The following is the established format for referencing this article:
Díaz-Vallejo, M., S. Chaparro-Herrera, A. Lopera-Salazar, M. Castaño-Díaz, R. Correa, and J. Parra. 2023. Use of acoustic monitoring to estimate occupancy of the Antioquia Brushfinch (Atlapetes blancae), a critically endangered species, in San Pedro de los Milagros, Antioquia. Journal of Field Ornithology 94(2):4.ABSTRACT
The Antioquia Brushfinch (Atlapetes blancae) is a passerine bird from the Passerellidae family, endemic to the Santa Rosa de Osos high elevation plateau (ASRO) in the department of Antioquia-Colombia. It is threatened nationally and globally, presumably by the destruction of its habitat and small population size. The Antioquia Brushfinch was described in 2007 from three specimens collected in 1971 and was considered possibly extinct until its rediscovery in 2018. Despite the difficulty in obtaining information on its vital rates, some methodologies have recently been proposed to estimate population state variables, such as occupancy. Occupancy is a basic population state variable reflecting the probability that a sampling unit, taken at random from the study area, is occupied. In the present work, we sampled 80 sites (30 m radius circles) to monitor acoustic activity using autonomous recorders and estimated the probability of occupancy of the Antioquia Brushfinch in an area of the municipality of San Pedro de los Milagros, Antioquia. Based on our observations of habitat use by the species, we evaluated the following set of covariates for occupancy: the amount of shrubby and herbaceous vegetation, the presence of water bodies, and terrain concavity. For detection probability (the probability of recording an organism given that the site is occupied), we evaluated the time of each record as a covariate. The average occupancy and detection probability of the Antioquia Brushfinch across sampling units was 0.11 ± 0.06 SE and 0.1 ± 0.07 SE, respectively, indicating low occupancy and probability of detection. We found that areas with intermediate proportions of shrubby and herbaceous vegetation and concave terrain relief promote the occupancy of the Antioquia Brushfinch. The probability of obtaining acoustic records of this species with the set of autonomous recorders increased toward the morning hours. The information and results obtained will provide guidelines for conservation actions in the area of occupancy of the Antioquia Brushfinch.
RESUMEN
El Pinzón de Antioquia (Atlapetes blancae) es un ave paseriforme de la familia Passerellidae, endémica del altiplano de Santa Rosa de Osos (ASRO) en el departamento de Antioquia-Colombia. Está amenazada a nivel nacional y mundial, presumiblemente por la destrucción de su hábitat y el pequeño tamaño de su población. El Pinzón de Antioquia fue descrito en 2007 a partir de tres ejemplares colectados en 1971 y se consideró posiblemente extinto hasta su redescubrimiento en 2018. A pesar de la dificultad para obtener información sobre sus tasas vitales, recientemente se han propuesto algunas metodologías para estimar variables de estado poblacional, como la ocupación. La ocupación es una variable básica del estado de la población que refleja la probabilidad de que una unidad de muestreo, tomada al azar del área de estudio, esté ocupada. En el presente trabajo, muestreamos 80 sitios (círculos de 30 m de radio) para monitorear la actividad acústica utilizando grabadoras autónomas y estimamos la probabilidad de ocupación del Pinzón de Antioquia en un área del municipio de San Pedro de los Milagros, Antioquia. En base a nuestras observaciones de uso de hábitat por esta especie, evaluamos el siguiente conjunto de covariables para la ocupación: la cantidad de vegetación arbustiva y herbácea, la presencia de cuerpos de agua y la concavidad del terreno. Para la probabilidad de detección (la probabilidad de grabar un organismo dado que el lugar está ocupado), evaluamos el tiempo de cada grabación como una covariable. La probabilidad media de ocupación y detección del Pinzón de Antioquia en todas las unidades de muestreo fue de 0,11 ± 0,06 SE y 0,1 ± 0,07 SE, respectivamente, lo que indica una baja ocupación y probabilidad de detección. Se encontró que las áreas con proporciones intermedias de vegetación arbustiva y herbácea y relieve de terreno cóncavo promueven la ocupación del Pinzón de Antioquia. La probabilidad de obtener registros acústicos de esta especie con el conjunto de grabadoras autónomas aumentó hacia las horas de la mañana. La información y los resultados obtenidos darán pautas para acciones de conservación en el área de ocupación del Pinzón de Antioquia.
INTRODUCTION
Threatened species generally have small population sizes and restricted distribution ranges, which represents a challenge to obtain information on their vital rates (e.g., mortality, birth rate, among others; Beissinger and Westphal 1998). For example, in the Red Book of Birds from Colombia, about 142 species were cataloged within some category of threat, and of these, only 5 were categorized using information on abundance or vital rates, whereas the rest were defined based on the area of distribution and extent of occurrence (Renjifo et al. 2014, 2016). The difficulty in estimating population state variables has caused researchers to generate simpler alternatives such as occupancy (MacKenzie et al. 2005, 2018), which can be interpreted as the probability that a species is present in a sampling unit. Occupancy models, a type of hierarchical model (Kéry and Royle 2015), recognize that detection methods are imperfect, meaning the absence of records does not imply the absence of the species (Guillera-Arroita 2017, MacKenzie et al. 2018). In addition, these models can associate environmental and landscape variables with occupancy and detection, providing useful information that may improve guidelines for conservation (Devarajan et al. 2020).
The Antioquia Brushfinch (Atlapetes blancae) is a passerine bird from the Passerellidae family, endemic to the Santa Rosa de Osos high-elevation plateau (ASRO) in the department of Antioquia-Colombia (Fig. 1), and it is listed on the International Union for Conservation of Nature (IUCN) Red List as “critically endangered” (CR; BirdLife International 2021). Due to habitat destruction, small population size, and lack of detection for over 10 years, it was listed as “possibly extinct” (PE; Donegan 2007, Renjifo et al. 2014, Correa et al. 2019, BirdLife International 2021). Since the rediscovery of this species, multiple efforts were made to identify the places where it is present (Chaparro-Herrera and Lopera-Salazar 2019, Chaparro-Herrera et al. 2021, 2022), and recent observations have generated valuable information, including the description of its species-specific song and its vocal activity (Correa et al. 2019).
The Antioquia Brushfinch exhibits territorial behavior and includes a vocal display, especially between the months of January and May, where the only reproduction events have been recorded (Chaparro-Herrera and Lopera-Salazar 2019). Even though there is not quantitative information about territory size, studies on related species with similar ecologies indicate small territories (5000-20,000 m²; Oppel et al. 2003, Krabbe 2004). This suggests that the species is suitable for acoustic monitoring. This method constitutes an innovative, affordable, and efficient tool that minimizes the degree of intervention in situ and can generate data for occupancy models (Campos-Cerqueira and Aide 2016). The current situation of the Antioquia Brushfinch requires informed decision making aimed at its immediate protection, thus occupancy models have emerged as a viable strategy to generate useful information in this context (Clement et al. 2014, Campos-Cerqueira and Aide 2016, Balantic and Donovan 2019). We believe that the use of automated recorders to monitor endangered species can be a critical tool in their conservation because these recorders allow the investment of enormous sampling efforts to maximize detections with minimal disturbance and without observer biases. For example, Campos‐Cerqueira and Aide (2016) conducted occupancy models through acoustic monitoring in a population of the Elfin-woods Warbler (Setophaga angelae) in Puerto Rico and found that the use of these passive methodologies improved the detectability of this species.
Recent observations of the Antioquia Brushfinch indicate potential associations with patches of shrubby vegetation and low-canopy height forests (Chaparro-Herrera and Lopera-Salazar 2019, Valencia-C et al. 2019, Chaparro-Herrera et al. 2021), contrary to what was predicted when the species was originally described (e.g., large forest remnants, Donegan 2007), but consistent with what was reported in other species of the genus (regenerating vegetation in early successional stages; Oppel et al. 2004, Botero-Delgadillo et al. 2022a, b). Preliminary studies also suggest associations with water bodies and flooded vegetation (Proyecto Atlapetes, personal communication). The spatial scale at which these factors impact occupancy can be variable and establishing which scales are relevant for the species can be another important component in conservation decision making. In terms of detection, the time of day at which sampling occurs can be critical (Symes et al. 2022). Analysis of daily vocalization activity in tropical birds has found that the highest probability of observation is within a few hours after dawn (Arbeláez-Cortés et al. 2011; O. Laverde-R., P. Caycedo-Rosales, P. C. Pulgarín-R., and C. D. Cadena, unpublished manuscript). However, some visual observations from the Antioquia Brushfinch have occurred at different hours during the day (Valencia-C et al. 2019). In this study, we estimate occupancy of the Antioquia Brushfinch in a region within the municipality of San Pedro de los Milagros using acoustic monitoring. We evaluate the hypothesis that the area of shrubby and herbaceous vegetation, aspects of terrain relief, the presence of water bodies, and the spatial scale at which these variables are measured determines the species’ occupancy. This is the first effort to establish quantitative information about the distribution and occupancy patterns of this species, which we hope will be useful for conservation actions in the short term.
