The following is the established format for referencing this article:
Grunst, M., A. Grunst, M. Barcelo Serra, Z. Delefortrie, L. Forrette, E. Tuttle, and R. Gonser. 2023. Aberrant singing is associated with developmental stress and zero fitness in a male White-throated Sparrow (Zonotrichia albicollis). Journal of Field Ornithology 94(1):2.ABSTRACT
Oscine birdsong signals species identity, and individual quality, and functions as a cue for mate selection. Thus, producing an atypical song may have deleterious fitness effects. Nevertheless, learning during song development deems the vocal phenotype labile, creating the potential for errors, which may be elevated by developmental stress. Indeed, developmental stress has been shown to interfere with song learning in laboratory studies, but in the field, developmental conditions are rarely known, and knowledge of reproductive success is typically also lacking. We report on a male White-throated Sparrow (Zonotrichia albicollis) that we observed producing an atypical song comprised of a sparrow-like introductory phrase followed by a normal song. We banded this male as a nestling and observed potentially stressful developmental conditions that may have elevated the chances for learning errors. Specifically, this male hatched last in an asynchronous second clutch of three eggs, was structurally smaller than his siblings and fledged at nearly the latest date documented during that season. As an adult, he was unsuccessful at securing a social mate or extra-pair paternity, suggesting that females selected against his atypical vocal phenotype. Our observations of this unique individual support a connection between developmental stress, atypical song, and fitness outcomes.
RESUMEN
El canto de los oscines señala la identidad de la especie y la calidad individual, y funciona como una señal para la selección de pareja. Por ello, producir un canto atípico puede tener efectos perjudiciales para el éxito biológico. Sin embargo, el aprendizaje durante el período de desarrollo del canto hace que el fenotipo vocal sea lábil, creando la posibilidad de errores, que pueden ser elevados por el estrés del desarrollo. De hecho, se ha demostrado que el estrés en el desarrollo interfiere con el aprendizaje del canto en estudios de laboratorio, pero en el campo, las condiciones de desarrollo rara vez se conocen, y el conocimiento del éxito reproductivo también suele ser escaso. Presentamos el caso de un macho de Chingolo gorjiblanco (Zonotrichia albicollis) al que observamos producir un canto atípico compuesto por una frase introductoria similar a la de un gorrión, seguida de un canto normal. Anillamos a este macho cuando era un polluelo y observamos condiciones de desarrollo potencialmente estresantes que pueden haber elevado las posibilidades de errores de aprendizaje. En concreto, este macho fue el último en eclosionar de una segunda nidada asincrónica de tres huevos, era estructuralmente más pequeño que sus hermanos y voló casi en la fecha más tardía documentada durante esa temporada. De adulto, no consiguió pareja social ni paternidad fuera de la pareja, lo que sugiere que las hembras seleccionaron contra su atípico fenotipo vocal. Nuestras observaciones de este individuo único apoyan una conexión entre el estrés del desarrollo, el canto atípico y los resultados de la aptitud biológica.
INTRODUCTION
Passerine birdsong functions as a multifaceted signal that is used in mate selection, intra-sexual competition, and territory acquisition (Beecher et al. 2000, Byers 2007, Catchpole 2008, Botero et al. 2009, Rivera-Gutierrez et al. 2010). Genetic factors predispose individuals to learn species-typical songs from conspecific tutors (Marler 1984, 1990), while filtering out superfluous noise (Byers et al. 2013). Anatomical constraints (Thorpe 1961, Martin et al. 1995) and behavioral and ecological factors that increase conspecific associations (Beecher and Burt 2004) further limit probability of acquiring a heterospecific song, contributing to stability of song traits. Nevertheless, learning influences song characteristics (Hultsch and Todt 2004), accounting for phenomena such as song dialects (Marler and Tamura 1962, Podos and Warren 2007, Benedict et al. 2009) and culture evolution (Koetz et al. 2007), and creating potential for imitation errors (Johnston 1982, Byers et al. 2013). The potential for mistakes in song learning may be further elevated by genetic factors, such as mutations within FoxP1 and FoxP2 (Haesler et al. 2007, Chen et al. 2013) and stressful developmental conditions, which may decrease investment into brain regions involved in song acquisition (Nowicki et al. 2000, 2002).
