The following is the established format for referencing this article:
Phillips, R. A., K. Wolf, R. P. Bourbour, J. A. Urbina, J. E. Ruano, V. M. Bonilla, V. Gamez, I. I. Mai, I. Morataya, R. A. Martinez, L. Pop, P. M. Balderamos, H. L. Jones, R. E. Melcer Jr., L. A. Trulio, and E. Ruelas Inzunza. 2023. Intratropical migration of the Hook-billed Kite (Chondrohierax uncinatus) in Middle America. Journal of Field Ornithology 94(1):5.ABSTRACT
The Hook-billed Kite (Chondrohierax uncinatus) is a specialized and secretive Neotropical raptor that has received little research attention. Despite scattered records of Hook-billed Kite movements, this species was long thought to be non-migratory. We studied the southbound autumn migration of Hook-billed Kites in Veracruz, Mexico, and Belize and investigated whether environmental conditions on their breeding grounds influenced migration. We collected migration count data over 8 years (2013–2020) in Belize and 25 years (1995–2019) in Veracruz. In Belize, we recorded 39,928 Hook-billed Kites on their southbound autumn migration during a total of 3093.1 count hours, with an autumn seasonal mean (±SE) count of 4991 ± 1083 kites/year, with flocks consisting of up to 200 individuals. In comparison, during a total of 42,531 count hours at two count sites in Veracruz, we recorded 3870 Hook-billed Kites with a seasonal mean (±SE) count of 154.8 ± 12.1 kites/year. Most kites in Veracruz migrated as single individuals or groups of < 4 birds and the largest flock ever recorded had 12 individuals. The mean 95% seasonal passage window of Hook-billed Kites in Belize lasted 44.9 ± 2.5 d (N = 8 years) from 26 October to 9 December compared to an earlier passage from 13 September–11 November in Veracruz (N = 25 years). Precipitation on the breeding grounds had no influence on the timing or magnitude of the kite migration through Belize; whereas, in Veracruz, migration timing occurred later as precipitation north of the count site increased. Notably, age, sex, and color morph classes migrated in mixed flocks; therefore, we detected no signs of differential migration, which is uncommon among raptors. Our study describes, for the first time, the largest known southbound autumn migration of Hook-billed Kites. Working to understand the life histories of tropical raptors provides critical ecological information, which can aid in identifying potential threats, conservation needs, and population statuses.
RESUMEN
Chonrrohierax uncinatus es una ave rapaz Neotropical especializada y sigilosa que ha recibido poca atención en la investigación. A pesar de los registros dispersos de los movimientos de C. uncinatus, por largo tiempo se ha pensado que esta especie no es migratoria. Estudiamos la migración de otoño hacia el sur de C. unicnatus en Veracruz, México y Belice e investigamos si las condiciones ambientales en la zona de reproducción influencian la migración. Colectamos datos de conteos migratorios durante 8 años (2013-2020) en Belice y 25 años (1995-2019) en Veracruz. En Belice registramos 39,928 individuos durante su migración de otoño hacia el sur durante un total de 3093.1 horas de conteo, con un promedio de los conteos de otoño estacionales (±SE) de 4991 ± 1083 aves/año, con bandadas compuestas por hasta 200 individuos. En comparación, durante un total de 42,531 horas de conteo en dos sitios en Veracruz, registramos 3870 C. uncinatus con un promedio en los conteos estacionales (±SE) de 154.8 ± 12.1 aves/año. La mayoría de las aves en Veracruz migraron solas o en grupos de < 4 aves y las bandadas mas grandes que se registraron tuvieron 12 individuos. La ventana de paso promedio del 95% de C. uncinatus en Belice duró 44.9 ± 2.5 (N=8 años) desde el 26 de octubre hasta el 9 de diciembre comparado con un paso mas temprano del 13 de Septiembre – 11 de Noviembre en Veracruz (N=25 años). La precipitación en la zona de reproducción no tuvo una influencia en el momento o la magnitud de la migración de C. uncinatus a través de Belice; sin embargo, en Veracruz, el momento de la migración ocurrió mas tarde a medida que la precipitación en el norte de los sitios de conteo incrementó. Notablemente aves en diferentes clases de edad, sexo y morfotipo de color migraron en bandadas mixtas; consecuentemente, no detectamos ninguna señal diferencial de la migración, lo que es poco común en aves rapaces. Trabajar con el fin de comprender las historias de vida de rapaces tropicales provee información ecológica crítica que puede ayudar en la identificación de amenazas potenciales, necesidades de conservación y estatus poblacional.
INTRODUCTION
The most diverse raptor communities are found in the Neotropics, encompassing approximately one-third of all raptor species globally (Bildstein 2004). Despite having high species richness, raptors in this region have received relatively little research attention, creating critical information gaps in our understanding of the ecology of these species (Whitacre 2012, Buechley et al. 2019). Tropical raptors are integral components in maintaining the function of tropical ecosystems (Sergio et al. 2006, Estes et al. 2011), but they are declining globally and locally because of anthropogenic factors, such as habitat loss, persecution, and, to an unknown extent, climate change (Carrete et al. 2009, McClure et al. 2018).
Studying the movements and migration of tropical raptors informs their conservation by providing a better understanding of how these species are impacted and potentially threatened by anthropogenic activities and changing environmental conditions (Chapman et al. 2011, Jahn et al. 2020). Monitoring raptor species using migration counts has been a viable and important conservation tool since the mid-20th century (Bildstein 2006, Bildstein et al. 2007). It has allowed us to estimate indices of population abundance, demography, and helped identify migration corridors and regions for protection and management of species (Zalles and Bildstein 2000, Hoffman and Smith 2003). Because raptors are generally secretive, wide-ranging, highly mobile, and occur at low abundances, they are logistically challenging to survey and monitor during both breeding and non-breeding seasons (Zalles and Bildstein 2000, Bildstein et al. 2007).
The establishment of hundreds of raptor migration count sites globally has allowed researchers and conservationists to gather critical information on raptors when they are most abundant, visible, and concentrated. The few raptor migration count sites in the Neotropical region have focused primarily on long-distance migrations of Nearctic-Neotropical migrants. There has been little focus on documenting intratropical migrants (Jahn et al. 2020), which are species that breed in the tropics and annually migrate to another area within the tropics (Hayes 1995). Nearctic-Neotropical monitoring programs have different study objectives and do not specifically capture the movements of raptors that spend their annual cycle exclusively within the Neotropical region. Consequently, the intratropical migrations and other movement patterns of most Neotropical raptors have not been studied, making information on the migratory patterns of this elusive group a high research priority (Bildstein 2004, Areta and Juhant 2019, Buechley et al. 2019, Jahn et al. 2020, Juhant 2022).
The Hook-billed Kite (Chondrohierax uncinatus) is an example of a Neotropical raptor with many unknown aspects of its life history, especially their migratory behaviors and patterns (Greeney and Boesman 2022). This species’ range extends from extreme southern Texas to northern Argentina. Despite this wide distribution, Hook-billed Kites are generally considered uncommon to rare, with some local declines (Ferguson-Lees and Christie 2001, Colorado et al. 2006, Johnson et al. 2007). Hook-billed Kites are dietary specialists, feeding almost exclusively on land snails (Whitacre and Vásquez 2012, Phillips et al. 2022). These kites are found in a variety of forest types, including but not limited to tropical lowland evergreen forest, tropical deciduous forest, gallery forest, montane evergreen forest, and xeric woodland (Ferguson-Lees and Christie 2001, Whitacre and Vásquez 2012, Clark and Schmitt 2017). However, researchers believe the kites occupy these habitats only if land snails occur in high densities, as prey abundance is considered the primary driver in their presence (Smith 1988, Clark and Schmitt 2017).
Although the breeding ecology of the Hook-billed Kite has been little studied, previous research suggests that the kite species occurs from mid-March through mid-October throughout most its northern distribution (Texas through northern South America), with peak nesting activity and most nesting observations coinciding with the wet season (Whitacre and Vásquez 2012, Phillips et al. 2022). It appears that nesting is synchronized closely with either the onset of the wet season or during the wettest months (Haverschmidt 1964, Marroquin et al. 1992). Therefore, precipitation may influence the abundance and availability of the Hook-billed Kite’s prey, which may subsequently influence its reproductive success and movement patterns.
The Hook-billed Kite is primarily a sedentary species throughout its range, but scattered observations suggest it could be a facultative migratory species (Zalles and Bildstein 2000, Ferguson-Lees and Christie 2001, Bildstein 2004, BirdLife International 2020). Collective movements of Hook-billed Kites were first documented in the early 1990s at the two Veracruz, Mexico migration monitoring sites, with an average autumn count of 120 kites from 1991 to 2001, and small groups observed throughout the season (Ruelas Inzunza et al. 2010a, Whitacre and Vásquez 2012). In Belize in 1999, HLJ first observed a flock of Hook-billed Kites during the autumn migration period, and in 2001, counted 1030 Hook-billed Kites moving along the southern coast in the Toledo District, all between 19 October and 15 November (Jones 2002). In South America, mainly in Brazil, observations of Hook-billed Kite flocks are rare and do not occur annually (de Melo Dantas et al. 2018). This range of observations highlights our limited understanding of the species’ movement ecology and whether this is truly a migratory species.
Given the uncertainty of this species’ movement patterns, we sought to characterize the migration of Hook-billed Kites in northern Middle America. Our study is the first to capture the complete autumn migration of Hook-billed Kites and provides the first comprehensive description of their migration. Using data from three raptor migration monitoring sites in Belize and Mexico, our specific objectives were to (1) document migratory movements of the Hook-billed Kite, (2) characterize the timing and magnitude of Hook-billed Kite post-breeding, southbound autumn migration in Belize and Veracruz, (3) determine population demographic parameters (sex, age, and color morph) of Hook-billed Kites from migration count data and assess differential migration using Belize count site data, and (4) determine if environmental conditions on the breeding grounds are related to the timing and magnitude of Hook-billed Kite migration in Belize and Veracruz.