MATERIALS AND METHODS
Study area
The Santa Rosa de Osos high elevation plateau (ASRO) is located north of the Central Cordillera of the Andes, in the department of Antioquia, Colombia. It is limited to the south and east by the Medellín-Porce River canyon and to the west by the Cauca River canyon (Arias and González 2003; Fig. 1). We sampled the southeastern limit of the ASRO, close to the rediscovery site of the Antioquia Brushfinch (Correa et al. 2019). The study area (~44.0 km²) encompasses an elevational range between 2218 and 2849 m.a.s.l. with an annual monthly average temperature of 16 °C, with considerable variation within a day. The dominant vegetation cover in 2019 was clean pastures, i.e., permanent grasslands and herbs usually under grazing by domestic animals (58.2% of the total study area). This was followed by 20.2% of high secondary vegetation, 11.0% of weedy grasses, 6.2% of low secondary vegetation, 2.5% of crops, 2.2% of bare land dedicated mostly to livestock, and 1.3% of fragmented forest (UMATA 2019, Chaparro-Herrera et al. 2021). Despite being a plateau, the ASRO varies in slope percentages, ranging between 5.0% and 9.0%. This slope variability includes morphologies such as hills, depressions, and plains that determine the flow of water courses (Arias et al. 2000, Arias and González 2003, Arias 2007).
Experimental design
We used 20 AudioMoth recorders (v1.1.0) distributed randomly in 80 sites across our study area, including the 9 localities of the previously known records (Fig. 1B). At each sampling site, an automatic recorder was deployed and it recorded for seven consecutive days, and on the eighth day, the equipment and stored information were retrieved and moved to the next site. Each day of recordings was interpreted as a repetition, so each sampling site was “visited” repeatedly on seven occasions. The recorders were installed ~1.5 m from the ground, with a minimum distance of 380.0 m from each other to guarantee independence between sampling units. In cases where the sampling point was in the middle of a large grassland area, it was relocated to the closest vegetation cover, to maximize the probability of detection. To assess the acoustic reach of the recorder, we conducted experiments playing a taped song at a reference volume, at 10 m intervals up to 50 m distance from the recorder within high secondary vegetation cover. After visually inspecting the sonograms of the recordings, we concluded that the maximum distance at which songs could be identified visually and through hearing for our study area was 30 m. Therefore, a sampling unit was defined as a circular area with a radius of 30 m, containing 2827.4 m². To protect the recorders, we built plastic containers with a hermetic lid and a hole in the back where the microphone was located. We protected the hole with a hydroponic cloth (Scosche-SGCWP) that allows sound to enter but prevents water and moisture from entering the interior of the recorder. Each recorder was configured with a 32.0 kHz sampling rate, medium gain, and a cyclic recording period of 60 s every 900 s (15 min) between 05:00 and 18:30 h. All the fieldwork was conducted between April and June 2021 for a total period of 49 days, covering part of the breeding season of the species (Chaparro-Herrera and Lopera-Salazar 2019). All recordings were aurally and visually inspected using Raven Pro 1.6.1 software (Center for Conservation Bioacoustics 2019). The sampling unit for occupancy analysis was defined as the combination of each site for one day, so any day in which there was at least one recording of the Antioquia Brushfinch vocalizations, was included in the database as a presence record (1), and any day when there were no vocalizations of the species of interest, was coded as not recorded (0).
Occupancy models
Time was proposed as the only detection covariate and five occupancy covariates were included: area with shrubby vegetation, average canopy height, the second derivative of the terrain, slope, and the presence of water bodies. The time of day was represented by a number from 1 (5:00) to 51 (18:30). In this way, on the days in which there were detections, we assigned the number corresponding to the time of detection. In the event that there was more than one record in a day, we chose the time of the first registration, and on days without vocalizations, we established a random value between 1 and 51. For each site, two spatial scales were used to quantify each covariate, a circular polygon of 50 and another of 500 m radius. The area in square meters of shrubby vegetation was obtained from a land cover layer developed using the Corine Land Cover methodology based on satellite images of 5 m spatial resolution for the year 2019 (UMATA 2019). Eight land use coverages were defined: fragmented forest, crops, weedy grasses, clean pastures, urban areas, bare lands, as well as high and low secondary vegetation (Fig. 1B). Based on documented sightings and field observations of the Antioquia Brushfinch (Chaparro-Herrera and Lopera-Salazar 2019, Correa et al. 2019, Valencia-C et al. 2019, Chaparro-Herrera et al. 2021), we reclassified this layer, combining all the areas of low secondary vegetation and weedy grasses to represent what we call the “shrub and herbaceous vegetation” category (SHV). The average canopy height (CH) was obtained from a 25 m resolution layer developed by the Ecosystem Dynamics Research Center (GEDI; Patterson and Healey 2015). The average of the second derivative of terrain (SDT) and the slope were obtained from the Geomorph90m database at a spatial resolution of 90 m (Amatulli et al. 2020). The slope percentage represents the rate of change in elevation in the direction of the water flow (e.g., the percent change in meters that occurs along a 100 m stretch), and the SDT is the rate of change of the slope and represents the curvature or concavity of the ground. When SDT is positive, it represents a convex surface, and when it is negative a concave surface (Amatulli et al. 2020). We calculated the length of water courses present within each polygon with a layer of surface watersheds (IGAC 2019).
Model analysis
We used the R package “unmarked” to generate the occupancy models (Fiske and Chandler 2011, R Core Team 2021). We standardized all variables before running the models and performed a Spearman correlation (Spearman 1904) among them to detect multicollinearity. We structured the models using the sequential model strategy, where the best detection (p) submodel is first identified holding occupancy (Psi) constant [p(covariates)~Psi(.)], and then, occupancy model candidates are evaluated using the best detection submodel [p(best)~Psi(covariates)] (Andrade-Ponce et al. 2021). Two models for detection and 27 occupancy models (Table 1) were proposed based on prior knowledge of the species and preliminary results, including models with quadratic relationships. We chose the best model following the Akaike information criterion corrected for sample size (AICc; Akaike 1998). Models with a delta AICc greater than two units were considered to represent a relatively poor fit to the data. If two models had a similar fit (< 2 AICc units) and the comparison involved a simple (one variable) and a complex model (more than two variables including that of the simple model), we chose the more parsimonious model as the best. To check the fit of the model, we performed a goodness-of-fit test for single-season occupancy models based on the Pearson chi-square distribution (MacKenzie et al. 2018). This test also estimates the overdispersion of the data in the model (c-hat parameter), where values close to 1 represent a good fit to the data and greater than 1, overdispersion (Burnham and Anderson 2002). Finally, we made a spatial projection of the best model in raster format at a spatial resolution of 53 m to approximate the area of the prediction with the area of the sampling unit (2809.0 m²) using the “terra” (Hijmans 2021) and unmarked (Fiske and Chandler 2011) packages.
RESULTS
We obtained 51, 1-minute recordings each day, 357 per site, and a total of 28,560 across sites; the Antioquia Brushfinch was detected in 17 (0.06%), corresponding to 7 sites (naive occupancy ~9.0%). Five new localities are reported for the species (12, 33, 38, 52, 70; Fig. 2), and two with previous records (51 and 69). Detections came from recorders located in three types of coverage: fragmented forest (5), high secondary vegetation (6), and low secondary vegetation (6). Most of the detections were through songs (13) and on four occasions they were based on calls (Table 2). In five sites with previous records, we did not detect the species (34, 50, 55, 60, 68), and in one site where we did not detect it through recordings, it was observed during the installation of the recorders (55; Fig. 2).