Indeed, even in species with stereotyped and simple songs, such as the White-crowned Sparrow (Zonotrichia leucophrys), laboratory studies report learning of heterospecific song (Baptista and Morton 1981). Although documented instances of individuals producing atypical songs in the wild are few, examples exist from a number of genera (Payne 1984, Martin et al. 1995, Boves et al. 2010, Byers et al. 2013), including Emberizid Sparrows (Baptista 1988, Murray et al. 2004). These cases normally involve a heterospecific song template from a species that is closely related or occupies the same habitat type (Kroodsma 2004). The fitness consequences of singing atypical songs in the wild have rarely been investigated, and observers commonly lack knowledge regarding developmental conditions.
We report on the atypical song of a male White-throated Sparrow (Zonotrichia albicollis). This species’ typical song consists of a long, descending or ascending series of high-pitched notes (Borror and Gunn 1965), and population-typical song types are positively associated with lifetime fitness (Grunst et al. 2018). The White-throated Sparrow displays a genetic polymorphism, which is associated with differences in reproductive behaviors and song use. White-morph males are more promiscuous, sing at higher rates, and have larger song-specific brain regions than tan-morph males (Thorneycroft 1966, DeVoogd et al. 1995, Tuttle 2003, Falls and Kopachena 2010). The subject of this study is a white-morph male, which we banded as a nestling. He recruited into the population during his first breeding season and returned to the same area for a second season. Thus, we are in a unique position to examine whether developmental conditions may have increased the potential for errors in song learning and assess whether this male’s atypical vocal phenotype may have had negative fitness consequences.
METHODS
Subject and study population
We banded the focal male (fish and wildlife band: 1541-43272; color combination: FROP) as a nestling in 2013 near Cranberry Lake Biological Station (State University of New York College of Environmental Science and Forestry; 44.15N, 74.78W), Adirondack Mountains, New York. This population is the subject of a long-term study initiated in 1988. Each year, we locate all nests and check nests every two days until nestlings hatch or the nest fails. We normally measure nestling mass and tarsus length daily until nestlings are 6 days old, allowing calculation of growth rates. Unfortunately, this male’s nest was found on day 4 of the nestling stage. Thus, we reported the weight on day 5 when the male was banded. Upon banding nestlings, we also take ~80–200 μl blood samples from the brachial vein for genetic paternity analysis (Formica and Tuttle 2009, Grunst et al. 2018). Thermochrons placed in nests allowed us to confirm nest success and determine fledging date.
Song recording and analysis
We recorded the focal male’s unsolicited singing on July 15, 2014, (3.5 min) and May 14 and 18, 2015 (11.75 and 6 min), using an Olympus LS-10 recorder. In addition, the focal male’s father (1541-43117; RFRR; social father confirmed by paternity analysis) was recorded singing on June 20, 2015 (6 min). Since the father was a tan-morph male, which sings at relatively low rates (Borror and Gunn 1965, Falls and Kopachena 2010), we elicited his singing with playback.
We used Raven Pro version 1.5 (Cornell Lab of Ornithology) to create sonograms and perform analyses on frequency characteristics and durations of song components. We created sonograms using WAV files, with a sampling rate of 44,100 Hz and sample size of 24 bits. For sonogram visualization, we used a Hann window, with a 3 dB filter bandwidth of 124 Hz, a DFT size of 512, a grid spacing of 86.1, and a step size of 256. Measurements were made by making selections in the spectrogram and waveform view. After identifying possible heterospecific sources of the abnormal song segment, we used Raven’s correlation function to compare these phrases to those produced by a candidate tutor species: the Song Sparrow (Melospiza melodia), also recorded at Cranberry Lake.
We captured the focal male as an adult on July 9, 2015, using conspecific playback. Upon capture, we measured mass, tarsus length, and wing chord.
RESULTS
The focal male’s song
The focal male had a repertoire of one species-typical song, which was sometimes preceded by atypical notes. Of 29/47 (661.7%) songs recorded, 29 contained the atypical portion (18/18 on May 18, 2015, 11/19 (57.9%) on May 14, 2015, 0/10 on July 15, 2014). These songs consisted of an atypical introductory phrase of two broadband note types (Fig. 1a), a 0.22 ± 0.03 s (mean ± SE) pause, and the male’s normal, descending White-throated Sparrow song (Fig. 1a). Atypical songs contained 12.03 ± 0.59 separate notes, with 2.93 ± 0.15 atypical type 1 notes, 4.72 ± 0.47 atypical type 2 notes, and 5.13 ± 0.13 wild-type notes. Songs lasted 4.56 ± 0.14 s, with atypical elements comprising 33 ± 3 % of the total song length. The bandwidth, central frequency, and duration of atypical notes are listed in Table 1. Atypical type 1 notes are higher in frequency and have a lesser bandwidth than atypical type 2 notes (Table 1; Fig. 1a).