METHODS
Study sites
Belize migration count site
RAP established a migration count site in Belize in 2013. The count site was located on a soccer field with open viewing in all directions in Cattle Landing, a coastal village ~2.5 km north of Punta Gorda, Toledo District (16.12024° N and -88.79436° W; Fig. 1). The count site is surrounded on the north by lowland broadleaved forest habitat (Meerman and Sabido 2001), on the south by the town of Punta Gorda, and is located immediately along the coastline of the Caribbean Sea, at ~3 meters above sea level. Belize has a subtropical climate with two seasons: dry (February–May) and wet (June–November). The mean annual rainfall at the count site is approximately 375 cm with a mean monthly temperature of 26.2 °C (National Meteorological Service of Belize 2021). Phillips (2014) describes the site in detail.
Veracruz, Mexico migration count site
We collected data of Hook-billed Kites from two migration count sites established by ERI, Dr. Laurie Goodrich, and Stephen Hoffman in central Veracruz, Mexico, which are the longest operating raptor migration monitoring sites in the Neotropics. Ruelas Inzunza et al. (2010b) describes the sites in detail: Cardel, Veracruz (19.37110° N, -96.37653° W) and Chichicaxtle, Veracruz (19.34308° N, -96.46419° W; Fig. 1). The Cardel count site is in the lowlands of the Gulf Coast Plain (29 meters above sea level) 7 km from the ocean. The Chichicaxtle count site is located 9.5 km west of the Cardel site (17 km from the coast) at 120 meters above sea level. The two sites are roughly at the same latitude, perpendicular to the migration front, in the geographic bottleneck formed by the proximity of the coast and two major mountain ranges, resulting in the largest concentration of migrating raptors in the world (Zalles and Bildstein 2000, Ruelas Inzunza et al. 2010b).
Regional climate
The climate of this region is highly variable, especially in Mexico (Kottek et al. 2006). The Hook-billed Kite’s northern extreme distribution is ~300 km north of the Tropic of Cancer. This area and south along the Gulf Coast Plain has an annual mean temperature between 24 and 28 °C, and temperature fluctuates little (5 °C) between the (boreal) summer and winter months (Parkes et al. 2021). Rainfall varies greatly by location and season but is greatest in the southeastern region (Howell and Webb 1995, Rhoda and Burton 2010). Areas with higher precipitation receive a mean annual rainfall of 100–200 cm (Parkes et al. 2021). Seasons are separated into a dry (February–May) and wet season (June–November; Howell and Webb 1995). The study area is in the hurricane belt and is susceptible to major storms from June to November, a significant factor in the variability of weather patterns (Young 2008).
Field data collection
We collected 8 years of count data in Belize (2013–2020) and 25 years (1995–2019) of count data from the Veracruz sites. In Belize, the count period was between 1 October and 15 December. The count period in Veracruz extended from 20 August to 20 November (Ruelas Inzunza et al. 2010a).
The count procedure at all count sites followed a standardized and widely used count protocol created by Hawk Migration Association of North America, described in detail by Bildstein et al. (2007). In Belize, Hook-billed Kites and all raptor species were counted daily from 0800 to 1600 H (CST) during the study period. Veracruz counts extended from 0900 to 1900 H during daylight savings and then reverted to 0800–1800 after daylight savings or the last Sunday of October (CST; Ruelas Inzunza et al. 2010b).
In Belize, starting in mid-2014 and in addition to counting the kites, we recorded date, time, flock size, flock number, and sex, age, and morph (Phillips 2014). Hook-billed Kites are sexually dimorphic, and plumage also differs dramatically within both juvenile and adult age classes, which allowed for accurate identification of each bird’s sex and age as it passed over the count station. Both juvenile and adult Hook-billed Kites exhibit different plumage morphs as well, allowing for additional classification of individuals according to their different plumage characteristics within sex and age categories. We attempted to identify each kite accordingly, but this was not always achievable because of the height of flight and distance from the site of some individuals. However, we made the assumption that the subset of kites that were identified to age and sex class were representative of all the kites passing the count site. In Veracruz, the proportion of individuals with recordable sex, age, and color morph class was minimal primarily because of flight height, and these data are not recorded consistently during the migration season.
At all sites, data we recorded at the start of each count hour included number of observers, number of visitors, wind speed, wind direction, temperature, barometric pressure, cloud cover, visibility, and precipitation; mean flight direction and height of flight was recorded at the end of each hour (Ruelas Inzunza 2007, Phillips 2014). We used a Kestrel weather meter (https://kestrelmeters.com/) to record wind speed, temperature, and barometric pressure. Standard hawk watch parameters widely used for precipitation and height of flight were adopted (Bildstein et al. 2007).
Weather data
We obtained weather data within the breeding range of the Hook-billed Kite north of the count sites from the TerraClimate dataset for 1995–2019 count years (Fig. 1). These weather data were extracted because this is the presumed breeding range and origin of the migratory Hook-billed Kite populations that pass through eastern Mexico and Belize on their annual southbound autumn migration (Fig. 1). Most of the breeding range north of the Belize count site includes the eastern foothills and Gulf Coast Plain of Mexico, extreme southern Texas, Yucatán Peninsula in Mexico, Petén in northern Guatemala, and all but extreme southern Belize (Greeney and Boesman 2022).
TerraClimate is a monthly, high-spatial-resolution (4-km grid) weather dataset spanning the years 1958–2019 (Abatzoglou et al. 2018). It uses climatically aided interpolation, combining high-spatial resolution climatological normals from the WorldClim dataset (Abatzoglou et al. 2018). The spatiotemporal aspects of TerraClimate were validated from weather station data, which showed a noted improvement in error and increased spatial accuracy relative to other datasets (Abatzoglou et al. 2018). Therefore, these TerraClimate data were used for this ecological modeling analysis. The weather (explanatory) variables that we extracted from the dataset were precipitation (monthly total), minimum temperature (monthly mean), and maximum temperature (monthly mean).
We randomly selected TerraClimate weather grids where there were confirmed eBird records (Sullivan et al. 2009) of Hook-billed Kites during the breeding season (May–August) to ensure that these were potential breeding areas. March–April was not included because records may coincide with migrating individuals, not breeding kites. For the Belize count, the sample size was 50 weather grids, of which 20 were in the Mexico Gulf Coast Plain, 10 in the Yucatán Peninsula, and 20 in Petén, Guatemala and Belize (Fig. 1). Ten weather grids for each biogeographic region appeared representative of the region. For the Veracruz count, the sample size was 10 weather grids, which were representative of the breeding range north of the count site, since it was smaller and only included the north Gulf Coast Plain.
Data analyses
Statistics
We standardized the count data by count effort (number of kites counted in a count season per total hours counted), which was the response variable for the magnitude of the Hook-billed Kite migration. We described the seasonal passage window of the Hook-billed Kite migration as the period during which the central 95% of individuals passed through the count sites (Rodríguez-Santana et al. 2014). We excluded the 0 to 2.5% and 97.5% to 100% tails of the distribution (very early and very late migration) of the migration to avoid using outlier observations. However, the full passage window is reported to include all migrating individuals from the date of the first Hook-billed Kite observed to the last individual observed.
We conducted analyses of the sex, age, and morph class data for Belize but not for Veracruz. We used a Chi-square test for goodness of fit for statistical comparison of the female-male sex ratio. Observed values were the number of identified individuals of each sex and the expected values were calculated based on a 1:1 sex ratio. We used a Kruskal-Wallis non-parametric rank sum test to compare hourly migration rates during the daily passage window in Belize. We compared the timing of migration using the 50% and 95% seasonal passage window. The count dates for each class (age and sex) were converted to Julian days to obtain continuous distributions. The Belize migration timing distributions of the Julian day of the 10%, 50%, 90% passage for each age and sex class were compared using Mann-Whitney U-tests over a 5-year period (2016–2020) to determine if Hook-billed Kites displayed a differential migration. Structure data (age, sex, morph) and flock sizes were not collected in 2013 and commenced in mid-2014. The 2015 season was excluded in analysis because the count started later, 15 October, compared to 1 October from 2016 to 2020.
Assessing the effects of weather
We used an information theoretic approach and generalized linear models to determine how weather influenced the movement of Hook-billed Kites on their breeding grounds pre-migration. We used a Gaussian distribution for Veracruz and Belize data because the response variables were normally distributed. We treated the two Veracruz count stations as a single site given that they were at the same latitude and only 9.5 km apart, which precluded double counting kites from the same breeding flow of migrants (Ruelas Inzunza et al. 2010b). Therefore, the counts are referred to as Veracruz and Belize.
We tested variables for relatedness to avoid multicollinearity and model dredging issues. We did not include variables that were highly or relatively highly correlated (r > 0.5) in the same set of candidate models. For each count site and response variable, we developed a set of a priori models based on ecological knowledge and theory (Anderson and Burnham 2002, Lukacs et al. 2007, Burnham et al. 2011). We used two response variables (magnitude and migratory timing) at count sites (Belize and Veracruz) to build four sets of candidate models and ranked each to determine which models were most supported by the observed data (Table 1).
The predictor variables we analyzed included precipitation for Belize and precipitation and temperature for Mexico. We omitted temperature as a predictor for Belize to reduce model dredging because of the high number of variables (Anderson and Burnham 2002) from the regional factors, and because we determined that precipitation was the more ecologically important variable and showed greater variation than temperature in all regions (Li and Meng 2017). Specifically, land snails, the main prey of the Hook-billed Kite, are moisture dependent and influenced by precipitation more than temperature, making precipitation a critical environment factor for kite presence (Barrientos 2000, Perez et al. 2021). Because of the lack of variation in monthly minimum and maximum temperatures, we used the difference between the maximum and minimum temperature of the annual monthly means to evaluate the changes between years and determine if the magnitude change influenced the timing and the number of kites that passed over the Veracruz sites. Changes in temperatures may impact soil moisture and weather patterns that could influence snail abundance, directly impacting Hook-billed Kite movements. We accumulated the monthly precipitation totals for each time window to obtain the total precipitation for that time frame.