The presence records were in sites with SHV areas above the average for all points (mean SHV in presence sites = 3101 m², SD = 1449), with greater slopes (8.3%), in more concave terrain, and with lower canopy heights (mean in presence sites = 12.9 m, SD = 2.7) than the average for all recorders (Fig. 3). No correlation was found among the proposed occupancy covariates (Appendix 1). The best detection submodel indicated a decrease with the time of day (AICc = 97.27), followed by a null model that did not include covariates (AICc = 108.57, ΔAICc = 11.30; Table 1, Fig. 4). The best occupancy model suggested that sites with intermediate SHV (a quadratic relationship) measured in a 50 m radius circle, and negative values of the SDT (concave terrain) measured in a 500 m radius circle promoted occupancy (AICc = 83.55; Table 1, Fig. 4). The best model presented a negative relationship with SDT (Coefficient [logit scale] = -1.63, P = 0.03) and a marginally significant quadratic relationship with SHV (Coefficient [linear term] = 4.93, P = 0.06; Coefficient [squared term] = -2.67, P = 0.08; Table 3). The Antioquia Brushfinch occupancy is favored in more concave terrain with intermediate extensions of SHV (Fig. 4A-B). The models that included vegetation aspects (SHV and canopy height) showed a better fit in reduced extensions (50 m), while the variables associated with topography (SDT and slope of the terrain) showed a better fit when measured at wide extensions (500 m; Table 3).
The goodness-of-fit test for the best model showed no evidence of overdispersion (c-hat = 0.9, P = 0.384). The best occupancy model indicated suitable sites close to the limits with the municipalities of Bello, Copacabana, and Don Matías (Fig. 2). The average detection probability across occupied sampling units (with detections) was 0.1 ± 0.07 SE; the average occupancy across the 80 sampled sites was 0.11 ± ;0.065 SE, and the average occupancy across the study area in the municipality of San Pedro de los Milagros 0.02 ± 0.016 SE. The latter had a strong asymmetric distribution with only 0.7% of the area with an occupancy probability greater than 0.5, while 58.57% of the area presented occupancy probabilities below 0.4.
DISCUSSION
Our results suggest that the occupancy of the Antioquia Brushfinch in our study area is low (total average in our survey sites ~0.11 and ~0.02 in all the study area) and confirm the assumptions about the reliance of this species in shrub cover instead of tree cover, as was considered by Donegan (2007) and Donegan et al. (2009). This is consistent with what has been reported for other endangered species of the genus such as the Yellow-headed Brushfinch (Atlapetes flaviceps; Botero-Delgadillo et al. 2022a, 2022b, Chaparro-Herrera et al. 2020) and Pale-headed Brushfinch (Atlapetes pallidiceps; Oppel et al. 2004). In addition to the importance of vegetation cover, our results corroborate the significance of topography, specifically the terrain profile, indicating that occupancy is favored in concave terrain in which water tends to accumulate and thus may have repercussions for the type of vegetation present (Mahe et al. 2005). Even though our results provide evidence for the utility of acoustic monitoring to track presence sites for the species by recording seven new sites, it also allows us to suggest some changes in the sampling scheme for future similar studies. Under our sampling design, acoustic detection probabilities were low, and Antioquia Brushfinch was not detected at five of seven known sites. The results of this study provide quantitative guidelines about the habitat characteristics occupied by this species and indicate that even within its range, the species is not common. For example, if we take the entire study area, less than 5.0% would have occupancy probabilities above 0.1 and about 0.6% of the area (46,110 m²) would have probabilities above 0.5 (Fig. 2). These results highlight the fragile situation of the species in the southern end of its distribution and invites all participants to raise awareness and take action to help protect the species and its habitat.
As we expected, the detection probability in the study area was high a few hours after dawn (Fig. 4C), as has been documented for most tropical birds (Arbeláez-Cortés et al. 2011). Our results suggest that the detection of the Antioquia Brushfinch was low (~0.1 on occupied sites with detections), suggesting either a low abundance in the surveyed area, a low vocal activity during the sampling period, or both. Early morning hours largely explained the variation in detection, consistent with previously made observations (Valencia-C et al. 2019) and with other occupancy studies in neotropical birds (Betancur et al. 2020). However, this might also be an artifact of choosing the first record of the day to assign the time of detection. Two vocal detections were obtained at noon and in the early afternoon (Table 1), and other studies have also reported vocal activity in the afternoon (Valencia-C et al. 2019). Some authors have suggested that for rare and elusive species with low detection probability, the number of survey events should be maximized instead of the number of sites, as increased survey events improve the probability of detecting more species (Guillera-Arroita et al. 2010, Shaw et al. 2022, Symes et al. 2022). Due to the lack of knowledge of the Antioquia Brushfinch and its daily vocal activity patterns, we used very short time intervals (1 min.) to increase the survey events during the day, however, we noted that increasing the time of recordings should also increase detection probabilities. The low detection probability could also be explained by the stage of the breeding season sampled. Our study was carried out in the last months of the breeding season (April and May; Chaparro-Herrera and Lopera-Salazar 2019), when competition and sexual display may be reduced (Chambert et al. 2012). Hence, given our results, we highlight and recommend considering longer recordings and short intervals to sample the start of the breeding season to maximize detectability. In fact, if possible due to battery and memory constraints, it could be ideal to sample continuously throughout the day and this would allow for a proper characterization of the species daily vocal activity pattern and the training of automatic detection algorithms.
The area with shrubby and herbaceous vegetation with the highest probability of occupancy (Fig. 4) ranged between 2000 and 6000 m² within a buffer of ~8000 m². In other words, buffers with a quarter to three-quarters of its total area covered with shrubby and herbaceous vegetation, promote the occupancy of this species. This suggests that the species does not occupy areas with a small extension of SHV (< 2000 m²), and that it favors some heterogeneity in vegetation but the uncertainty of the model prediction toward areas with uniform coverage of SHV was high (> 6000 m²; Fig. 4). The importance of this type of cover emphasizes the level of threat for this species in the face of the transformation rate of landscapes in San Pedro de los Milagros (Gobernación de Antioquia, Ministerio de Ambiente 2016). Personal observations also indicate that replacements of crop varieties and transformations from cattle farming to agriculture and vice versa are fast and often unpredictable, evidencing the need for small-sized reserves across this region. Thus, one of the recommendations from this study is to promote the conservation of thickets, low secondary vegetation, and fragments in succession (e.g., through remote sensing techniques). These vegetation covers are not only important for the occupancy of the Antioquia Brushfinch but also for other species that co-occur in these ecosystems (e.g., Black-throated Flowerpiercer, Diglossa brunneiventris vuilleumieri). These types of vegetation are traditionally not included in conservation plans, which added to their fragmented and isolated status, leaves this critically endangered species in a worrisome situation and without protection.
Occupancy of the Antioquia Brushfinch increases in concave terrain despite the topographic homogeneity of the ASRO. It is possible that this relationship is associated with the presence of water bodies, or the accumulation of water (e.g., wetlands). Swampy or slightly flooded areas may promote shrub-type vegetation, also important for this species, and they may be the only shrub vegetation patches left due to inaccessibility. On the other hand, the effect of the occupancy variables changed with the measurement scale and was different for vegetation than for topographic relief. This result highlights the importance of recognizing multiple scales in occupancy models and, in general, in distribution models (Miguet et al. 2016). As expected, some variables such as vegetation cover were more informative at small extents, while other variables such as terrain profile were more informative at large extents, supporting results from other studies (Luoto et al. 2007, Jetz et al. 2012, Mertes and Jetz 2018).
Different investigations in the ASRO around the Antioquia Brushfinch have noted that one of the greatest threats to the species corresponds to deforestation due to the expansion of the livestock and agricultural frontier (Donegan 2007, Donegan et al. 2009, Chaparro-Herrera et al. 2021). This result reflects the critical situation of the species and emphasizes the need to implement livestock and agricultural practices that include vegetation corridors or fragments that connect isolated vegetation patches (Edwards et al. 2012, Gliessman 2002). In addition, the promotion of agroecosystems as a kind of production system would work as a conservation strategy, and it can be a useful tool in the recovery of the Antioquia Brushfinch populations. Other initiatives, such as monitoring the state of vegetation patches through time and space and identifying important conservation areas for this species, are urgently needed. For example, organizations, such as CuencaVerde, have dedicated part of their efforts to the protection and restoration of the vegetation associated with high-elevation water bodies. Our results provide guidelines to identify such areas and reveal the current occupancy state of the species in an important section of its distribution.