The atypical introductory notes are similar to certain song phrases of the Song Sparrow (Fig. 1b), which shares habitat with White-throated Sparrows at our study site. However, correlations with the Song Sparrow song in Raven were low (~0.05 U, with complete similarity indicated by 1 U). We were unable to locate other cases of atypical singing in the White-throated Sparrow when checking the acoustical collections of the Cornell Lab of Ornithology (Macaulay Library) and Xeno-Canto.
As for most White-throated Sparrows, the male’s wild-type descending song was stereotyped, with the highest note at 4306.6 Hz and the lowest at 3100.8 Hz. Wild-types songs had a relatively consistent bandwidth of 1205 Hz and lasted an average of 3.46 ± 0.06 s. In our population, male White-throated Sparrows display four distinct different singing patterns: descending (as in this male), ascending, ascending and then descending, and monotone (constant pitch). The frequency of songs is distinct between individuals and thus may serve as a signal of individual identity (Brooks and Falls 1975a, b, Grunst et al. 2018). Frequency characteristics associated with each singing pattern can be found in Table 2 in Grunst et al. (2018).
The father’s song
The focal male’s father sang two song types, with no apparent abnormalities (Fig. 1c, d). Although most White-throated Sparrows sing a single song type, singing two song types is not uncommon. His dominant song type is a descending song with a bandwidth of 1205 Hz (Fig. 1c). The father sang at a lower frequency than the focal male, with the highest note at 4134.4 Hz and the lowest at 2928.5 Hz. The father’s ascending song has a bandwidth of 861 Hz, with an initial low note produced at 2928.5 Hz followed by higher notes at 3789.8 Hz.
Developmental conditions
The focal male was one of three nestlings hatched in the second clutch of a tan-morph male by white-morph female pair (White-throated Sparrows pair disassortatively by color morph; Lowther 1961, Tuttle 2003), all of which were within-pair offspring. This clutch was initiated late in the 2013 breeding season after the male’s parents had fledged 4 nestlings. The pair’s first clutch fledged on June 18, and the second fledged on August 8. Among 40 clutches banded in 2013, the latest fledge date was August 9, the earliest June 5, and the median June 20. Many second clutches hatch asynchronously, and the focal male hatched one day later than his siblings; both were tan-morph males. The focal male was 5 days old on banding day, whereas, his nest mates were both 6 days old. On day 5, the focal male weighed 12.75 g and had a tarsus length of 20.9 mm. His 6-day-old siblings weighed 13.7 and 12.1 g and had tarsus lengths of 21.0 and 21.3 mm. The male’s weight was in the 2nd quartile for day 5 nestlings, his tarsus length in the 4th quartile, and his mass-size residuals (a metric of body condition) in the 1st quartile (Table 2a).
Fitness and breeding behavior
The focal male was unable to secure a social mate during two breeding seasons. During 2014, he may have been sighted singing in the Woosh Pond area of the study site on June 11, but the earliest confirmed sighting was July 14, after which he was frequently re-sighted in the same area. In 2015, he was observed singing early in the breeding season, on May 9, close to the area in which he was present in 2014, and appeared to have sole control of the territory.
Although White-throated Sparrows normally mate disassortatively by color morph (Lowther 1961, Tuttle 2003, Falls and Kopachena 2010), late in the 2015 breeding season, the focal male appeared to be attempting to usurp a white-morph female paired with a tan-morph male on a neighboring territory. He was observed singing immediately above the female’s second nest and closely associating with her during foraging bouts. Despite observed off-territory forays, the focal male did not obtain extra-pair paternity.
As a breeding adult, the focal male’s mass was in the 1st quartile among white-morph males, and his tarsus length at the top of the 1st quartile, his wing chord in the 4th quartile, and his mass-size residuals in the 1st quartile (Table 2b). The focal male showed no physical evidence of abnormality and appeared to have normal crown plumage for a white-morph male (Fig. 2).
DISCUSSION
Our study provides a rare example of atypical singing in the wild and, to our knowledge, the first of a White-throated Sparrow producing atypical song elements. Our focal male produced a broadband, Song Sparrow-like introductory phrase before a normal, species-typical song. Conclusively determining the source of the atypical song elements is impossible. The song’s hybrid nature suggests that this male may have interacted with a heterospecific bird, such as a Song Sparrow, during song crystallization. However, we were unable to find a close match to the abnormal portion of the vocalization. As Song Sparrow songs are diverse, obtaining a close correlation might be unlikely unless the actual tutor was recorded. Although beyond the scope of this work, a laboratory study involving acoustically training isolated nestlings with heterospecific songs could help clarify the plausibility of this explanation.