For the weather variables, we analyzed four time frames and three regional factors of the breeding grounds north of the count sites (Table 1) to determine if weather patterns during a specific time of year and in a specific location seemed to influence the Hook-billed Kite migration. The time frames for weather data included (1) the full annual cycle (January–December of the count year), (2) the prior year (January–December of the year prior to the count), (3) breeding season (May–August), and (4) one month prior to the onset of migration (September for Belize and August for Mexico). We evaluated these windows to determine if precipitation during a specific time triggered individuals to migrate, thereby influencing the timing and/or magnitude of migration. To potentially delineate the breeding origins and identify environmental triggers on the breeding grounds of the migratory kites counted in Belize, we included three regions as factors in the analysis: (1) the Mexico Gulf Coast Plain’s, northernmost region; (2) the Yucatán Peninsula; and (3) Petén, Guatemala, and Belize north of the count site (Fig. 1). We used these region factors for Belize because the specific breeding region of the migrating birds is unknown, and these regions were delineated by distinct bioregions. For Mexico, the breeding area north of the Veracruz count is relatively small and only includes the north Gulf Coast Plain; thus, data for this region are only included in the Veracruz count analysis.
We ranked all sets of models using Akaike’s Information Criteria (AIC; Akaike 1973), adjusted for small sample size (AICc) to select the most parsimonious models (Anderson and Burnham 2002), which was achieved by evaluating AICc values and Akaike weights (wi) in the R package MuMIn (Bartoń 2019). We considered a sample small if it had < 40 samples (count years; Burnham and Anderson 2002). We created a confidence set of models for each set with a ΔAICc < 2 of the best fitting models (Burnham and Anderson 2002). In these analyses, we used R version 3.6.3 and considered P ≤ 0.05 as statistically significant (R Core Team 2020). We made all figures using the R package ggplot2 (Wickham 2016).
RESULTS
Belize migration
During a total of 3093 hourly counts (536 days, a mean of 67 days/season) over 8 years (2013–2020) in Belize, we counted 39,928 Hook-billed Kites (12.9 individuals/h) moving past the count site. The mean (± SE) annual count of Hook-billed Kites in Belize was 4991 ± 1083 kites/year (N = 8 year). The highest annual count was 9755 kites in 2020 during 405.6 h, which equates to 24.1 kites/h, while the lowest annual count was 744 in 2013 during 510.5 h, which equated to 1.5 kites/h. A significant positive trend in Hook-billed Kite migration numbers was observed in Belize from 2013 to 2020 (F1,6 = 16.45, R² = 0.73, P = 0.007); Fig. 2a). The single day high count over 8 years in Belize was 1610 on 7 November 2020 (Table 2), followed by the second largest single day count of 1441 on 8 November 2020. These two dates followed many consecutive days of rain across Belize starting from 12 October followed by Hurricane Eta on 3–6 November, which grounded Hook-billed Kites during the first movement of kites past the count site. The next three largest single day counts were 1101 on 11 November 2018, 981 on 11 November 2019, and 921 on 19 November 2018. The mean peak day was 11 November (N = 8) with a mean (± SE) of 378.6 ± 157.2 kites.
The mean 95% seasonal passage window of Hook-billed Kites that flew over the Belize count site was 44.9 d ± 2.5 (N = 8) from 26 October to 9 December with little variation (Table 2). The mean full passage window (total days of kites observed over the season) in which kites were migrating through Belize was 68.3 d ± 2.8 and the mean 50% passage (median) was 15 November (N = 8).
Hook-billed Kites in Belize did not migrate in equal numbers throughout the daily passage window (χ² = 34.4, df = 7, P < 0.001). They were observed in all count hours (0800–1600 H), but 63.5% of kites passed between 900 and 1200 H and the peak (23.7% of all kites counted) was from 1000 to 1100 H (7 yr, N = 34,271 kites). Following 1100 H, activity slowed throughout the day (Fig. 3). Kite movement was primarily in a northeast to southwest direction during all years (2013–2020) with some deviation as flocks on occasion would circle back north or move west as they appeared to be searching for thermals or wind currents before continuing in the usual direction. Movement was primarily directly over or within 500 m of the count site and often kites would move through the base of cumulus clouds estimated to be between ~1000 and 2000 m.
The height of flight varied throughout the day. Typically, during the morning hours kites were low flying (~50–300 m above the ground) and the height of flight increased throughout the day where birds were difficult to spot, even with the aid of binoculars. Migrating kites were either soaring with little flapping-flight in a thermal (kettling; Figs. 4a, 4c) or in a glide with occasional flapping (streaming; Fig. 4b). Flock size varied annually from 1 to 217 individuals (Table 3). The largest flock of 217 was observed on 12 November 2020. In 7 years (2014–2020) there were 8 flocks that were over 100 kites: 150 on 19 November 2018, 136 on 2 November 2019, 110 on 8 November 2020, 106 on 24 November 2020, 105 on 19 November 2018, 100 on 11 November 2018, and 100 on 19 November 2018. The mean (± SE) flock size was 7.6 individuals ± 0.4 (N = 4490). A Pearson’s r data analysis revealed a strong positive correlation, r(5) = 0.79, P = 0.03, between the annual mean flock size (M = 7.6, SD = 1.8, N = 7) and annual kite count (M = 5597.7, SD = 2739.2, N = 7).
Of the 34,098 Hook-billed Kites counted in Belize from 2015 to 2020 (N = 6 year), 24.2% (8264 individuals) were identified to sex and 40.3% (13,700 individuals) were identified to age (juvenile or adult; Figs. 4d-h). The sex ratio was significantly skewed toward females with 54.1% females making up the annual mean passage (N = 8264, χ² = 56.3, df = 1, P < 0.001). The age ratio was skewed toward adult kites, with annual counts averaging 71.0% adults and 29.0% juveniles. We found a positive correlation between juveniles during years where more kites were counted, but not statistically significant r(4) = 0.47, P = 0.35. The proportion of juveniles counted annually varied between 0.149 and 0.434. We found no difference in the migration timing of adults and juveniles (10%: U = 8.0; P = 0.398, 50%: U = 11.0; P = 0.830, and 90%: U = 14.0; P = 0.833), or between females and males (10%: U = 13.5; P = 0.913, 50%: U = 12.0; P = 1.000, and 90%: U = 16.5; P = 0.443). They migrated in mixed flocks throughout the entire migration season (Figs. 4a-c, Fig. 5).
The mean (± SE) percentage of dark morph Hook-billed Kites (Figs. 4f, 4h) in the migratory population in Belize from 2014 to 2020 that were identified to morph was 13.6% ± 0.7 (N = 16,061). The percentage of individuals identified to morph each year ranged from 25.1 to 81.8% with a mean of 53.9% ± 9.4.
Veracruz migration
During a total of 42,531 hourly counts from 1995 to 2019 (25 years) at the Chichicaxtle and Cardel count sites in Veracruz, 3870 Hook-billed Kites were counted from 20 August to 20 November, which was a rate of 0.09 kites/h. The mean (± SE) annual count of migrating Hook-billed Kites in Veracruz (two sites pooled) was 154.8 kites/year ± 12.1 (N = 25). The highest annual count was 299 kites in 2000 during 1699.8 h, equating to 0.18 kites/h and the lowest annual count was 27 in 2013, a rate of 0.02 kites/h. Interestingly, the two lowest counts in Veracruz and Belize occurred in 2013 (Veracruz: 27, Belize: 744). We found no significant trend in migratory kite numbers at the Veracruz count sites from 1995 to 2019 (F1,23 = 1.35, R² = 0.06, P = 0.26; Fig. 2b). The highest single day count over the 25 years in Veracruz was 51 individuals on 18 October 2001. The high count for the Cardel site was 35 individuals on 28 September 2000, and for the Chichicaxtle site 23 individuals on 18 October 2001. The mean peak day was 14 October (N = 25, Fig. 6) with a mean of 6.6 ± 0.9 kites.
The mean 95% seasonal passage window of Hook-billed Kites that passed over the Veracruz count sites was from 13 September to 11 November (29 d, N = 25) and the mean 50% passage (median) was 14 October (N = 25), which was the same day as the peak day. The earliest migrating Hook-billed Kite was recorded on 23 August and the latest on 20 November. From flock size data collected between 1992 and 2001 (N = 873 flocks), we found most Hook-billed Kites migrated as single individuals, pairs, or small groups of 3–4 individuals, and the maximum number of kites ever observed in a single flock was 12 individuals.
Influence of weather in Belize and Veracruz
Precipitation did not explain the variation in the magnitude (kites/h) of kites passing over the count site in Belize on their post-breeding migration (Table 4). Of the 17 a priori candidate models for precipitation influencing kite numbers, the model best supported by the data was the null model (Wi = 0.36). The only other model that was below the acceptable < 2 ΔAICc was the model including annual precipitation in the Gulf Coastal Plain in eastern Mexico (ΔAICc =1.54, Wi = 0.17; Table 4). The candidate models of precipitation on the timing of Hook-billed Kite movements southward over the Belize count site in the various regions of the breeding grounds were not supported by the data and did not explain the variation of when kites moved (Table 4).
Likewise for the Veracruz sites combined, the top model with respect to the magnitude of kite migration was the null model (Wi = 0.19). But four other models were below the acceptable < 2 ΔAICc threshold and these top 5 models explained 65% of the variation (Table 4). With respect to the timing of Hook-billed Kite migration in Veracruz, two models explained 88% of the variation and were below the < 2 ΔAICc threshold (Table 4). The top model was the total annual precipitation of the count year north of the count-site (north Gulf Coast Plain; Wi = 0.53). The second model was cumulative annual temperature and cumulative annual rainfall (Wi = 0.35). When accounting for temperature and rainfall, we found that the annual precipitation north of the Veracruz count site was positively correlated with the median Julian day of Hook-billed Kite migration in Veracruz (R² = 0.33, P = 0.003); thus, kites stayed longer on their breeding grounds in years with greater precipitation.
DISCUSSION
This study is the first report of migration of the Hook-billed Kite and largest known movement aggregations of the species. Our results provide a robust documentation of significant, directional, post-breeding migration of the Hook-billed Kite using 8–25 years of standardized migration counts at two localities near the northern limit of its range. We found that precipitation on the breeding grounds was not important in predicting the timing or magnitude of the Hook-billed Kite migration we documented through Belize, but this may be due to small sample size. However, we found that models including annual precipitation were supported by the count data, where increased annual precipitation was correlated with delayed migration timing in Veracruz. Additionally, we did not detect signs of differential migration between age, sex, and color morph classes, which is uncommon among raptors.