CONCLUSION
Occupancy models, through acoustic sampling, represent a viable alternative for monitoring rare species. Our results support previous information on the importance of shrubby and low-lying vegetation cover for occupancy of the Antioquia Brushfinch and highlight its problematic situation due to the fragmentation and scarcity of its habitat. We found that concave terrain profiles are important for the presence of the Antioquia Brushfinch. These results allow the identification of priority areas for the species. With additional information, for example, direct evidence of occupancy, these sites could be recommended for its protection. Finally, the determination of variables related to the ecology of the species provides crucial information for future studies. We hope that the results obtained through this work will help promote the protection and appropriation of this species as another signature element of Andean biodiversity.
RESPONSES TO THIS ARTICLE
Responses to this article are invited. If accepted for publication, your response will be hyperlinked to the article. To submit a response, follow this link. To read responses already accepted, follow this link.
ACKNOWLEDGMENTS
We thank IdeaWild for its invaluable support in realizing this project, and the Atlapetes Project for its guidance and help during data collection. To Juliana Villa for her help in the field phase. To the families and owners of San Pedro de los Milagros for caring for, protecting, and claiming this fascinating species as a vital part of their territory and for their willingness to do this research. Finally, to the University of Antioquia and the Ecología y Evolución de Vertebrados research group.
DATA AVAILABILITY
The recordings are available through the corresponding author. The code that support the findings of this study are available in Github at https://doi.org/10.5281/zenodo.6872141, reference number da05d85. These codes were derived from the following resources available in the public domain: https://github.com/Mauriciodiazz/A_blancae_occ_code/tree/main/A_blancae_occ_code.
LITERATURE CITED
Akaike, H. 1998. Information theory and an extension of the maximum likelihood principle. Pages 199-213 in E. Parzen, K. Tanabe, and G. Kitagawa, editors. Selected papers of Hirotugu Akaike. Springer, New York, New York, USA. https://doi.org/10.1007/978-1-4612-1694-0_15
Amatulli, G., D. McInerney, T. Sethi, P. Strobl, and S. Domisch. 2020. Geomorpho90m, empirical evaluation and accuracy assessment of global high-resolution geomorphometric layers. Scientific Data 7(162):162. https://doi.org/10.1038/s41597-020-0479-6
Andrade-Ponce, G., J. C. Cepeda-Duque, S. Mandujano, K. L. Velásquez-C, B. Gómez-Valencia, and D. J. Lizcano. 2021. Modelos de ocupación para datos de cámaras trampa. Mammalogy Notes 7(1):200. https://doi.org/10.47603/mano.v7n1.200
Arbeláez-Cortés, E., H. A. Rodríguez-Correa, and M. Restrepo-Chica. 2011. Mixed bird flocks: patterns of activity and species composition in a region of the Central Andes of Colombia. Revista Mexicana de Biodiversidad 82(2):639-651. https://doi.org/10.22201/ib.20078706e.2011.2.468
Arias, L. A. 2007. Las concavidades de primer orden: expresión del mecanismo activo de modelado en el Altiplano de Santa Rosa de Osos. Boletín de Ciencias de la Tierra 20:9-38. http://www.scielo.org.co/scielo.php?script=sci_arttext&pid=S0120-36302007000100001
Arias, L. A., and L. H. González. 2003. La diversidad del relieve y los suelos en el altiplano de Santa Rosa de Osos (Antioquia): Sus significados ambientales. Boletín de Ciencias de la Tierra 15:51-72. https://revistas.unal.edu.co/index.php/rbct/article/view/95468
Arias, L. A., L. H. González, and G. Arias. 2000. Historia del relieve y los Suelos en el Altiplano de Santa Rosa de Osos Antioquia Región el Vergel. Universidad Nacional de Colombia, Sede Medellín, Facultad de Ciencias, Corantioquia, Antioquia, Columbia.
Balantic, C., and T. Donovan. 2019. Dynamic wildlife occupancy models using automated acoustic monitoring data. Ecological Applications 29(3):e01854. https://doi.org/10.1002/eap.1854
Beissinger, S. R., and M. I. Westphal. 1998. On the use of demographic models of population viability in endangered species management. Journal of Wildlife Management 62(3):821-841. https://doi.org/10.2307/3802534
Betancur, J. S., A. Morales-Rozo, and J. L. Parra. 2020. Occupancy of the Sooty Ant-Tanager (Habia gutturalis) and White-Mantled Barbet (Capito hypoleucus) in fragmented forests of the Central Andes in Colombia. Ornitologia Colombiana 18:eA03. https://asociacioncolombianadeornitologia.org/ojs/index.php/roc/article/view/383/309
BirdLife International. 2021. Atlapetes blancae. The IUCN Red List of threatened species 2021:e.T22735460A181746724. https://dx.doi.org/10.2305/IUCN.UK.2021-3.RLTS.T22735460A181746724.en
Botero-Delgadillo, E., N. J. Bayly, J. Sanabria-Mejía, P. Caicedo, and S. Escudero-Páez. 2022a. On the habitat use and foraging ecology of the Yellow-headed Brushfinch (Atlapetes flaviceps), an endemic species of conservation concern from Colombia. Wilson Journal of Ornithology 134(1):106-113. https://doi.org/10.1676/20-00132
Botero-Delgadillo, E., S. Escudero-Páez, J. Sanabria-Mejía, P. Caicedo, and N. J. Bayly. 2022b. Sequential use of niche and occupancy models identifies conservation and research priority areas for two data-poor endemic birds from the Colombian Andes. Ornithological Applications 124(1):duab063. https://doi.org/10.1093/ornithapp/duab063
Burnham, K. P., and D. R. Anderson. 2002. Model selection and multimodel inference: a practical information-theoretic approach. Second edition. Springer, New York, New York, USA. https://doi.org/10.1007/b97636
Campos-Cerqueira, M., and T. M. Aide. 2016. Improving distribution data of threatened species by combining acoustic monitoring and occupancy modelling. Methods in Ecology and Evolution 7(11):1340-1348. https://doi.org/10.1111/2041-210X.12599
Center for Conservation Bioacoustics. 2019. Raven Pro: interactive sound analysis Software. Version 1.6.1. Cornell University, Ithaca, New York, USA. http://ravensoundsoftware.com/
Chambert, T., D. Pardo, R. Choquet, V. Staszewski, K. D. McCoy, T. Tveraa, and T. Boulinier. 2012. Heterogeneity in detection probability along the breeding season in Black-legged Kittiwakes: implications for sampling design. Journal of Ornithology 152:371-380. https://doi.org/10.1007/s10336-010-0542-8
Chaparro-Herrera, S., M. Hernández-Schmidt, and A. Lopera-Salazar. 2021. Notas sobre la dieta y el hábitat del Gorrión-Montés paisa Atlapetes blancae (Passerellidae). Actualidades Biológicas 43(115):1-14. https://doi.org/10.17533/udea.acbi.v43n115a06
Chaparro-Herrera, S., and A. Lopera-Salazar. 2019. Descripción del nido, huevos y comportamientos reproductivos del Gorrión-montés paisa (Atlapetes blancae). Ornitología Colombiana 17:eNB08. https://asociacioncolombianadeornitologia.org/ojs/index.php/roc/article/view/403/327
Chaparro-Herrera, S., A. Lopera-Salazar, R. Correa Peña, and J. L. Parra. 2022. Inventario y nuevos registros de la Avifauna de San Pedro de los Milagros, Antioquia, Colombia. Cotinga 44(2022):24-42.
Chaparro-Herrera, S., A. Lopera-Salazar, R. D. Ruíz-Correa, and A. M. Castaño. 2020. Sobre la distribución del Gorrión-Montés de Anteojos (Atlapetes flaviceps) en Antioquia. Boletín SAO 29(1-2):1-5. https://sao.org.co/publicaciones/boletinsao/29_1n2/29_1n2.html
Clement, M. J., T. J. Rodhouse, P. C. Ormsbee, J. M. Szewczak, and J. D. Nichols. 2014. Accounting for false-positive acoustic detections of bats using occupancy models. Journal of Applied Ecology 51(5):1460-1467. https://doi.org/10.1111/1365-2664.12303
Correa, R., S. Chaparro-Herrera, A. Lopera-Salazar, and J. L. Parra. 2019. Rediscovery of the Antioquia Brush Finch Atlapetes blancae. Cotinga 41(2019):101-108.