Genetics predispose species to learn species-typical song (Marler 1990), and given the role of song in female choice (Catchpole 1987, Nowicki and Searcy 2004) and signaling species identity (Marler 1957), strong selection should exist against errors. Indeed, outside of mimetic species, examples of individuals copying heterospecific song in the wild are rare (Byers et al. 2013). However, a Prairie Warbler (Setophaga discolor) was observed producing a song seemingly acquired by imitating a Field Sparrow (Spizella pusilla) (Byers et al. 2013), and a Song Sparrow was observed singing White-throated Sparrow songs (Baptista 1988). Laboratory studies of song learning have also demonstrated that isolated individuals can learn heterospecific song (Baptista and Morton 1981). In addition, a Red-eyed Vireo (Vireo olivaceus) was recorded singing a song that was completely aberrant, that is, not resembling that of any other species (Rice 1981).
Developmental stress may increase errors in song learning (Nowicki 2000, 2002), and this may have been the case for the focal male. The focal male was raised in a second clutch. In such cases, parents are often still caring for fledglings decreasing provisioning of the current brood. In our population, nestlings from second clutches are lighter on day 6 than nestlings from first clutches (Tuttle et al., unpublished data). Field observations confirm that the focal male’s father was caring for fledglings during the nestling period of the second clutch. Second clutches in our population also often hatch asynchronously. The focal male hatched one day later than his siblings, placing him at a competitive disadvantage; he was structurally smaller than his siblings on banding day and was structurally large but light in mass for a five-day-old nestling. These data are consistent with low body condition, which could indicate developmental stress.
Unfortunately, we have no measurements of physiological stress in our subject from the nestling period. However, White-throated Sparrow nestlings that are the smallest member of their brood have elevated oxidative-stress levels when compared to siblings (Grunst et al. 2019). Elevated oxidative stress may impair learning and reduce singing performance (von Schantz et al. 1999, Casagrande et al. 2014). Hatching order has also been shown to negatively affect nestling survival and body condition in many avian species (Maness and Anderson 2013).
The focal male’s late fledging date may have further increased potential for developing an atypical song by increasing physiological stress and limiting opportunities for song learning during the fledging period (Kaliński et al. 2019). To accumulate body reserves prior to migration, late-fledging nestlings may need to prioritize foraging over song learning. Further, singing of White-throated Sparrow declines late in the season, which could decrease interactions with singing conspecifics. Laboratory experiments have demonstrated that absence of appropriate social interactions with conspecifics can promote erroneous song learning (Baptista and Petrinovich 1986, Baptista 1988). The fact that the focal male’s father is tan might have further reduced exposure to conspecific song since tan-morph male White-throated Sparrows sing relatively infrequently (Tuttle 2003, Falls and Kopachena 2010).
Our data suggest that atypical singing may have deleteriously affected the focal male’s fitness as he was unable to secure a social mate or gain extra-pair paternity. This bird showed no physical abnormalities, his adult morphometric measurements were within the normal range, and he was capable of defending a territory. Therefore, atypical song and failure in mate attraction is a possible explanation for reproductive failure. However, our study is limited by the correlational nature of data and sample size of 1, which precludes statistical analysis. In contrast to our findings, Byers et al. (2013) found that a male Prairie Warbler that sang an atypical song successfully attracted a mate, but others have reported that individuals with atypical songs were unpaired (Boves et al. 2010). To our knowledge, success in acquiring extra-pair paternity has not been previously evaluated in relation to atypical singing. In the future, a behavioral mate-choice assay could be used to test whether females avoid atypical song.
In summary, our study provides a novel example of atypical singing in a male White-throated Sparrow. This individual’s atypical song was associated with potentially stressful developmental conditions and zero fitness returns. Thus, although our sample size clearly presents limitations, our data are consistent with the hypothesis that atypical song may be indicative of developmental stress, such that song serves as a reliable indicator of developmental conditions and phenotypic quality during adulthood. Results also suggest that females discriminate against these songs when choosing mating partners, hence exerting sexual selection on the vocal phenotype.
RESPONSES TO THIS ARTICLE
Responses to this article are invited. If accepted for publication, your response will be hyperlinked to the article. To submit a response, follow this link. To read responses already accepted, follow this link.