The Veracruz data provided further evidence of directional, post-breeding migratory movements of the Hook-billed Kite, however, at a lesser magnitude than in Belize (Table 5). Given southeastern directionality of the movement, the post-breeding seasonality, and consistency of this phenomena across the years, we posit that within the northern extent of its range, the Hook-billed Kite is a partial migrant, having both migratory and non-migratory individuals.
The number of Hook-billed Kites counted annually in Belize is > 30 times larger than the two Veracruz sites combined (the latter represents only 3% of the number of birds observed in Belize). The differences in the number of Hook-billed Kites observed at migration count sites in Veracruz and Belize may be explained by the geographic origin of kite populations in Middle America. Veracruz documents the largest concentration of migratory raptors of any one site in the world (Ruelas Inzunza 2007), but a considerably higher proportion of migrant Hook-billed Kites are observed in southern coastal Belize. These differences are not unexpected for several reasons. For example, the Veracruz count sites are closer to the northern extreme of this species’ range. Consequently, the kites recorded in central Veracruz likely originate in northern Veracruz, Tamaulipas, Nuevo León, or the southern Texas region because of their southern movement where drier conditions and resource scarcity may limit the density of breeding kites (Montiel de la Garza and Contreras-Balderas 1990, Clark 2002, 2003, Brush 2005). Thus, the pool of birds available to pass by the Veracruz sites is much smaller, given the smaller breeding area to the north, compared to the Belize site.
We originally hypothesized that the Hook-billed Kites counted in Veracruz may also be passing through Belize because the median date in Veracruz is nearly a month earlier than the passage in Belize (14 October versus 11 November, respectively; Fig. 6), thereby adding to the Belize numbers.
The distance between the Veracruz and Belize sites is ~1000 km. Capitolo et al. (2020) tracked migrating Broad-winged Hawks (Buteo platypterus), where daily straight-line migration distances were between 110 and 265 km. If Hook-billed Kites migrate at approximately the same rate, then kites in Veracruz would reach Belize in 4–10 d, the estimated lag time between sites based on distance. Although it is plausible that the Veracruz Hook-billed Kites are moving through Belize and are counted at all migration monitoring sites, the much greater numbers of kites passing through Belize and its geographic position due east of Veracruz make this unlikely. Most or all the Hook-billed Kites migrating through Belize migrate on a different schedule and represent a larger breeding pool that encompasses the Yucatán Peninsula and perhaps the southern Gulf Coast Plain south of Veracruz as well. The timing of Hook-billed Kite migration was also consistent across years at both the Veracruz and Belize count sites. As quantified by the passage windows, the difference in both timing and numbers also supports that kites recorded in Veracruz and Belize belong to different populations.
Based on observations of Hook-billed Kites from 1999 to 2001 in southern Belize, Jones (2002) estimated that as many as 5000 Hook-billed Kites could be migrating each year through the region and concluded that this kite is the most common autumn raptor migrant in Belize. Our findings in Belize provide a robust confirmation of Jones (2002) hypothesis based on 8 years of observations gathered through standardized field methods. Further, we have provided a quantitative characterization of the migration phenology at the daily and seasonal timescales and elucidate significant interannual variation in number of kites observed at the Belize study site. The counts were higher in years with a greater number of juvenile kites observed, however, this does not account for the extreme variation among years.
Interestingly, very few observations of collective movements by Hook-billed Kites have been observed south of Belize (Porras-Peñaranda and McCarty 2005, McCrary and Young 2008, de Melo Dantas et al. 2018, Tenorio et al. 2020). Hook-billed Kites that migrate through Belize spend the non-breeding season at an unknown location south of Belize, probably mixing in with resident populations, and thus, going undetected. Tenorio et al. (2020) observed flocking and movements of Hook-billed Kites for the first time in Costa Rica in 2018; however, this report was on 8 September, which is much earlier than our observations within Belize, or even Mexico. McCrary and Young (2008) observed a single individual in September in Nicaragua that appeared to be migrating, which represents the only known observation of movement in Nicaragua. Therefore, the migrating populations that are observed in Veracruz and Belize have not been detected migrating south of Belize. These September observations south of Belize may be movements by local breeding populations too, because these kites do regularly flock (Paulson 1983), however, more study is needed to better understand this.
The presence of some Hook-billed Kites year-round in the Rio Grande Valley of Texas suggests that at least the population at the northern end of its range is a combination of facultative migrants and resident individuals (Brush 2005, Clark and Schmitt 2017, Greeney and Boesman 2022). Further, most obligate migratory raptor species in both tropical and boreal regions exhibit a differential migration where the timing of age and sex classes differ (Kjellén 1992, Goodrich and Smith 2008, Hull et al. 2012, Vansteelant et al. 2020). In Belize, we found no difference in the timing between age and sex classes, which supports the suggestion that the northern populations are facultative migrants. Last, the species employing a facultative migration strategy could account for the significant annual variation in the number of kites that migrate through Belize each fall. It appears that in good prey years kites do not need to migrate, or not as many, and the lack of differences between the timing of age and sex classes suggests that resources are in short supply and so all ages and sexes migrate south in search of snails.
Documenting Hook-billed Kite migration in Middle America provides an opportunity to explore how tropical raptor species employ intratropical migratory movements in response to environmental conditions and other drivers, such as prey availability. The highly specialized ecology of the species and its close predator-prey association with land snails presents a powerful model for evaluating the influence food resources may have on the migration dynamics described in this study. In the Yucatán Peninsula, which is the largest area with suitable breeding habitat for Hook-billed Kites, deforestation is a major concern that is currently or could become a threat to Hook-billed Kite populations and movement patterns (Ellis et al. 2017). Land snails have limited mobility and are highly sensitive to environmental changes, such as forest loss and fragmentation (Forys et al. 2001; D. Dourson, personal communication; B. Breure, personal communication). Because Hook-billed Kites critically rely on the availability of snails across their annual cycle (Phillips et al. 2022), arboreal snail declines in heavily deforested areas may negatively impact Hook-billed Kite populations in the future (D. Dourson, personal communication).
Our findings support the idea that Hook-billed Kite migration is complex. Therefore, we categorize the Hook-billed Kite as a partial migrant, and these migratory patterns may vary considerably at the local population level, as is the case in many avian species (Chapman et al. 2011). Furthermore, because these regular, predictable, seasonal movements occur within the tropics, and their breeding range is mostly restricted to the tropical zone, we can safely designate Hook-billed Kites as an intratropical migrant. However, the factors that trigger Hook-billed Kite migration and influence their annual movement patterns in certain parts of their range requires further investigation. Future research priorities could explore the ecological drivers behind the variation in numbers of birds migrating each year and track movements between breeding and non-breeding sites.
From a conservation perspective, improving our understanding of the non-breeding component of the Hook-billed Kite’s annual cycle, including the distribution, habitat requirements, and the movement ecology will inform land protection efforts and reserve establishment. Importantly, understanding Neotropical land snail ecology and their response to habitat fragmentation and changing precipitation patterns will also assist in deciphering the complex migration ecology of the Hook-billed Kite and help determine how anthropogenic factors, such as habitat loss and climate change, are impacting regional populations of this highly specialized Neotropical raptor.
RESPONSES TO THIS ARTICLE
Responses to this article are invited. If accepted for publication, your response will be hyperlinked to the article. To submit a response, follow this link. To read responses already accepted, follow this link.
ACKNOWLEDGMENTS
We would like to thank the numerous volunteers involved with the Hawk Watch Programs in Belize and Veracruz counting raptors over the many years. We acknowledge the pioneering work by Dr. Laurie Goodrich and Stephen Hoffman, who co-founded the Veracruz River of Raptors and showed us that hawk watching in the tropics is achievable and critical. Without the financial and continued support from Rebecca Harvey, Diane and Tom Bennett, Richard Kuehn, and Dean Schuler, and Katie Brown the Belize Hawk Watch would not have been possible. We greatly appreciate the generous grant and support received from the Hawk Migration Association of North America. We would like to thank Wil Maheia, Sheila Nale, Chaab’il B’e Lodge and Casitas, and Laguna Maya Lodge for providing critical local support in Belize, and Pronatura Veracruz, A.C. for their dedication to raptor research and conservation in Mexico. A special thank you to Reymundo Chen and Matthew Sacul for their passion and assistance counting raptors in Belize. We acknowledge all the information provided by our colleagues on ecology and weather; Dr. John Abatzoglou, Dan Dourson, and Dr. Bram Breure. This research was reviewed and authorized by the San Jose State University Institutional Animal Care and Use Committee (Protocol #2018-E). ERI thanks Mexico’s Consejo Nacional de Ciencia y Tecnología for fellowship 47135 of the Sistema Nacional de Investigadores and additional support from the Secretaría de Educación Pública’s PRODEP Program.
DATA AVAILABILITY
The data/code that support the findings of this study are available upon request from the corresponding authors, RAP and ERI.
LITERATURE CITED
Abatzoglou, J. T., S. Z. Dobrowski, S. A. Parks, and K. C. Hegewisch. 2018. TerraClimate, a high-resolution global dataset of monthly climate and climatic water balance from 1958-2015. Scientific Data 5:170191. https://doi.org/10.1038/sdata.2017.191.
Akaike, H. 1973. Information theory and an extension of the maximum likelihood principle. Pages 267-281 in B. N. Petrov and B. F. Csaki, editors. Second International Symposium of Information Theory. Acadamei Kiado, Budapest, Hungary.
Anderson, D. R., and K. P. Burnham. 2002. Avoiding pitfalls when using information-theoretic methods. Journal of Wildlife Management 66:912-918. https://doi.org/10.2307/3803155
Areta, J. I., and M. A. Juhant. 2019. The Rufous-thighed Kite Harpagus diodon is not an endemic breeder of the Atlantic Forest: lessons to assess Wallacean shortfalls. Ibis 161:337-345. https://doi.org/10.1111/ibi.12645
Barrientos, Z. 2000. Population dynamics and spatial distribution of the terrestrial snail Ovachlamys fulgens (Stylommatophora: Helicarionidae) in a tropical environment. Revista de Biologia Tropical 48:71-87.
Bartoń, K. 2019. MuMIn: Multi-modal inference. Version 1.43.6.
