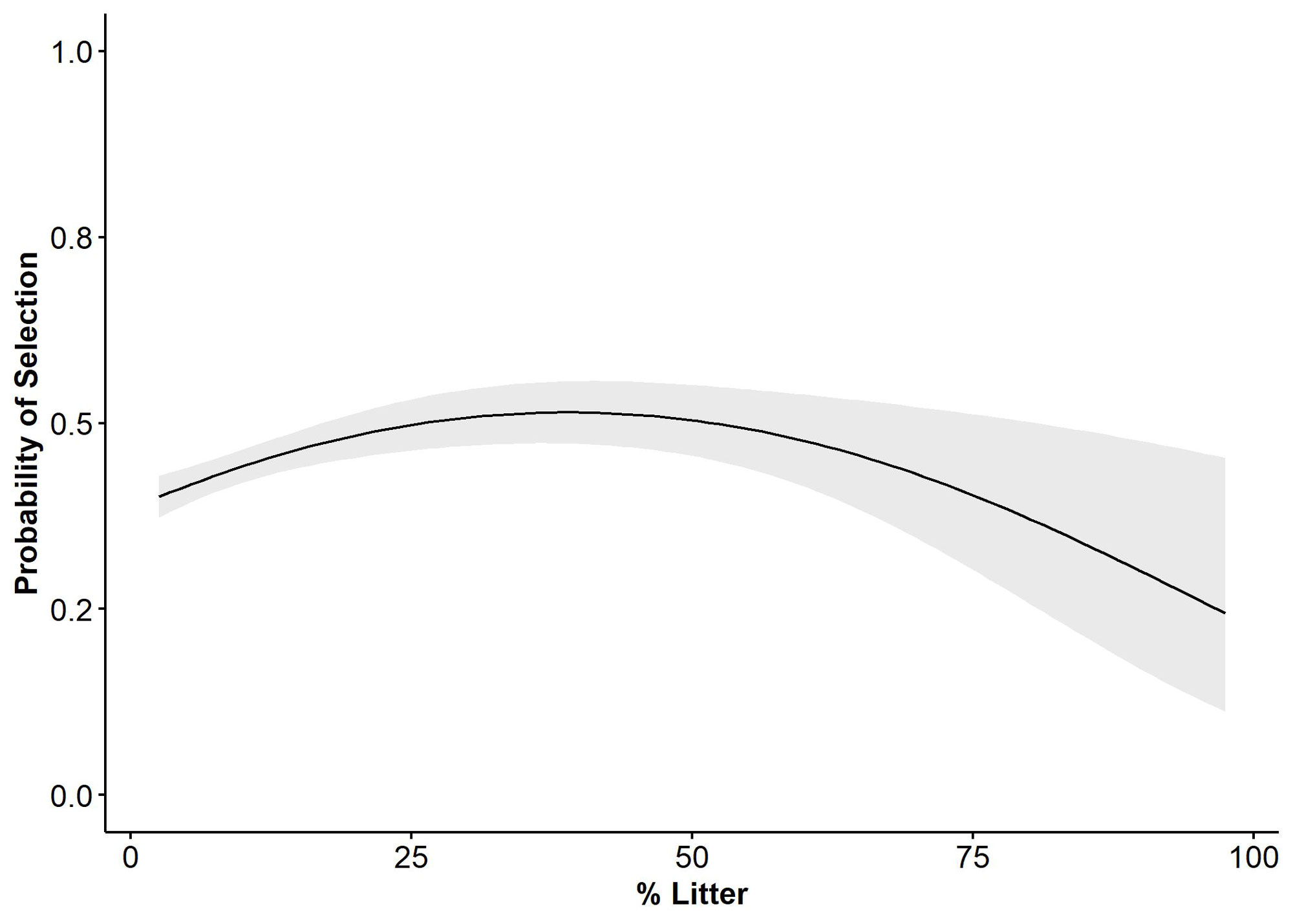
Fig. 2. Predicted probability of a breeding season (April–August) roost site being selected by a radio-collared Northern Bobwhite (Colinus virginianus) as a function of the percent coverage of litter at B. Bryan Farms, Clay County, Mississippi, USA, 2021–2022.