AUTHOR CONTRIBUTIONS
M.L.G and A.S.G conducted statistical analysis, analyzed songs, and wrote the initial draft of the manuscript. M.B.S., Z.D., and L.F collected and organized field data. R.A.G. and E.M.T. provided project leadership and intellectual input. All authors provided inputs on manuscript drafts and gave final approval for publication. The outline of the manuscript was formulated prior to Elaina Tuttle's passing.
ACKNOWLEDGMENTS
We acknowledge the White-throated Sparrow field crews, Cranberry Lake Biological Station, The Center for Genomic Advocacy at Indiana State University, the Tuttle and Gonser labs, C.A.T. Gonser, and Zonotrichia Zeke. Funding sources included the School of Graduate and Professional Studies at Indiana State University, the National Science Foundation (grant DUE-0934648) and the National Institutes of Health (grant 1R01GM084229 to E.M.T. and R.A.G.). Finally, we thank anonymous reviewers whose comments and critique greatly improved this manuscript.
DATA AVAILABILITY
The data are available in Zenodo. https://doi.org/10.5281/zenodo.7441124
LITERATURE CITED
Baptista, L. F., and M. L. Morton. 1981. Interspecific song acquisition by a White-crowned Sparrow. Auk 98:383-385.
Baptista, L. F., and L. Petrinovich. 1986. Song development in the White-crowned Sparrow: social factors and sex differences. Animal Behaviour 35:1359-1371. https://doi.org/10.1016/S0003-3472(86)80207-X
Baptista, L. F. 1988. Imitations of White-crowned Sparrow songs by a Song Sparrow. Condor 90:486-488. https://doi.org/10.2307/1368579
Beecher, M. D., S. E. Campbell, and J. C. Nordby. 2000. Territory tenure in song sparrows is related to song sharing with neighbors, but not repertoire size. Animal Behaviour 59:29-37. https://doi.org/10.1006/anbe.1999.1304
Beecher, M. D., and J. M. Burt,. 2004. The role of social interaction in bird song learning. Current Directions in Psychological Science 13:224-228. https://doi.org/10.1111/j.0963-7214.2004.00313.x
Benedict, L., and R. C. K. Bowie. 2009. Macrogeographical variation in the song of a widely distributed African warbler. Biology Letters 5:484-487. https://doi.org/10.1098/rsbl.2009.0244
Borror, D. J., and W. W. H. Gunn. 1965. Variation in white-throated sparrow songs. Auk 82:26-47. https://doi.org/10.2307/4082793
Botero, C. A., R. J. Rossman, L. M. Caro, L. M. Stenzler, I. J. Lovette, S. R. De Kort, and S. L. Vehrencamp. 2009. Syllable type consistency is related to age, social status and reproductive success in the tropical mockingbird. Animal Behaviour 77:701-706. https://doi.org/10.1016/j.anbehav.2008.11.020
Boves, T. J., D. A. Buehler, and P.C. Massey. 2010. Interspecific song imitation by a Cerulean Warbler. Wilson Journal of Ornithology 122:583-587. https://doi.org/10.1676/09-157.1
Brooks, R. J., and J. B. Falls. 1975a. Individual recognition by song in white-throated sparrows. I. Discrimination of song of neighbors and strangers. Canadian Journal of Zoology 53:879-888. https://doi.org/10.1139/z75-101
Brooks, R. J., and J. B. Falls. 1975b. Individual recognition by song in white-throated sparrows. III. Song features used in individual recognition. Canadian Journal of Zoology 53:1749-1761. https://doi.org/10.1139/z75-210
Byers, B. E. 2007. Extrapair paternity in chestnut-sided warblers is correlated with consistent vocal performance. Behavioral Ecology 13:130-136. https://doi.org/10.1093/beheco/arl058
Byers, B. E., B. A. Kramer, M. E. Akresh, and D. I. King. 2013. Interspecific song imitation by a prairie warbler. Journal of Field Ornithology 84:181-186. https://doi.org/10.1111/jofo.12016
Casagrande, S., R. Pinxten, E. Zaid, and M. Eens. 2014. Carotenoids, birdsong and oxidative status: Administration of dietary lutein is associated with an increase in song rate and circulating antioxidants (albumin and cholesterol) and a decrease in oxidative damage. PLoS ONE 9:e115899. https://doi.org/10.1371/journal.pone.0115899
Catchpole, C. K. 1987. Bird song, sexual selection, and female choice. Trends in Ecology and Evolution 2:94-97. https://doi.org/10.1016/0169-5347(87)90165-0
Catchpole, C. K., and P. J. B. Slater. 2008. Birdsong: Biological Themes and Variations. 2nd ed. Cambridge University Press, New York, NY, USA. https://doi.org/10.1017/CBO9780511754791