Devarajan, K., T. L. Morelli, and S. Tenan. 2020. Multi-species occupancy models: review, roadmap, and recommendations. Ecography 43(11):1612-1624. https://doi.org/10.1111/ecog.04957
Donegan, T. M. 2007. A new species of brush finch (Emberizidae: Atlapetes) from the northern Central Andes of Colombia. Bulletin-British Ornithologists Club 127(4):255-268. https://www.biodiversitylibrary.org/page/40881795#page/5/mode/1up
Donegan, T. M., J. E. Avendaño-C, B. Huertas, and P. Flórez. 2009. Avifauna de San Pedro de los Milagros, Antioquia: una comparación entre colecciones antiguas y evaluaciones rápidas. Boletín Científico. Centro de Museos. Museo de Historia Natural 13(1):63-72. http://www.scielo.org.co/scielo.php?pid=S0123-30682009000100005&script=sci_abstract&tlng=es
Edwards, D. P., B. Fisher, and D. S. Wilcove. 2012. High conservation value or high confusion value? Sustainable agriculture and biodiversity conservation in the tropics. Conservation Letters 5(1):20-27. https://doi.org/10.1111/j.1755-263X.2011.00209.x
Fiske, I., and R. Chandler. 2011. Unmarked: an R package for fitting hierarchical models of wildlife occurrence and abundance. Journal of Statistical Software 43(10):1-23. https://doi.org/10.18637/jss.v043.i10
Gliessman, S. R. 2002. Agroecología: procesos ecológicos en agricultura sostenible. First edition. E. Rodríguez, T. Benjamin, L. Rodríguez, and A. Cortés, editors. CATIE, Turrialba, Costa Rica.
Gobernación de Antioquia, Secretaría de Medio Ambiente, D. de S. de I.A. 2016. Anuario estadístico de Antioquia: tasa de deforestación entre los años 2000 a 2012, por municipios y subregiones. 2000 to 2012. Gobernación de Antioquia, Antioquia, Columbia. https://antioquia.gov.co/planeacion/ANUARIO%202013/es-CO/capitulos/ambiente/impacto/suelo/cp-2-3-1-10.html?url=https://antioquia.gov.co/planeacion/ANUARIO%202013/es-CO/capitulos/transporte.html
Guillera-Arroita, G. 2017. Modelling of species distributions, range dynamics and communities under imperfect detection: advances, challenges and opportunities. Ecography 40(2):281-295. https://doi.org/10.1111/ecog.02445
Guillera-Arroita, G., M. S. Ridout, and B. J. T. Morgan. 2010. Design of occupancy studies with imperfect detection. Methods in Ecology and Evolution 1(2):131-139. https://doi.org/10.1111/J.2041-210X.2010.00017.X
Hijmans, R. J. 2021. terra: spatial data analysis. R package version 1.4-7. R Foundation for Statistical Computing, Vienna, Austria. https://cran.r-project.org/web/packages/terra/index.html
Instituto Geográfico Agustin Codazzi (IGAC). 2019. Datos hidrológicos y meterológicos (DHIME). Instituto Geográfico Agustin Codazzi, Bogota, Columbia. http://dhime.ideam.gov.co/webgis/home/item.html?id=cc12ae8165304d53b81d0d2d10154042
Jetz, W., J. M. McPherson, and R. P. Guralnick. 2012. Integrating biodiversity distribution knowledge: toward a global map of life. Trends in Ecology and Evolution 27(3):151-159. https://doi.org/10.1016/j.tree.2011.09.007
Kéry, M., and J. A. Royle. 2015. Applied hierarchical modeling in ecology: analysis of distribution, abundance and species richness in R and BUGS. Second edition. Dynamic and Advanced Models. Elsevier, San Diego, California, USA.
Krabbe, N. 2004. Pale-headed Brush-finch Atlapetes pallidiceps: notes on population size, habitat, vocalizations, feeding, interference competition and conservation. Bird Conservation International 14(2):77-86. https://doi.org/10.1017/S0959270904000115
Luoto, M., R. Virkkala, and R. K. Heikkinen. 2007. The role of land cover in bioclimatic models depends on spatial resolution. Global Ecology and Biogeography 16(1):34-42. https://doi.org/10.1111/j.1466-8238.2006.00262.x
MacKenzie, D. I., J. D. Nichols, J. A. Royle, K. H. Pollock, L. L. Bailey, and J. E. Hines. 2018. Occupancy estimation and modeling: inferring patterns and dynamics of species occurrence. Second edition. Elsevier, Burlington, Vermont, USA. https://doi.org/10.1016/C2012-0-01164-7
MacKenzie, D. I., J. D. Nichols, N. Sutton, K. Kawanishi, and L. L. Bailey. 2005. Improving inferences in population studies of rare species that are detected imperfectly. Ecology 86(5):1101-1113. https://doi.org/10.1890/04-1060
Mahe, G., J.-E. Paturel, E. Servat, D. Conway, and A. Dezetter. 2005. The impact of land use change on soil water holding capacity and river flow modelling in the Nakambe River, Burkina-Faso. Journal of Hydrology 300(1-4):33-43. https://doi.org/10.1016/j.jhydrol.2004.04.028
Mertes, K., and W. Jetz. 2018. Disentangling scale dependencies in species environmental niches and distributions. Ecography 41(10):1604-1615. https://doi.org/10.1111/ecog.02871
Miguet, P., H. B. Jackson, N. D. Jackson, A. E. Martin, and L. Fahrig. 2016. What determines the spatial extent of landscape effects on species? Landscape Ecology 31:1177-1194. https://doi.org/10.1007/s10980-015-0314-1
Oppel, S., H. M. Schaefer, and V. Schmidt. 2003. Description of the nest, eggs, and breeding behavior of the endangered Pale-headed Brush-Finch (Atlapetes pallidiceps) in Ecuador. Wilson Bulletin 115(4):360-366. https://doi.org/10.1676/03-020
Oppel, S., H. M. Schaefer, V. Schmidt, and B. Schröder. 2004. Habitat selection by the Pale-headed Brush-finch (Atlapetes pallidiceps) in southern Ecuador: implications for conservation. Biological Conservation 118(1):33-40. https://doi.org/10.1016/j.biocon.2003.07.006
Patterson, P. L., and S. Healey. 2015. Global ecosystem dynamics investigation (GEDI) LiDAR sampling strategy. In S. M. Stanton and G. A. Christensen, editors. Pushing boundaries: new directions in inventory techniques and applications: forest inventory and analysis (FIA) symposium, December 8-10 Portland, Oregon. General Technical Report PNW-GTR-931. USA Department of Agriculture, Forest Service, Pacific Northwest Research Station, Corvallis, Oregon, USA. https://www.fs.usda.gov/research/treesearch/50389
R Core Team. 2021. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.r-project.org/
Renjifo, L. M., M. F. Gómez, J. Velásquez-Tibatá, Á. M. Amaya-Villarreal, G. H. Kattan, J. D. Amaya-Espinel, and J. Burbano-Girón. 2014. Libro rojo de aves de Colombia. VOL. I. Bosques húmedos de los Andes y la Costa Pacífica. First edition. Pontificia Universidad Javerinana, Instituto Alexander von Humboldt, Bogotá, Colombia. http://repository.humboldt.org.co/handle/20.500.11761/32545
Renjifo, L. M., Á. M. Amaya-Villarreal, J. Burbano-Girón, and J. Velásquez-Tibatá. 2016. Libro Rojo de Aves de Colombia. VOL. II. Ecosistemas abiertos, secos, insulares, acuáticos continentales, marinos, tierras altas del Darién y Sierra Nevada de Santa Marta y bosques húmedos del centro, norte y oriente del país. First edition. Pontificia Universidad Javerinana, Instituto Alexander von Humboldt, Bogotá, Colombia. http://repository.humboldt.org.co/handle/20.500.11761/34285
Shaw, T., S. R. Schönamsgruber, J. M. Cordeiro Pereira, and G. Mikusiński. 2022. Refining manual annotation effort of acoustic data to estimate bird species richness and composition: the role of duration, intensity, and time. Ecology and Evolution 12(11):e9491. https://doi.org/10.1002/ece3.9491
Spearman, C. 1904. The proof and measurement of association between two things. American Journal of Psychology 15(1):72-99. http://digamoo.free.fr/spearman1904a.pdf
Symes, L. B., K. D. Kittelberger, S. M. Stone, R. T. Holmes, J. S. Jones, I. P. Castaneda Ruvalcaba, M. S. Webster, and M. P. Ayres. 2022. Analytical approaches for evaluating passive acoustic monitoring data: a case study of avian vocalizations. Ecology and Evolution 12(4):e8797. https://doi.org/10.1002/ece3.8797
Unidades Municipales de Asistencia Técnica Agropecuaria (UMATA). 2019. Análisis de coberturas de las veredas donde se llevará acabo el diagnóstico del SILAP de San Pedro, para propuesta de muestreo. San Pedro de los Milagros, Colombia.