Bildstein, K. L. 2004. Raptor migration in the Neotropics: Patterns, process, and consequences. Ornitología Neotropical 15:83-99.
Bildstein, K. L. 2006. Migrating raptors of the world: their ecology and conservation. Cornell University Press, Ithaca, New York, USA.
Bildstein, K. L., J. P. Smith, and R. Yosef. 2007. Migration counts and monitoring. Pages 101-116 in D. M. Bird and K. L. Bildstein, editors. Raptor research and management techniques. Hancock House, Blaine, Washington, USA.
BirdLife International. 2020. Chondrohierax uncinatus. IUCN Red List of Threatened Species 2020: e.T22694971A168997614. https://dx.doi.org/10.2305/IUCN.UK.2020-3.RLTS.T22694971A168997614.en
Brush, T. 2005. Nesting birds of the tropical frontier: the Lower Rio Grande Valley of Texas. Texas A & M University Press, College Station, Texas, USA.
Buechley, E. R., A. Santangeli, M. Girardello, M. H. Neate‐Clegg, D. Oleyar, C. J. McClure, and Ç. H. Şekercioğlu. 2019. Global raptor research and conservation priorities: tropical raptors fall prey to knowledge gaps. Diversity and Distributions 25(6):856-869. https://doi.org/10.1111/ddi.12901
Burnham, K. P., and D. R. Anderson. 2002. Model selection and multi-modal inference: a practical information-theoretic approach. Second edition. Springer-Verlag, New York, New York, USA.
Burnham, K. P., D. R. Anderson, K. P. Huyvaert. 2011. AIC model selection and multimodal inference in behavioral ecology: some background, observations, and comparisons. Behavioral Ecology and Sociobiology 65:23-35. https://doi.org/10.1007/s00265-010-1029-6
Capitolo, P. J., L. J. Jesus, A. B. Harper, A. M. Fish, and A. C. Hull. 2020. Fall migration of radio-tagged Broad-winged Hawks (Buteo platypterus) in California. Wilson Journal of Ornithology 132:15-21. https://doi.org/10.1676/1559-4491-132.1.15
Carrete, M., J. L. Tella, G. Blanco, and M. Bertellotti. 2009. Effects of habitat degradation on the abundance, richness and diversity of raptors across Neotropical biomes. Biological Conservation 142:2002-2011. https://doi.org/10.1016/j.biocon.2009.02.012
Chapman, B. B., C. Brönmark, J. Å. Nilsson, and L. A. Hansson. 2011. The ecology and evolution of partial migration. Oikos 120:1764-1775. https://doi.org/10.1111/j.1600-0706.2011.20131.x
Clark, W. S. 2002. First nesting of dark-morph Hook-billed Kite in the United States. North American Birds 56:260-262.
Clark, W. S. 2003. Observations on nesting Hook-billed Kites in the Rio Grande Valley. Bulletin of Texas Ornithological Society 36:40.
Clark, W. S., and N. J. Schmitt. 2017. Raptors of Mexico and Central America. Princeton University Press, Princeton, New Jersey, USA. https://doi.org/10.1515/9781400885077
Colorado, G. J., M. J. Bechard, C. Marquez, and A. M. Castano. 2006. Raptor migration in the Cauca River Valley of northern Colombia. Ornitología Neotropical 17:161-172.
de Melo Dantas, S., C. E. B. Portes, E. Pinheiro, and G. M. Kirwan. 2018. A review of flocking behavior by Hook-billed Kite, Chondrohierax uncinatus, in South America. Revista Brasileira de Ornitologia 26:9-11. https://doi.org/10.1007/BF03544411
Ellis, E. A., J. A. R. Montero, I. U. H. Gómez, L. Porter-Bolland, and P. W. Ellis. 2017. Private property and Mennonites are major drivers of forest cover loss in Central Yucatán Peninsula, Mexico. Land Use Policy 69:474-484. https://doi.org/10.1016/j.landusepol.2017.09.048
Estes, J. A., J. Terborgh, J. S. Brashares, M. E. Power, J. Berger, W. J. Bond, S. R. Carpenter, T. E. Essington, R. D. Holt, J. B. C. Jackson, R. J. Marquis, L. Oksanen, T. Okansen, R. T. Paine, E. K. Pikitch, W. J. Ripple, S. A. Sandin, M. Scheffer, T. W. Schoener, J. B. Shurin, A. R. E. Sinclair, M. E. Soule, R. Virtanen, and D. A. Wardle. 2011. Trophic downgrading of Planet Earth. Science 333:301-306. https://doi.org/10.1126/science.1205106
Ferguson-Lees, J., and D. A. Christie. 2001. Raptors of the world. Houghton Mifflin, New York, New York, USA.
Forys, E. A., C. R. Allen, and D. P. Wojcik. 2001. The likely cause of extinction of the tree snail Orthalicus reses reses (Say). Journal of Molluscan Studies 67:369-376. https://doi.org/10.1093/mollus/67.3.369
Goodrich, L. J., and J. P. Smith. 2008. Raptor migration in North America. Pages 27-150 in K. L. Bildstein, J. P. Smith, E. Ruelas Inzunza, and R. R. Veit, editors. State of North America’s birds of prey series in ornithology No. 3. Nuttall Ornithological Club, Cambridge, Massachusetts, USA and American Ornithologist’s Union, Washington, D.C., USA.
Greeney, H. F., and P. F. D. Boesman. 2022. Hook-billed Kite (Chondrohierax uncinatus), version 3.0. In B. K. Keeney, editor. Birds of the world. Cornell Lab of Ornithology, Ithaca, New York, USA. https://doi.org/10.2173/bow.hobkit.03
Haverschmidt, F. 1964. Beobachtungen an Chondrohierax uncinatus (Temminck) in Surinam. Journal für Ornithologie 105:64-66. https://doi.org/10.1007/BF01671092
Hayes, F. E. 1995. Definitions for migrant birds: What is a Neotropical migrant? Auk 112:521-523. https://doi.org/10.2307/4088747
Hoffman, S. W., and J. P. Smith. 2003. Population trends of migratory raptors in western North America, 1977̫2001. Condor 105:397-419. https://doi.org/10.1093/condor/105.3.397
Howell, S. N. G., and S. Webb. 1995. A guide to the birds of Mexico and northern Central America. Oxford University Press, New York, New York, USA.
Hull, J. M., S. Pitzer, A. M. Fish, H. B. Ernest, and A. C. Hull. 2012. Differential migration in five species of raptors in Central Coastal California. Journal of Raptor Research 46:50-56. https://doi.org/10.3356/JRR-10-116.1
Jahn, A. E., V. R. Cueto, C. S. Fontana, A. C. Guaraldo, D. J. Levey, P. P. Marra, and T. B. Ryder. 2020. Bird migration within the Neotropics. Auk 137:ukaa033. https://doi.org/10.1093/auk/ukaa033
Johnson, J. A., R. Thorstrom, and D. P. Mindell. 2007. Systematics and conservation of the Hook-billed Kite including the island taxa from Cuba and Grenada. Animal Conservation 10:349-359. https://doi.org/10.1111/j.1469-1795.2007.00118.x
Jones, H. L. 2002. Central America. North American Birds 56:115-117.
Juhant, M. 2022. Raptor migration across South America. Neotropical Birding 30:35-51.
Kjellén, N. 1992. Differential timing of autumn migration between sex and age groups in raptors at Falsterbo, Sweden. Ornis Scandinavica 23:420-434. https://doi.org/10.2307/3676673
Kottek, M., J. Grieser, C. Beck, B. Rudolf, and F. Rubel. 2006. World map of the Köppen-Geiger climate classification updated. Meteorologische Zeitschrift 15:259-263. https://doi.org/10.1127/0941-2948/2006/0130
Li, T., and Q. Meng. 2017. Forest dynamics to precipitation and temperature in the Gulf of Mexico coastal region. International Journal of Biometeorology 61:869-879. https://doi.org/10.1007/s00484-016-1266-0
Lukacs, P. M., W. L. Thompson, W. L. Kendall, W. R. Gould, P. F. Doherty Jr., K. P. Burnham, and D. R. Anderson. 2007. Concerns regarding a call for pluralism of information theory and hypothesis testing. Journal of Applied Ecology 44:456-460. https://doi.org/10.1111/j.1365-2664.2006.01267.x
Marroquin, V. M. A., E. Reyes Moreno, and T. D. Ortiz. 1992. Nesting biology of three species of kite. Pages 145-152 in D. Whitacre and R. Thorstrom, editors. Maya project progress report V, 1992. The Peregrine Fund, Inc., Boise, Idaho, USA.
McClure, C. J. W., J. R. S. Westrip, J. A. Johnson, S. E. Schulwitz, M. Z. Virani, R. Davies, A. Symes, H. Wheatley, R. Thorstrom, A. Amar, R. Buij, V. R. Jones, N. P. Williams, E. R. Buechley, and S. H. M. Butchart. 2018. State of the world’s raptors: distributions, threats, and conservation recommendations. Biological Conservation 227:390-402. https://doi.org/10.1016/j.biocon.2018.08.012
McCrary, J. K., and D. P. Young. 2008. New and noteworthy observations of raptors in southward migration in Nicaragua. Ornitología Neotropical 19:573-580.
Meerman, J., and W. Sabido. 2001. Central American ecosystems: Belize. Programme for Belize, Belize City, Belize.
Montiel de la Garza, F., and A. J. Contreras-Balderas. 1990. First Hook-billed Kite specimen from Nuevo León, Mexico. Southwestern Naturalist 35:370. https://doi.org/10.2307/3671966
National Meteorological Service of Belize. 2021. Belize topographical and meteorological information. National Meteorological Service of Belize, Ladyville, Belize. https://nms.gov.bz/about/met-info/
Parkes, H. B., G. R. Wiley, M. D. Berstein, H. F. Cline, E. C. Griffin, A. Palerm, and M. C. Meyer. 2021. Mexico. Encyclopedia Britannica. https://www.britannica.com/place/Mexico
Paulson, D. R. 1983. Flocking in the Hook-billed Kite. Auk 100:749-750. https://doi.org/10.1093/auk/100.3.749
Perez, K. E., B. S. Lewis Najev, B. Christoffersen, and J. C. Nekola. 2021. Biotic homogenization or riparian refugia? Urban and wild land snail assemblages along a subtropical precipitation gradient. Journal of Urban Ecology 7:juab002. https://doi.org/10.1093/jue/juab002
Phillips, R. 2014. BRRI Raptorwatch Manual. Second edition. Belize Raptor Research Institute, Campbell, California, USA.