Chen, Q., J. B. Heston, Z. D. Burkett, and S. A. White. 2013. Expression analysis of the speech-related genes FoxP1 and FoxP2 and their relation to singing behavior in two songbird species. Journal of Experimental Biology 216:3682-3692. https://doi.org/10.1242/jeb.085886
Devoogd, T. J., A. M. Houtman, and J. B. Falls. 1995. White-throated sparrow morphs that differ in song production rate also differ in the anatomy of some song-related brain areas. Journal of Neurobiology 28:202-213. https://doi.org/10.1002/neu.480280207
Falls, J. B., and J. G. Kopachena. 2010. White-throated sparrow (Zonotrichia albicollis). No. 128 in A. Poole, editor. The Birds of North America online. Cornell Laboratory of Ornithology, Ithaca, NY, USA. https://doi.org/10.2173/bna.whtspa.02
Formica, V. A., and E. M. Tuttle. 2009. Examining the social landscapes of alternative reproductive strategies. Journal of Evolutionary Biology 22:2395-2408. https://doi.org/10.1111/j.1420-9101.2009.01855.x
Grunst, M. L., A. S. Grunst, V. A. Formica, R. A. Gonser, and E. M. Tuttle. 2018. Multiple signaling functions of song in a polymorphic species with alternative reproductive strategies. Ecology and Evolution 8:1369-1383. https://doi.org/10.1002/ece3.3702
Grunst, A. S., M. L. Grunst, R. A. Gonser, and E. M. Tuttle. 2019. Developmental stress and telomere dynamics in a genetically polymorphic species. Journal of Evolutionary Biology 32:134-143. https://doi.org/10.1111/jeb.13400
Haesler, S., C. Rochefort, B. Georgi, P. Licznerski, P. Osten, and C. Scharff. 2007. Incomplete and inaccurate vocal imitation after knockdown of FoxP2 in songbird basal ganglia nucleus Area X. PLoS Biology 5:e321. https://doi.org/10.1371/journal.pbio.0050321
Hultsch, H., and D. Todt. 2004. Learning to sing. Pages 80-107 in P. Marler, and H. Slabbekoorn, editors. Nature’s music: The Science of Birdsong. Academic Press, New York, NY, USA. https://doi.org/10.1016/B978-012473070-0/50006-2
Johnston, T. D. 1982. Selective costs and benefits in the evolution of learning. Advances in the Study of Behavior 12:65-106. https://doi.org/10.1016/S0065-3454(08)60046-7
Kalinski, A., M. Banbura, M. Glaalski, M. Markowski, J. Skwarska, J. Wawrzyniak, P. Zieliński, and J. Bańbura. 2019. Physiological condition of nestling great tits (Parus major) declines with the date of brood initiation: a long term study of first clutches. Scientific Reports 9:843. https://doi.org/10.1038/s41598-019-46263-z
Koetz, A. H., D. A. Westcott, and B. C. Congdon. 2007. Geographical variation in song frequency and structure: the effects of vicarant isolation, habitat type and body size. Animal Behaviour 74:1573-1583. https://doi.org/10.1016/j.anbehav.2007.03.022
Kroodsma, D. E. 2004. The diversity and plasticity of birdsong. Pages 108-131 in P. Marler, and H. Slabberekoorn, editors. Nature’s Music: The Science of Birdsong. Academic Press, New York, NY, USA. https://doi.org/10.1016/B978-012473070-0/50007-4
Lowther, J. K. 1961. Polymorphism in the White-throated sparrow, Zonotrichia albicollis (Gmelin). Canadian Journal of Zoology 39:281-292. https://doi.org/10.1139/z61-031
Maness, T. J., and D. J. Anderson. 2013. Predictors of juvenile survival in birds. Ornithological Monographs 78:1-55. https://doi.org/10.1525/om.2013.78.1.1
Marler, P. 1957. Specific distinctiveness in the communication signals of birds. Behaviour 11:13-39. https://doi.org/10.1163/156853956X00066
Marler, P., and M. Tamura. 1962. Song ‘dialects’ in three populations of white-crowned sparrows. Condor 64:368-377. https://doi.org/10.2307/1365545
Marler, P. 1984. Song learning: innate species differences in the learning process. Pages 289-309 in P. Marler, and H. S. Terrace, editors. The Biology of Learning. Springer-Verlag, Berlin, Germany. https://doi.org/10.1007/978-3-642-70094-1_13
Marler, P. 1990. Innate learning preferences: signals for communication. Developmental Psychobiology 23:557-568. https://doi.org/10.1002/dev.420230703
Martin, P. R., J. R. Fotheringham, and R. J. Robertson. 1995. A Prairie Warbler with a conspecific and heterospecific song repertoire. Auk 112:770-774.