Fig. 3
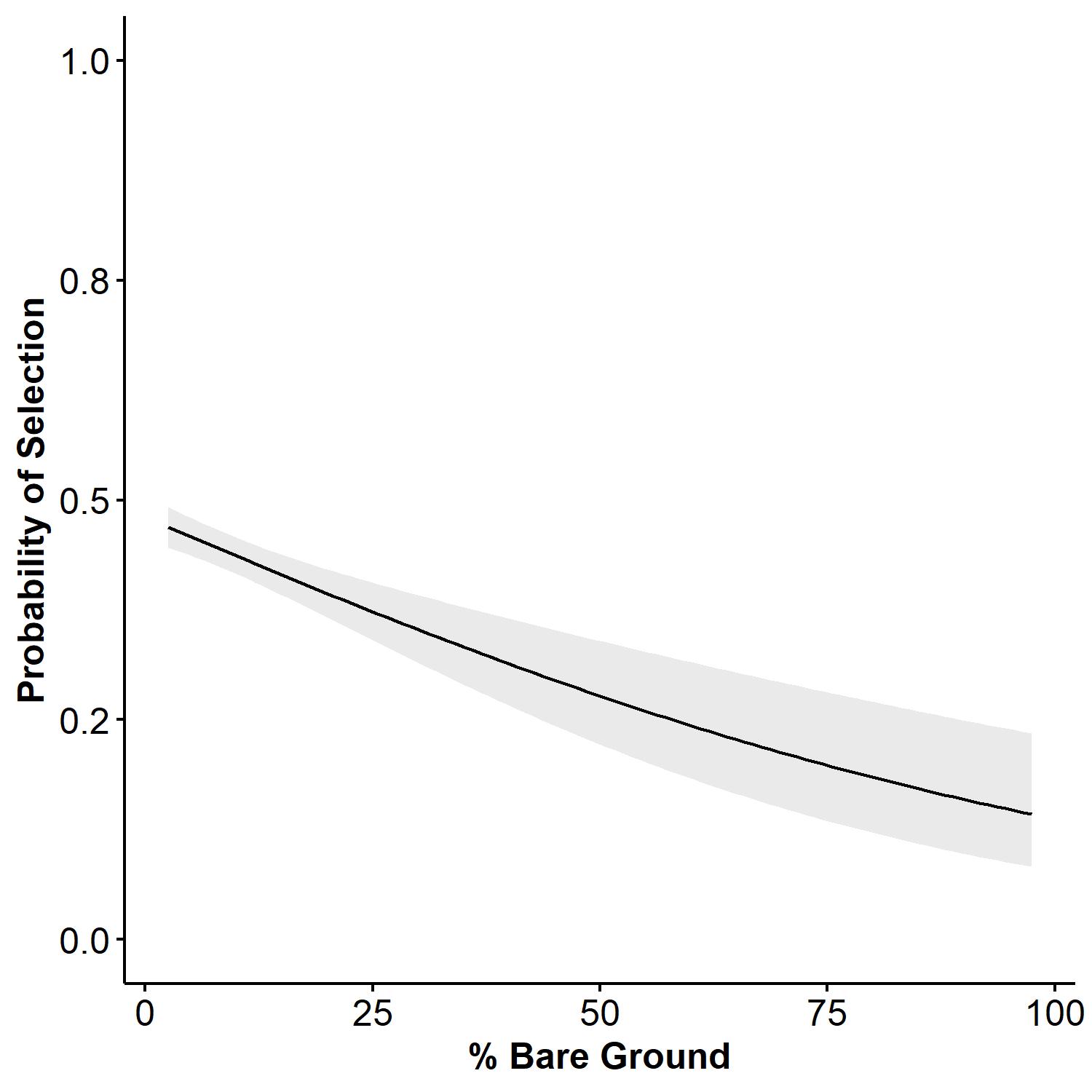
Fig. 3. Predicted probability of a breeding season (April–August) roost site being selected by a radio-collared Northern Bobwhite (Colinus virginianus) as a function of the percent coverage of bare ground at B. Bryan Farms, Clay County, Mississippi, USA, 2021–2022.

Table 1
Table 1. Covariates used to describe radio-collared Northern Bobwhite (Colinus virginianus) breeding season (April–August) third-order roost-site selection at B. Bryan Farms, Clay County, Mississippi, USA, 2021–2022.
| Abbreviation | Description | ||||||||
| Forb | Percent cover by forbs | ||||||||
| B_Ground | Percent cover by bare ground | ||||||||
| Litter | Percent cover by litter | ||||||||
| Native_G | Percent cover by native grass | ||||||||
| Nnative_G | Percent cover by any non-native grass | ||||||||
| Woody† | Percent cover by woody vegetation | ||||||||
| Canopy† | Percent canopy closure of woody vegetation ≥ 2 m utilizing densiometer | ||||||||
| Avg_Height | Average height (cm) of vegetation in 1 m x 1 m daubenmire frame based on 3 random plant measurements | ||||||||
| VOS‡ | Height (cm) at which 100% visual obstruction stops: average of 4 measurements taken 4 m from Robel pole in each cardinal direction | ||||||||
| Percent cover measurements were taken in a 1 m² Daubenmire frame based on Daubenmire cover classes. † Removed from roost-site selection analysis because of lack of category observation during data collection. ‡ Removed from roost-site selection analysis because of collinearity with Avg_Height. | |||||||||