Phillips, R. A., J. C. Meerman, T. Boomsma, R. P. Bourbour, B. L. Martinico, R. Howe, and R. Martinez. 2022. Breeding ecology of the Hook-billed Kite, Chondrohierax uncinatus, in Belize, Central America. Journal of Raptor Research 56:220-229. https://doi.org/10.3356/JRR-21-12
Porras-Peñaranda, P., and K. McCarty. 2005. Autumn 2004 raptor migration at Talamanca, Costa Rica. International Hawkwatcher 10:3-6.
R Core Team. 2020. R: A language and environment for statistical computing (version 3.6.3). https://www.r-project.org/
Rhoda, R., and T. Burton. 2010. Geo-Mexico: the geography and dynamics of modern Mexico. Sombrero Books, Ladysmith, British Columbia, Canada.
Rodríguez-Santana, F., Y. S. Vega, M. S. Padilla, C. P. León, Y. E. T. Adán, M. S. Losada, A. M. Lescay, and Y. Rivera. 2014. Magnitude and timing of autumn Osprey migration in southeastern Cuba. Journal of Raptor Research 48:334-344. https://doi.org/10.3356/JRR-OSPR-14-02.1
Ruelas Inzunza, E. 2007. Raptor and wading bird migration in Veracruz, Mexico: spatial and temporal dynamics, flight performance, and monitoring applications. Dissertation. University of Missouri, Columbia, Missouri, USA. https://doi.org/10.32469/10355/4778
Ruelas Inzunza, E., L. J. Goodrich, and S. W. Hoffman. 2010a. Cambios en las poblaciones de aves rapaces migratorias en Veracruz, México, 1995-2005. Acta Zoológica Mexicana 26:495-525. https://doi.org/10.21829/azm.2010.263797
Ruelas Inzunza, E., L. J. Goodrich, and S. W. Hoffman. 2010b. North American population estimates of waterbirds, vultures and hawks from migration counts in Veracruz, México. Bird Conservation International 20:124-133. https://doi.org/10.1017/S0959270909990293
Sergio, F., I. A. N. Newton, L. Marchesi, and P. Pedrini. 2006. Ecologically justified charisma: preservation of top predators delivers biodiversity conservation. Journal of Applied Ecology 43(6):1049-1055. https://doi.org/10.1111/j.1365-2664.2006.01218.x
Smith, T. B. 1988. Hook-billed Kite. Pages 102-108 in R.S. Palmer, editor. Handbook of North American birds, vol. 4. Yale University Press, New Haven, Connecticut, USA.
Sullivan, B. L., C. L. Wood, M. J. Iliff, R. E. Bonney, D. Fink, and S. Kelling. 2009. eBird: a citizen-based bird observation network in the biological sciences. Biological Conservation 142:2282-2292. https://doi.org/10.1016/j.biocon.2009.05.006
Tenorio, J., V. Abarca-Fallas, and O. Ramirez-Alan. 2020. Migration of Hook-billed Kite (Chondrohierax uncinatus) in Kèköldi, Costa Rica. Spizaetus 29:14-18.
Vansteelant, W. M. G., J. Wehrmann, D. Engelen, J. Jansen, B. Verhelst, R. Benjumea, S. Cavaillès, T. Kaasiku, B. Hoekstra, and F. de Boer. 2020. Accounting for differential migration strategies between age groups to monitor raptor population dynamics in the eastern Black Sea flyway. Ibis 162:356-372. https://doi.org/10.1111/ibi.12773
Whitacre, D. F. 2012. Neotropical birds of prey: biology and ecology of a forest raptor community. Cornell University Press, Ithaca, New York, USA. https://doi.org/10.7591/9780801464287
Whitacre, D. F., and M. A. Vásquez. 2012. Hook-billed Kite. Pages 48-59 in D. F. Whitacre, editor. Neotropical birds of prey: biology and ecology of a raptor forest community. Cornell University Press, Ithaca, New York, USA. https://doi.org/10.7591/9780801464287-007
Wickham, H. 2016. ggplot2: Elegant graphics for data analysis. Springer-Verlag, New York, New York, USA. https://ggplot2.tidyverse.org
Young, C. A. 2008. Belize’s ecosystems: threats and challenges to conservation in Belize. Tropical Science Conservation 1:18-30. https://doi.org/10.1177/194008290800100102
Zalles, J. I., and K. L. Bildstein. 2000. Raptor watch: a global directory of raptor migration sites. Birdlife Conservation Series No. 9. Birdlife International, Cambridge, UK and Hawk Mountain Sanctuary, Kempton, Pennsylvania, USA.
Fig. 1
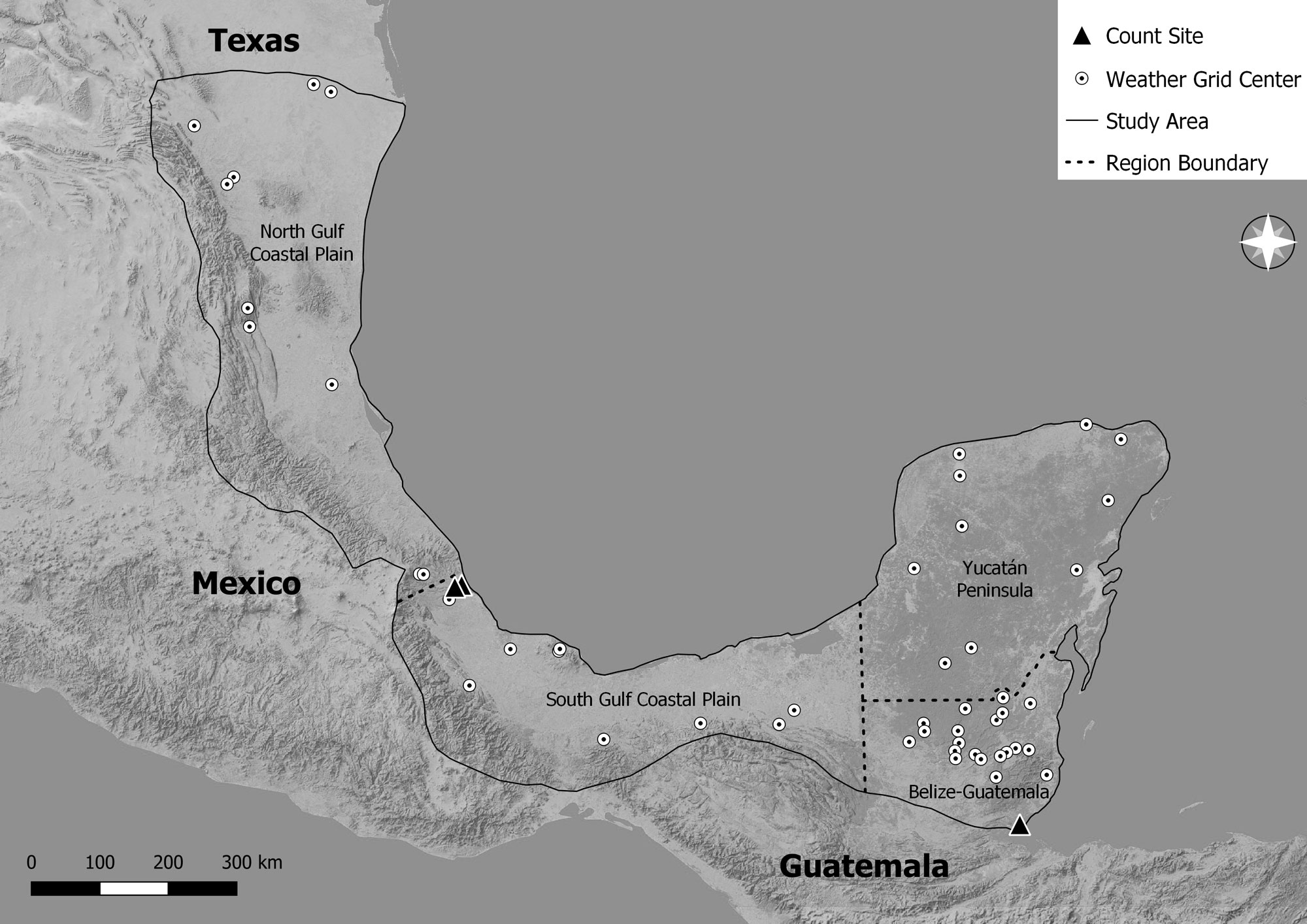
Fig. 1. The study area (outlined region) including count site locations, weather grid locations (dots represent the center of a 4x4 km grid), and regions (for Belize, the North and South Gulf Coastal Plain = Gulf Coast Plain and Mexico the North Gulf Coastal Plain only) used in the analysis.