Murray, R. L., T. P. Stanton, and V. R. Emrick. 2004. Bachman’s Sparrows mimic the vocalizations of the Common Yellowthroat and the Indigo Bunting. Journal of Field Ornithology 85:51-52. https://doi.org/10.1648/0273-8570-75.1.51
Nowicki, S., D. Hasselquist, S. Bensch, and S. Peters. 2000. Nestling growth and song repertoire size in great reed warblers: evidence for song learning as an indicator mechanism. Proceedings of the Royal Society of London B. 267:2419-2424. https://doi.org/10.1098/rspb.2000.1300
Nowicki, S., W. A. Searcy, and S. Peters. 2002. Brain development, song learning and mate choice in birds: a review and experimental test of the “nutritional stress hypothesis.” Journal of Comparative Physiology A. 188:1003-1014. https://doi.org/10.1007/s00359-002-0361-3
Nowicki, S., W. A. Searcy. 2004. Song function and the evolution of female preference: why birds sing, why brains matter. Annuals of the New York Academy of Science 1016:704-723. https://doi.org/10.1196/annals.1298.012
Payne, R. B., L. L. Payne, and S. M. Dohlert. 1984. Interspecific song learning in a wild Chestnut-sided Warbler. Wilson Bulletin 96:292-294.
Podos, J., and P. S. Warren. 2007. The evolution of geographic variation in birdsong. Advances in the Study of Behavior 37:403-458. https://doi.org/10.1016/S0065-3454(07)37009-5
Rice, J. C. 1981. Behavioral implications of aberrant song of a red-eyed vireo. The Wilson Bulletin 93:383-390.
Rivera-Gutierrez, H. F., R. Pinxten, and M. Eens. 2010. Multiple signals for multiple messages: great tit, Parus major, song signals age and survival. Animal Behaviour 80:451-459. https://doi.org/10.1016/j.anbehav.2010.06.002
Thorneycroft, H. B. 1966. Chromosomal polymorphism in the white-throated sparrow, Zonotrichia albicollis. Science 154:1571-1572. https://doi.org/10.1126/science.154.3756.1571
Thorpe, W. H. 1961. The biology of vocal communication and expression in birds. Cambridge University Press, Cambridge, UK.