Valencia-C, G., D. J. Sánchez-Londoño, A. I. Villamizar, and A. Ángel. 2019. Una nueva localidad de Atlapetes blancae (Passerellidae, Passeriformes), con comentarios sobre su hábitat. Ornitología Colombiana 17:eNB07. https://asociacioncolombianadeornitologia.org/ojs/index.php/roc/article/view/402/326
Fig. 1
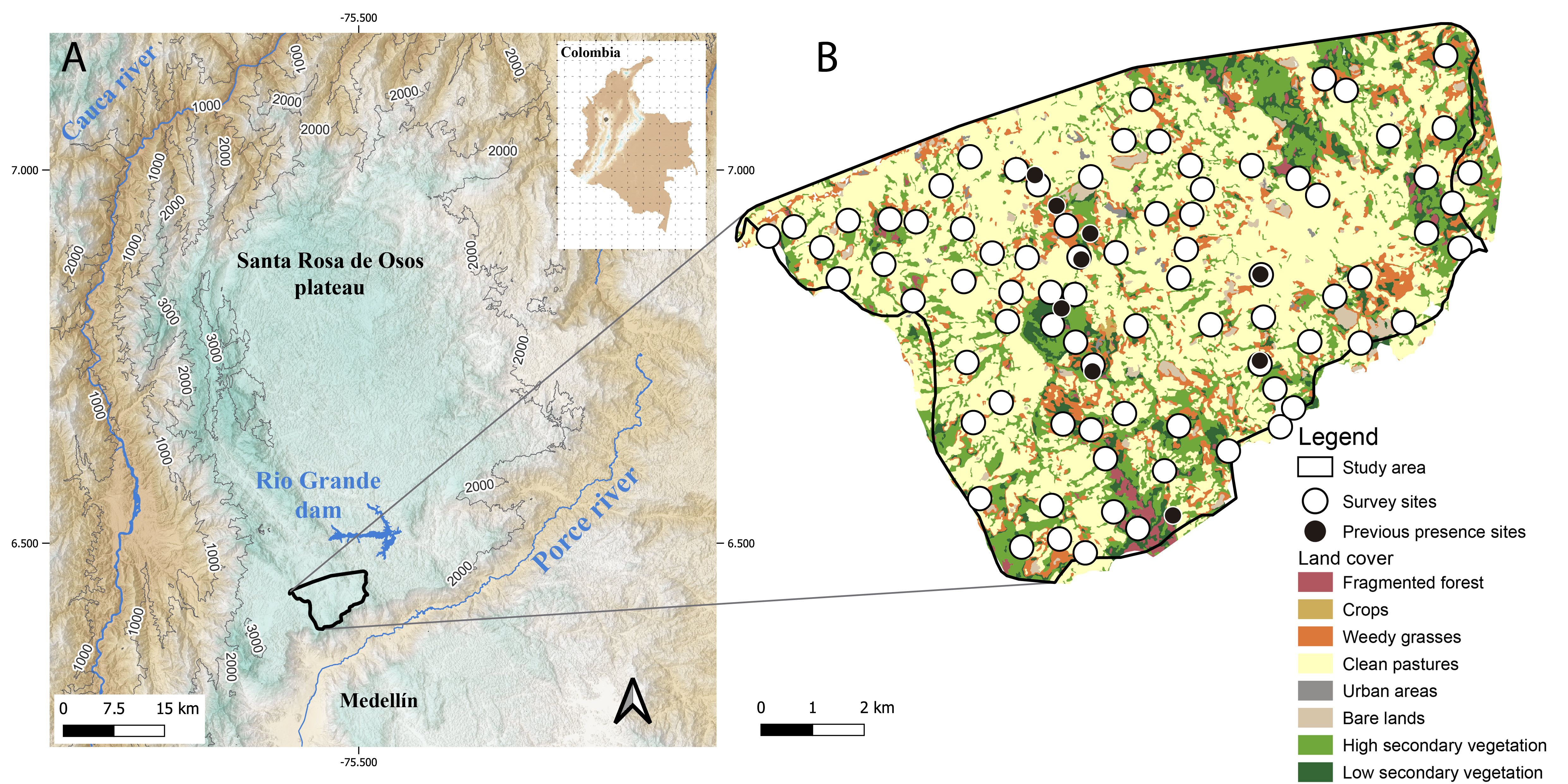
Fig. 1. Study area and location of sampling sites within the Santa Rosa de Osos high elevation plateau (ASRO; Antioquia, Colombia), for the occupancy model of the Antioquia Brushfinch (Atlapetes blancae). (A) The northern section of the Central Andes in Colombia includes ASRO. The study area is highlighted inside the black polygon. (B) The different land cover types within the study area. Previously known sites of occurrence for this species are represented by black dots, and white circles represent all sampling sites with acoustic recorders.

Fig. 2
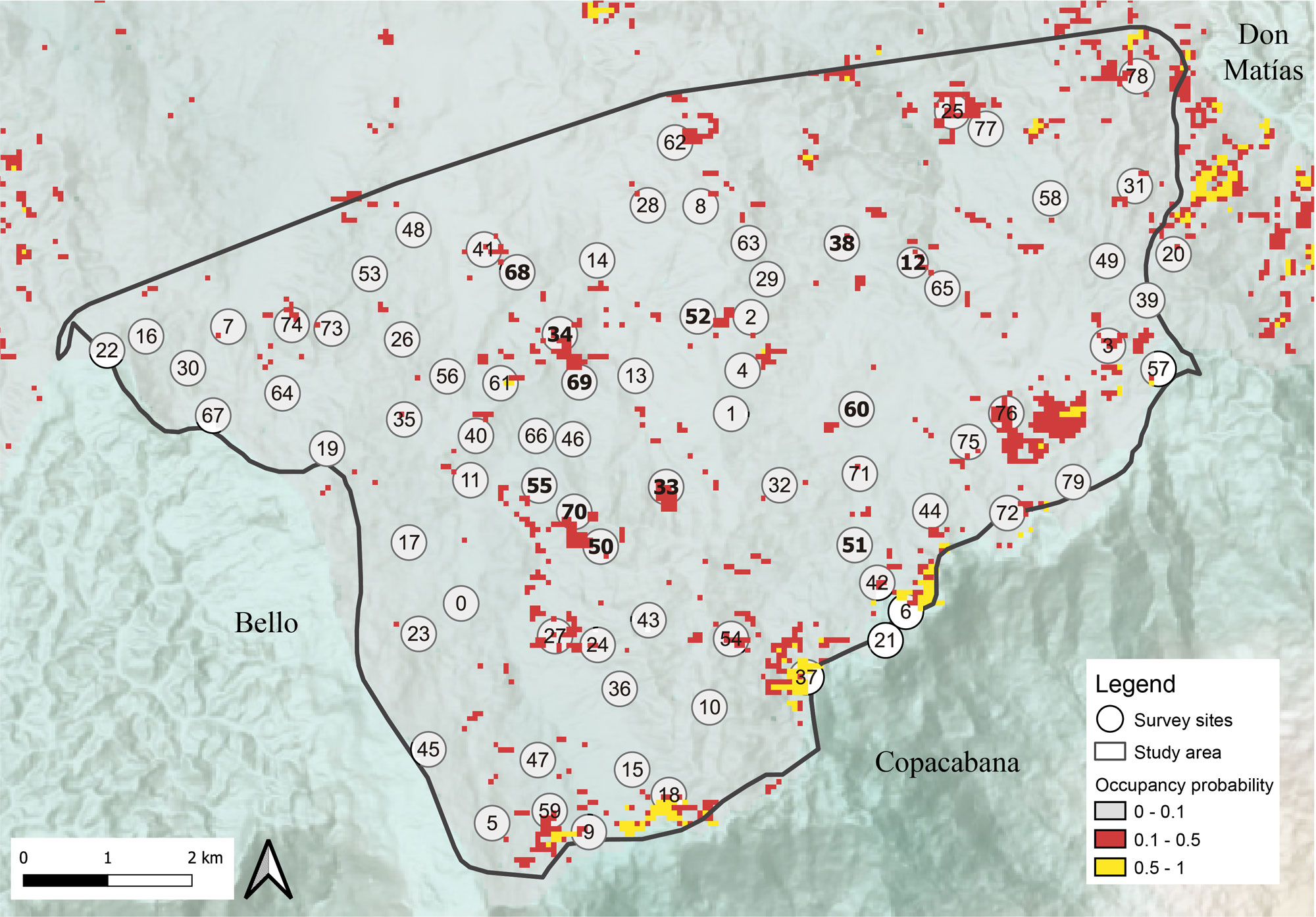
Fig. 2. Occupancy probability of the Antioquia Brushfinch (Atlapetes blancae) in the municipality of San Pedro de los Milagros, Antioquia, Colombiastudy area. Spatial resolution of the predicted grid is 53 m, resembling the area sampled in our sampling units. White circles represent the 80 study sites. Bold numbers represent sites where the species was detected with the recorders, during the installation of the recorders, or previous to this study.