Fig. 2
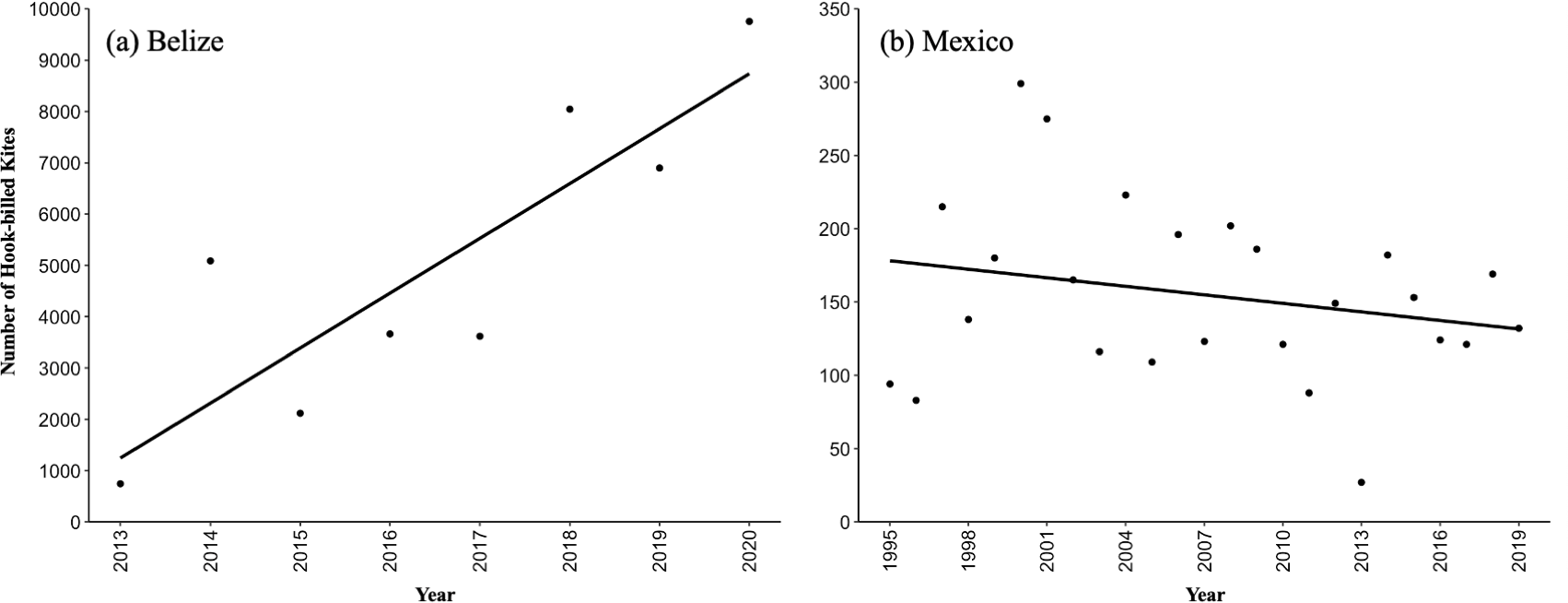
Fig. 2. (a) An increasing count trend from 2013 to 2020 (8 years) of the Hook-billed Kite (Chondrohierax uncinatus) autumn migration in Belize. (b) Population trend from 1995 to 2019 (25 years) of the Hook-billed Kite autumn migration in Veracruz, Mexico. Pooled counts from the two Veracruz count sites.

Fig. 3
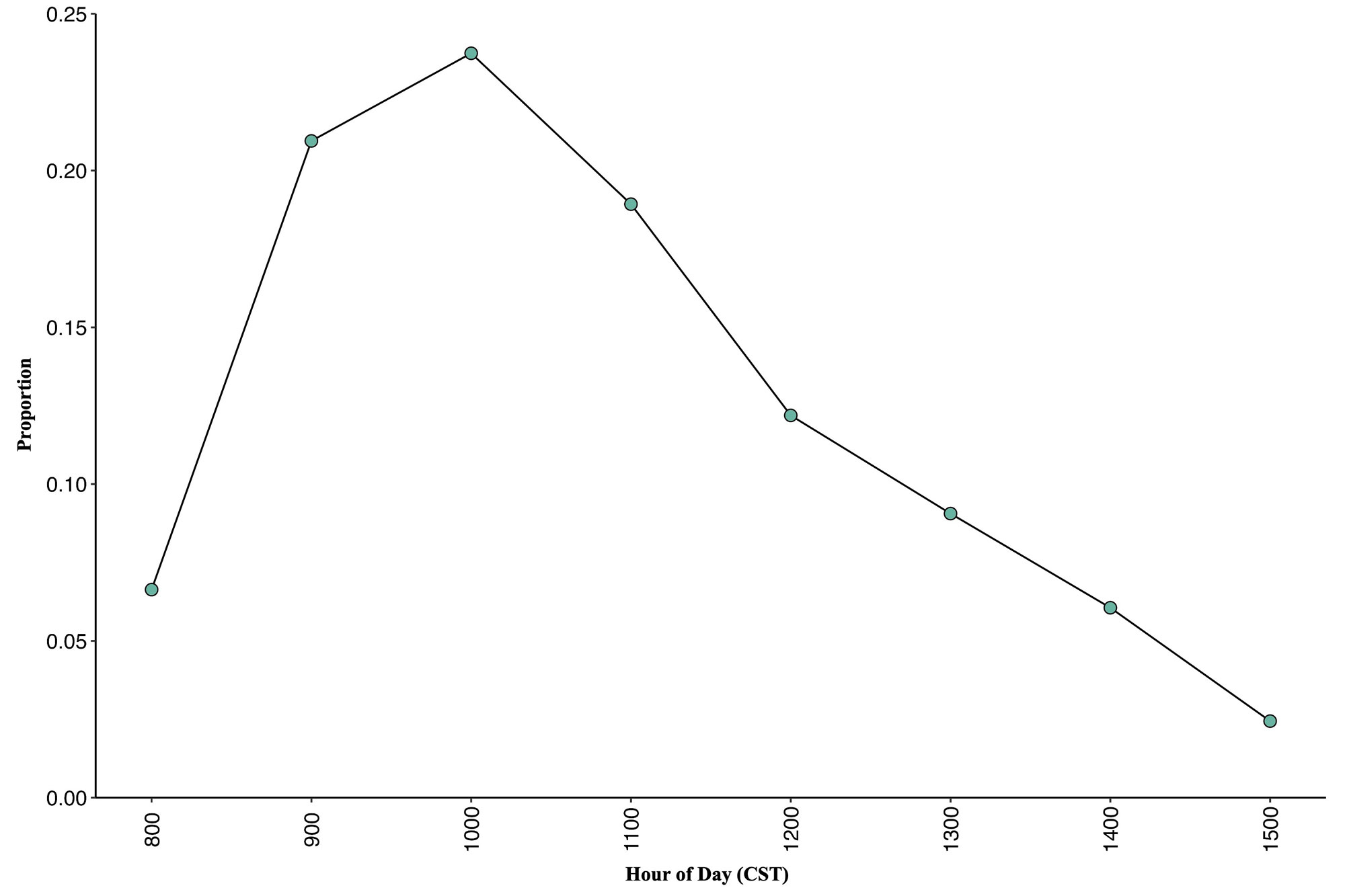
Fig. 3. Proportion of daily passage activity times of the Hook-billed Kites (Chondrohierax uncinatus) during fall migration at the Belize migration monitoring site from 2014 to 2020 (7 years, N = 34,271 kites).

Fig. 4

Fig. 4. Flocks, and age and sex differences of Hook-billed Kites (Chondrohierax uncinatus) in Belize and Mexico: (a) large migrating flock in Belize, (b) streaming flock in Belize, (c) migrating flock in Chichixactle, Mexico (photo courtesy John Garrett), (d) adult light morph female, (e) adult light morph male, (f) adult dark morph unknown sex, (g) juvenile (formative) light morph: female with rufous underwing coverts, and (h) juvenile dark morph with light banding in remiges.

Fig. 5
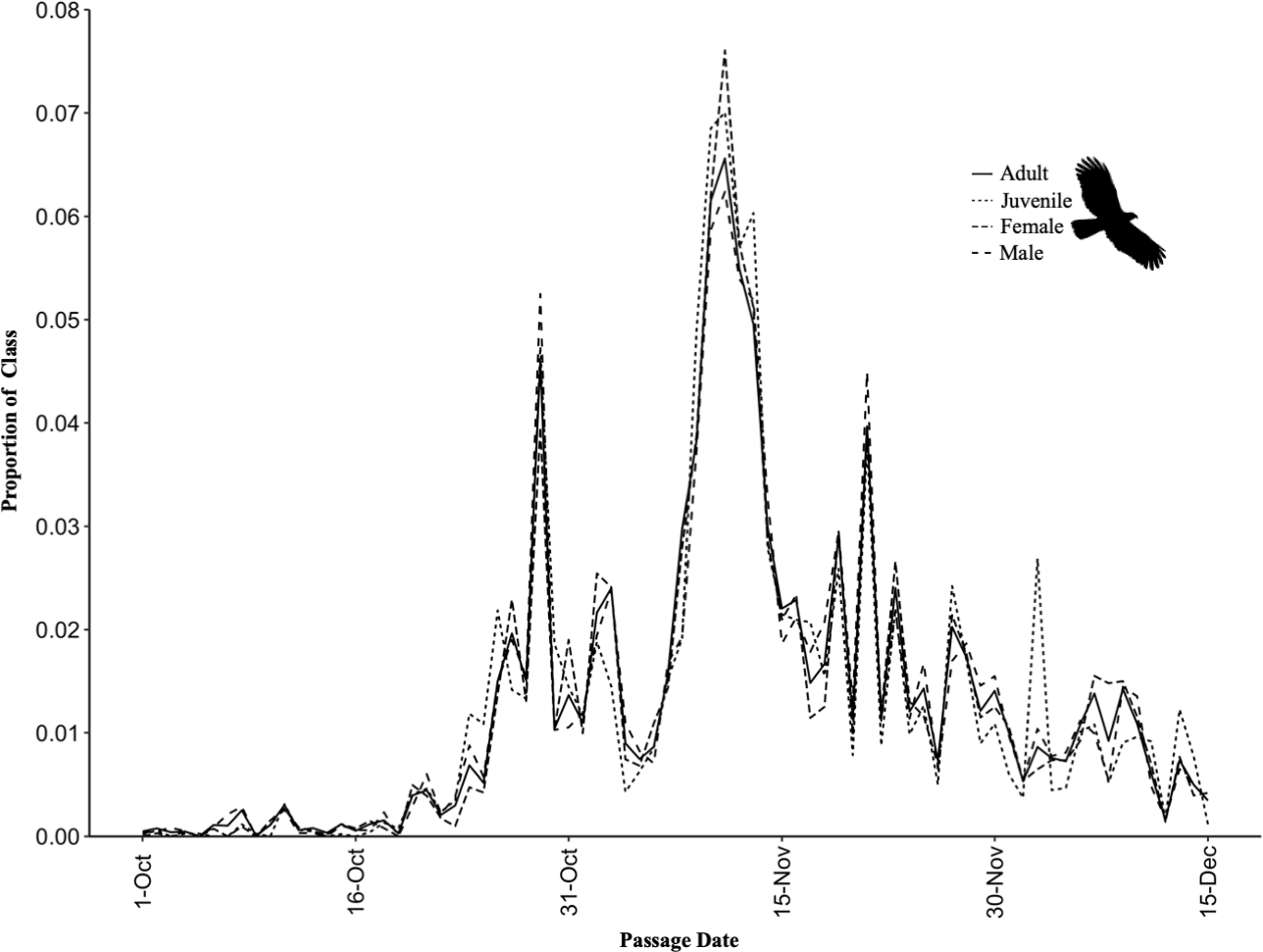
Fig. 5. Comparison of the timing (mean; N = 6 years) and proportions (individuals of class/total individuals of all class identified) of age and sex classes by migratory Hook-billed Kites (Chondrohierax uncinatus) in Belize from 2015 to 2020.

Fig. 6
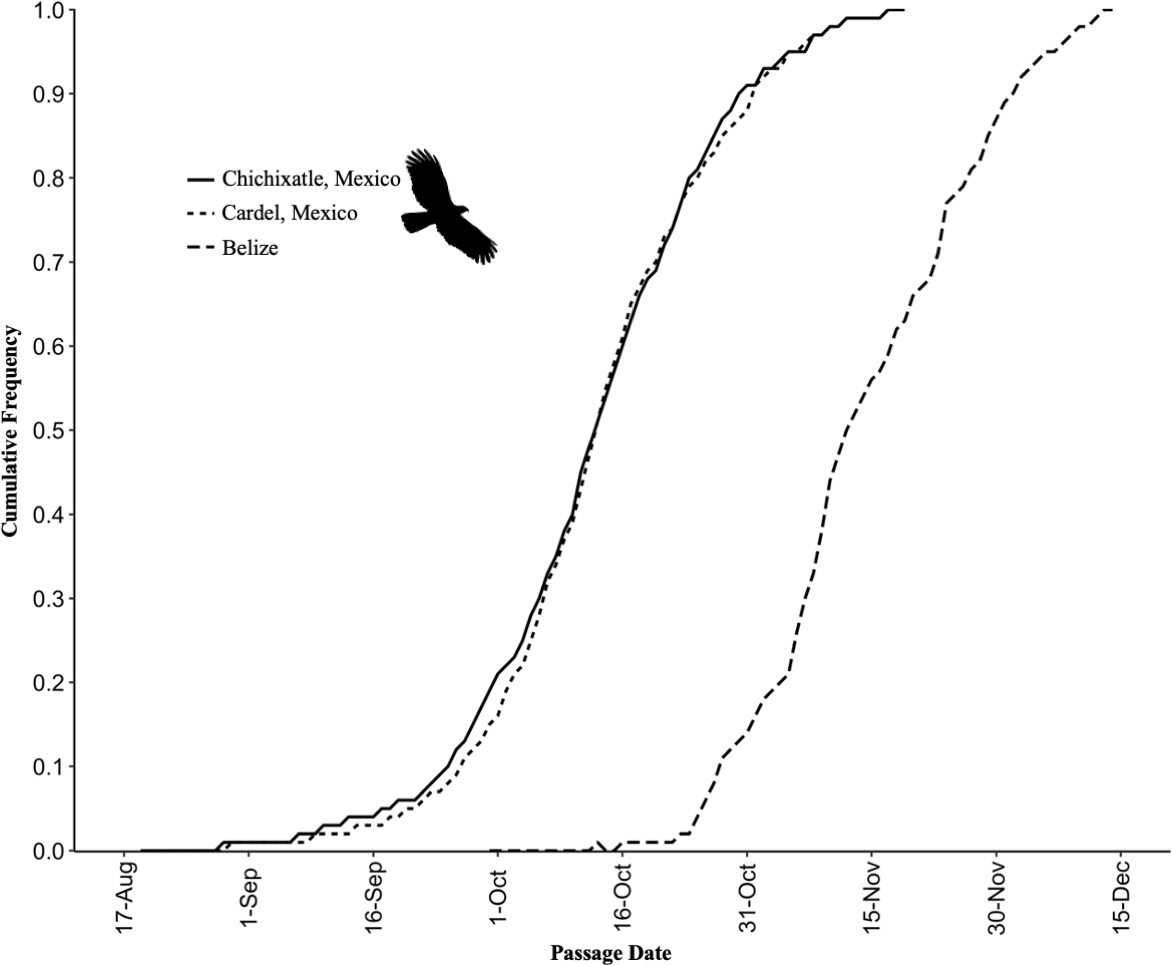
Fig. 6. Comparing the Hook-billed Kite (Chondrohierax uncinatus) timing (day of year) of the southbound migration in Belize (2013–2020) and Mexico (1995–2019).

Table 1
Table 1. Predictor and response variables, time frame factors, and regional factors used to analyze how weather on the breeding grounds influenced the Hook-billed Kite (Chondrohierax uncinatus) migration in Belize (BZ) and Mexico (MX).
| Response | Predictor | Time Frame Factors | Region Factors (Site) |
| Magnitude (kites/h) | Precipitation (BZ, MX) | Year/Annual (Jan–Dec) | North Gulf Plain (MX) |
| Timing (median) | Temperature (MX) | Year Prior (Jan–Dec) | Gulf Coast Plain (BZ) |
| Breeding (May–Aug) | Yucatán (BZ) | ||
| Month Prior (Sep or Aug)† | Guatemala/Belize (BZ) | ||
| †For Belize, September was the month prior to the initiation of migration and August for Mexico. | |||
Table 2
Table 2. Seasonal passage timing of the Hook-billed Kite (Chondrohierax uncinatus) migration at the Belize count site from 2013 to 2020.
| Year | 95% Passage (D) | Total Days† |
Median (50%) |
Daily High (Count) |
| 2013 | 3 Nov–2 Dec (30) | 54 | 25 Nov | 25 Nov (143) |
| 2014 | 23 Oct–13 Dec (52) | 71 | 10 Nov | 26 Oct (479) |
| 2015 | 22 Oct–10 Dec (45) | 58 | 26 Nov | 25 Nov (377) |
| 2016 | 26 Oct–11 Dec (47) | 73 | 15 Nov | 21 Nov (412) |
| 2017 | 27 Oct–12 Dec (47) | 68 | 15 Nov | 11 Nov (366) |
| 2018 | 24 Oct–14 Dec (52) | 76 | 11 Nov | 11 Nov (1101) |
| 2019 | 28 Oct–8 Dec (42) | 75 | 11 Nov | 11 Nov (981) |
| 2020 | 26 Oct–8 Dec (44) | 71 | 10 Nov | 7 Nov (1610) |
| Mean | 26 Oct–9 Dec (45) | 68 | 15 Nov | 13 Nov (684) |
| †Total days is the total passage window where all migrating Hook-billed Kites are included. | ||||
Table 3
Table 3. Annual Hook-billed Kite (Chondrohierax uncinatus) flock size and number of southbound migrating flocks observed at the Belize migration monitoring site from 2014 to 2020 (7 years).
| Year | Range | Mean ± SE | N |
| 2014 | 1–80 | 5.3 ± 0.3 | 575 |
| 2015 | 1–45 | 5.7 ± 0.3 | 374 |
| 2016 | 1–55 | 6.7 ± 0.3 | 548 |
| 2017 | 1–59 | 7.4 ± 0.3 | 476 |
| 2018 | 1–150 | 8.1 ± 0.5 | 994 |
| 2019 | 1–136 | 9.7 ± 0.5 | 711 |
| 2020 | 1–217 | 10.0 ± 0.6 | 812 |
| Mean | 1–106 | 7.6 ± 0.4 | 641 |
Table 4
Table 4. Best fitting models (ΔAICc < 2) evaluating the effects of precipitation and temperature during various times throughout the year (annual: January–December, breeding: May–August, prior month: September for Belize and August for Mexico) on the timing and magnitude of the Hook-billed Kite (Chondrohierax uncinatus) autumn migration in Belize and Mexico.
| Response Variable | Model | k | AICc | ΔAICc | Wi | LogLik |
| Belize Magnitude (no. of kites) | Null | 2 | 48.2 | 0.00 | 0.36 | -20.588 |
| Belize Magnitude (no. of kites) | Annual Precip Gulf Coast Plain | 3 | 49.7 | 1.54 | 0.17 | -17.859 |
| Belize Timing (median passage) | Null | 2 | 52.4 | 0.00 | 0.56 | -22.702 |
| Mexico Magnitude (no. of kites) | Null | 2 | -91.7 | 0.00 | 0.19 | 48.111 |
| Mexico Magnitude (no. of kites) | Breeding Precipitation | 3 | -91.2 | 0.45 | 0.15 | 49.186 |
| Mexico Magnitude (no. of kites) | Annual Temperature | 3 | -90.8 | 0.90 | 0.12 | 48.959 |
| Mexico Magnitude (no. of kites) | Prior Month Precipitation | 3 | -90.5 | 1.18 | 0.11 | 48.818 |
| Mexico Magnitude (no. of kites) | Annual Precipitation | 3 | -89.8 | 1.87 | 0.08 | 48.474 |
| Mexico Timing (median passage) | Annual Precipitation | 3 | 136.3 | 0.00 | 0.53 | -64.585 |
| Mexico Timing (median passage) | Annual Precip +Annual Temp | 4 | 137.1 | 0.83 | 0.35 | -63.57 |
Table 5
Table 5. Comparison of Hook-billed Kite (Chondrohierax uncinatus) autumn migration totals for Belize and Veracruz, Mexico count sites during overlapping years (2013–2020).
| Year | Cardel, Mexico | Chichicaxtle, Mexico | Mexico pooled | Toledo, Belize |
| 2013 | 11 | 16 | 27 | 744 |
| 2014 | 88 | 94 | 182 | 5086 |
| 2015 | 82 | 71 | 153 | 2119 |
| 2016 | 41 | 83 | 124 | 3661 |
| 2017 | 32 | 89 | 121 | 3616 |
| 2018 | 68 | 101 | 169 | 8047 |
| 2019 | 64 | 39 | 103 | 6900 |
| 2020 | — | 68 | — | 9755 |
| Mean ± SE | 55.1 ± 10.6 | 70.1 ± 9.6 | 125.6 ± 19.6 | 4991 ± 1083 |