Fig. 3
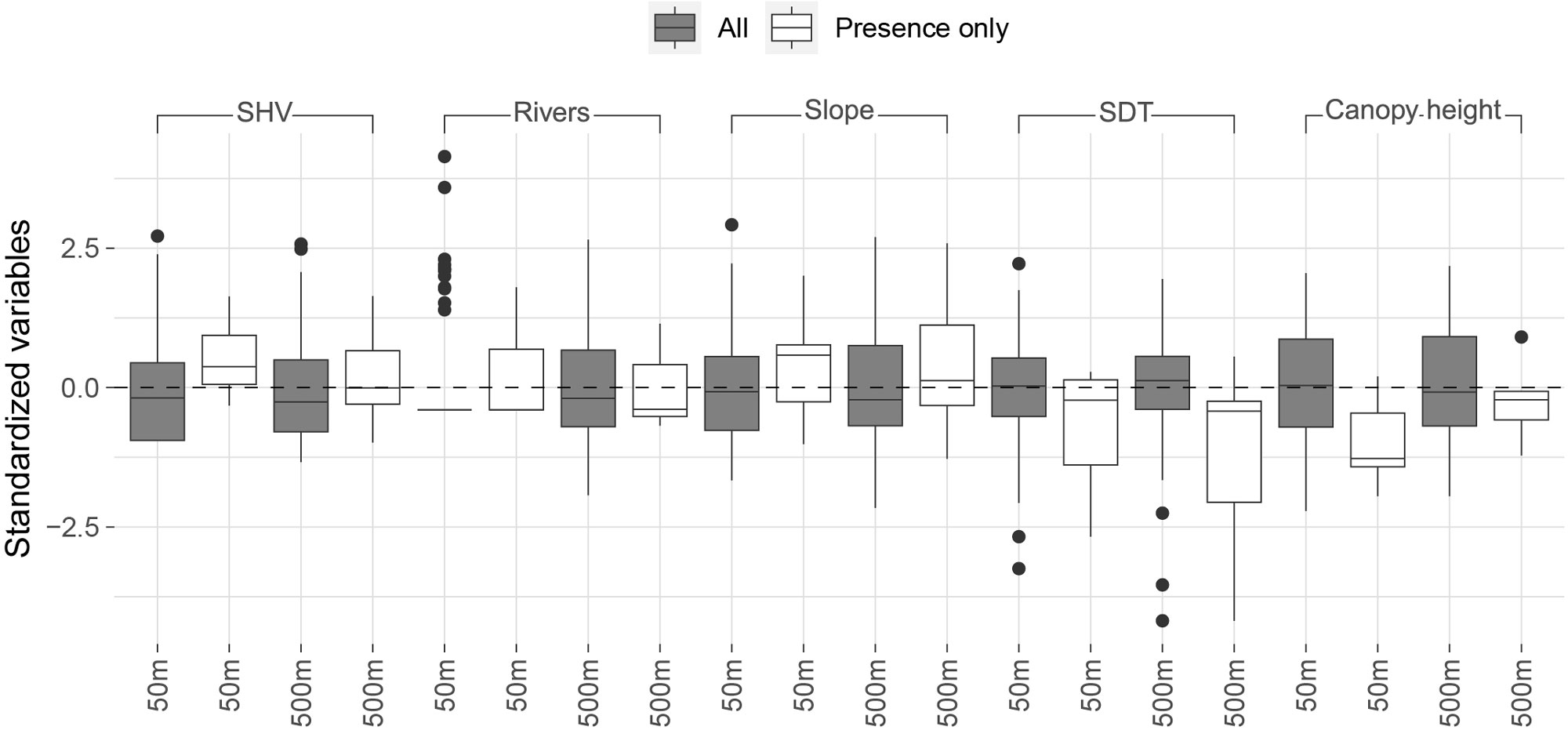
Fig. 3. Comparison of the distribution of values between sites with detections (white boxes) and all sites (gray boxes) for each standardized covariate, i.e., area of shrub and herbaceous vegetation (SHV), length of water bodies, average slope, average second derivative of terrain (SDT), and average canopy height measured at each of two scales (50 and 500 m radio). Notice the marked differences in the values for SHV, SDT, and canopy height, the three most important covariates contributing to variation in occupancy.

Fig. 4
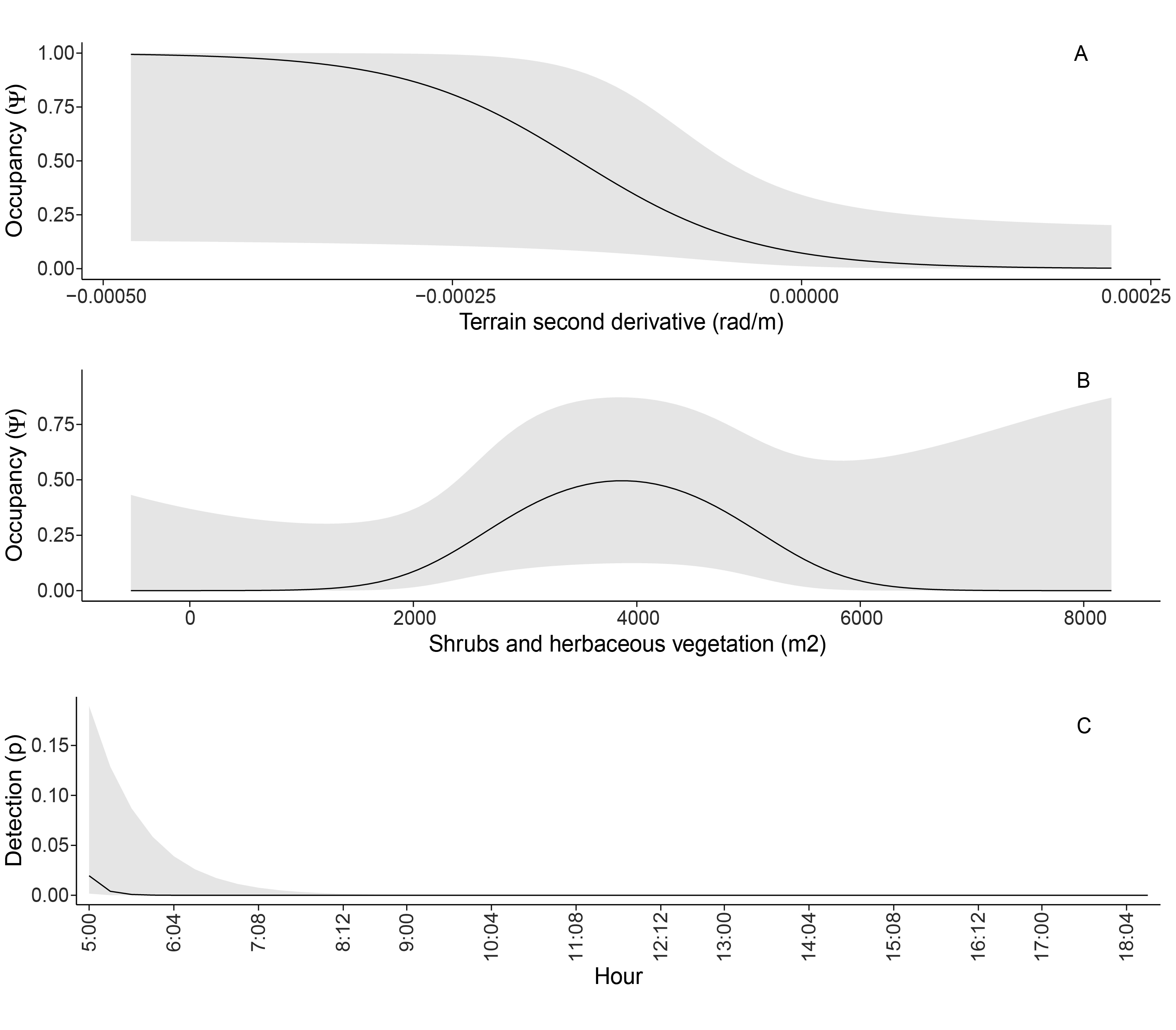
Fig. 4. Occupancy as a function of the second derivative of terrain (A), area of shrub and herbaceous vegetation (B), and detection as a function of the time of day (C) for the best occupancy model of the Antioquia Brushfinch (Atlapetes blancae) in the study area. The shaded area corresponds to the 95% confidence interval.

Table 1
Table 1. Results of the candidate occupancy models for the Antioquia Brushfinch (Atlapetes blancae). AICc: Akaike information criterion corrected for sample size. ΔAICc: the difference in AICc relative to the best model. A_vsbpe_x: coverage area of shrub and herbaceous vegetation and weedy grasses (SHV) in a circular area of 50 or 500 m radius. Rivers_bx: meters of watersheds in a circular area of 50 or 500 m radius. Slop_x: mean percentage of slope in a circular area of 50 or 500 m radius. Sdt_bx: mean second derivative of the terrain in a circular area of 50 or 500 m radius. CH_GEDI_50: mean height of the canopy in a circular area of 50 m radius.
| Candidate models | AICc | ΔAICc | |||||||
| Detection submodels | |||||||||
| 1 | p (hour) psi ( . ) | 97.27 | 0 | ||||||
| 2 | p ( . ) psi ( . ) | 108.57 | 11.30 | ||||||
| Occupancy submodels | |||||||||
| 1 | p (hour) psi (A_vsbpe_50^2 + Sdt_b500) | 83.55 | 0 | ||||||
| 2 | p (hour) psi (A_vsbpe_50^2 + CH_GEDI_50) | 88.14 | 4.59 | ||||||
| 3 | p (hour) psi (A_vsbpe_50 + Sdt_b500) | 89.23 | 5.67 | ||||||
| 4 | p (hour) psi (A_vsbpe_50^2 + Sdt_b50) | 89.41 | 5.86 | ||||||
| 5 | p (hour) psi (A_vsbpe_50 + Sdt_b50 + Sdt_b500 + slop_50 + slop_500 + CH_GEDI_50) | 90.33 | 6.78 | ||||||
| 6 | p (hour) psi (A_vsbpe_50^2 + A_vsbpe_500 + CH_GEDI_50) | 90.47 | 6.91 | ||||||
| 7 | p (hour) psi (A_vsbpe_50 + Sdt_b50 + slop_50 + CH_GEDI_50) | 92.11 | 8.55 | ||||||
| 8 | p (hour) psi (A_vsbpe_50^2 + Rivers_b50 + Rivers_b500 + CH_GEDI_50) | 92.86 | 9.30 | ||||||
| 9 | p (hour) psi (A_vsbpe_50 + CH_GEDI_50 ^2 + Sdt_b50) | 93.38 | 9.82 | ||||||
| 10 | p (hour) psi (A_vsbpe_50 + CH_GEDI_50) | 93.53 | 9.97 | ||||||
| 11 | p (hour) psi (A_vsbpe_50^2 + slop_500) | 93.93 | 10.37 | ||||||
| 12 | p (hour) psi (A_vsbpe_50 + Sdt_b50) | 94.05 | 10.50 | ||||||
| 13 | p (hour) psi (A_vsbpe_50^2 + slop_50) | 94.68 | 11.13 | ||||||
| 14 | p (hour) psi (A_vsbpe_50^2 + Rivers_b500) | 94.74 | 11.18 | ||||||
| 15 | p (hour) psi (A_vsbpe_50^2 + Rivers_b50) | 94.79 | 11.23 | ||||||
| 16 | p (hour) psi (Rivers_b50 + Rivers_b500 + Sdt_b50 + Sdt_b500 + A_vsbpe_50 + slop_50 + slop_500 +CH_GEDI_50) | 95.42 | 11.86 | ||||||
| 17 | p (hour) psi (A_vsbpe_50 + A_vsbpe_500 + CH_GEDI_50) | 95.75 | 12.19 | ||||||
| 18 | p (hour) psi (A_vsbpe_50 + A_vsbpe_500^2 + CH_GEDI_50) | 96.94 | 13.38 | ||||||
| 19 | p (hour) psi (A_vsbpe_50 + CH_GEDI_50 ^2) | 98.15 | 14.60 | ||||||
| 20 | p (hour) psi (A_vsbpe_50 + slop_500) | 98.70 | 15.14 | ||||||
| 21 | p (hour) psi (A_vsbpe_50 + slop_50) | 99.04 | 15.48 | ||||||
| 22 | p (hour) psi (A_vsbpe_50 + Rivers_b50) | 99.55 | 16.00 | ||||||
| 23 | p (hour) psi (A_vsbpe_50 + Rivers_b50 + Rivers_b500 + CH_GEDI_50) | 99.80 | 16.25 | ||||||
| 24 | p (hour) psi (A_vsbpe_50 + Rivers_b500) | 99.89 | 16.34 | ||||||
| 25 | p (hour) psi (A_vsbpe_50 + Rivers_b50 + Rivers_b500 + CH_GEDI_50 ^2) | 99.92 | 16.36 | ||||||
| 26 | p (hour) psi (A_vsbpe_50 + A_vsbpe_500^2 + Rivers_b50 + Rivers_b500 + CH_GEDI_50) | 101.59 | 18.03 | ||||||
| 27 | p ( . ) psi ( . ) | 108.57 | 25.02 | ||||||
Table 2
Table 2. Acoustic detections for the Antioquia Brushfinch (Atlapetes blancae) throughout the entire study. Seventeen presence records are included and for each record the site number, the land cover category, the vocalization type, and the date and the time of registration.
| Site | Land cover | Vocalization type | Date | Hour | |||||
| 12 | Fragmented forest | Song | 05/24/2021 | 06:20 | |||||
| 12 | Fragmented forest | Call and song | 05/25/2021 | 05:48 | |||||
| 12 | Fragmented forest | Song | 05/25/2021 | 12:28 | |||||
| 12 | Fragmented forest | Song | 05/26/2021 | 05:48 | |||||
| 12 | Fragmented forest | Song | 05/26/2021 | 06:20 | |||||
| 33 | High secondary vegetation | Call and song | 05/05/2021 | 14:20 | |||||
| 38 | Low secondary vegetation | Call and song | 05/30/2021 | 08:12 | |||||
| 51 | Low secondary vegetation | Song | 05/15/2021 | 06:52 | |||||
| 51 | Low secondary vegetation | Call and song | 05/16/2021 | 07:08 | |||||
| 51 | Low secondary vegetation | Song | 05/16/2021 | 07:40 | |||||
| 51 | Low secondary vegetation | Song | 05/18/2021 | 07:40 | |||||
| 52 | High secondary vegetation | Song | 05/14/2021 | 06:20 | |||||
| 52 | High secondary vegetation | Song | 05/15/2021 | 06:20 | |||||
| 52 | High secondary vegetation | Song | 05/18/2021 | 06:04 | |||||
| 52 | High secondary vegetation | Song | 05/20/2021 | 06:36 | |||||
| 69 | Low secondary vegetation | Song | 05/15/2021 | 06:20 | |||||
| 70 | High secondary vegetation | Song | 05/05/2021 | 07:24 | |||||
Table 3
Table 3. Results of the best occupancy model. SE = standard error; z = z-score for the coefficient of each covariate and its associated P-value. All estimates are in logit scale.
| Estimate | SE | z | P(> |z|) | ||||||
| Occupancy | |||||||||
| (Intercept) | -2.42 | 0.90 | -2.68 | 0.007 | |||||
| A_vsbpe_50 | 4.93 | 2.60 | 1.90 | 0.06 | |||||
| I(A_vsbpe_50^2) | -2.67 | 1.54 | -1.73 | 0.08 | |||||
| Sdt_b500 | -1.63 | 0.76 | -2.14 | 0.03 | |||||
| Detection | |||||||||
| (Intercept) | -2.16 | 0.67 | -3.22 | 0.001 | |||||
| hour | -1.65 | 0.60 | -2.73 | 0.006 | |||||