Tuttle, E. M. 2003. Alternative reproductive strategies in the white-throated sparrow: behavioral and genetic evidence. Behavioral Ecology 14:425-432. https://doi.org/10.1093/beheco/14.3.425
Von Schantz, T., S. Bensch, M. Grahn, D. Hasselquist, and H. Wittzel. 1999. Good genes, oxidative stress and condition-dependent sexual signals. Proceedings of the Royal Society of London B 266:1-12 https://doi.org/10.1098/rspb.1999.0597
Fig. 1
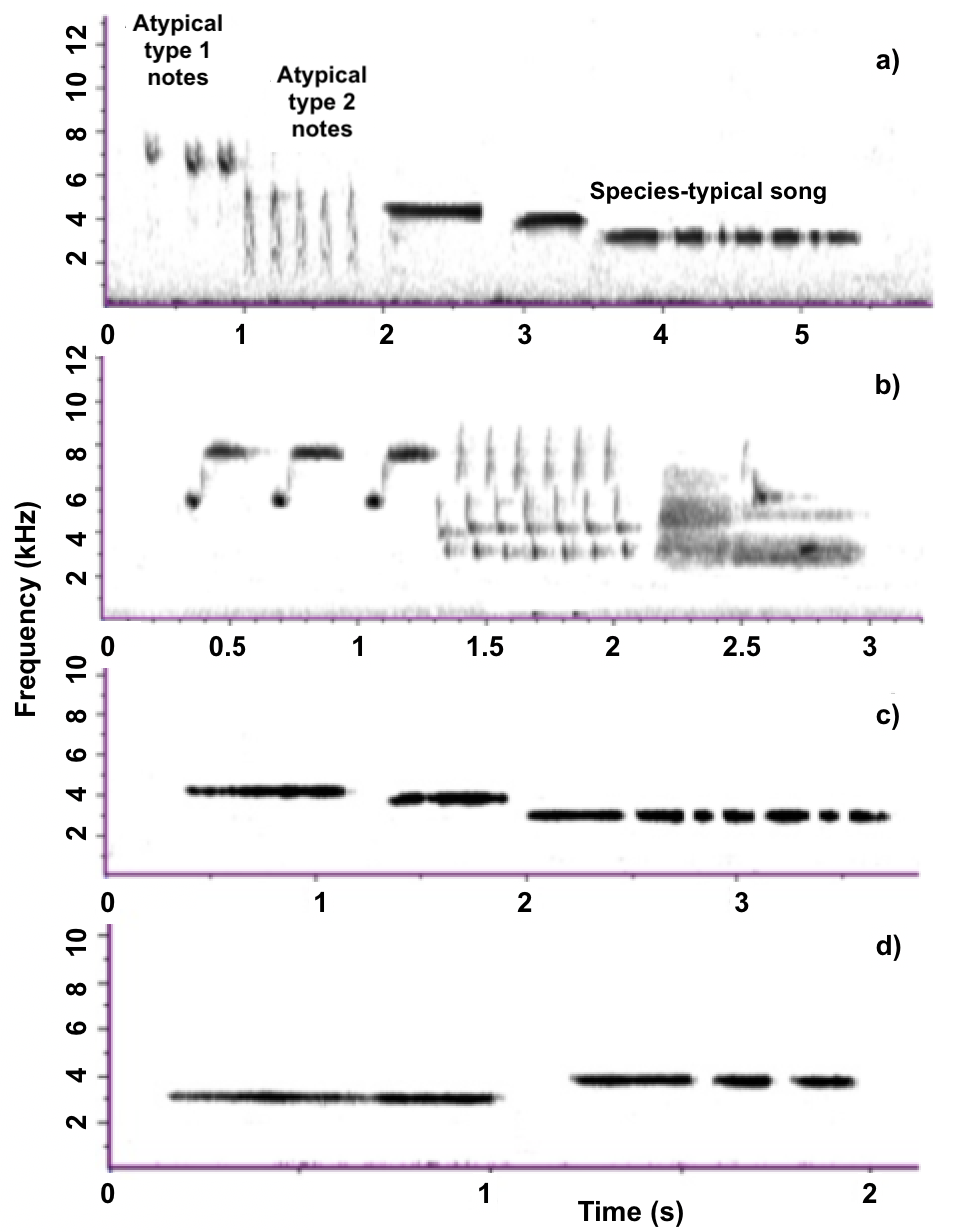
Fig. 1. (a) The focal male’s song, consisting of an atypical introductory phrase of two broadband note types, followed by a species-typical song (b) Song Sparrow song from the study site showing obtuse similarity to the introductory phrases of the focal male’s song (c) The focal male’s father’s descending song (d) The focal male’s father’s ascending song.

Fig. 2

Fig. 2. Front and side view of the focal male.

Table 1
Table 1. Frequency and duration characteristics of atypical note types
| Song element | Bandwidth (Hz) | Center frequency (Hz) | Duration of phrase (sec) |
| Atypical type 1 | 1304.80 ± 24.10 | 6572.42 ± 26.19 | 0.15 ± 0.002 |
| Atypical type 2 | 3700.02 ± 37.66 | 4715.28 ± 33.10 | 0.12 ± 0.001 |
Table 2
Table 2. Morphological measurements and condition estimates (mass-size (i.e. tarsus length) residuals) for the focal male as a nestling (a) and adult (b) relative to population-level statistics
| Focal male | Population min. | Population max. | Median | ||||
| (a) Nestling† | |||||||
| Weight (g) | 12.75 | 8.25 | 24.0 | 13.3 | |||
| Tarsus (mm) | 20.9 | 14.73 | 22.64 | 19.48 | |||
| Mass-size residuals | -1.87 | -3.58 | 10.79 | -0.129 | |||
| (b) Adult‡ | |||||||
| Weight (g) | 23.6 | 22.5 | 31.1 | 25.75 | |||
| Tarsus (mm) | 24.0 | 21.39 | 28.6 | 24.5 | |||
| Wing chord (mm) | 75.5 | 63.5 | 78.5 | 73.9 | |||
| Mass-size residuals | -1.53 | -2.71 | 3.12 | -0.092 | |||
| †N = 264 nestlings for weight, 251 nestlings for tarsus length, 229 for mass-size residuals (calculated from the equation: Mass~-2.966 (± 1.876 SE)+0.841(±0.096 SE)(tarsus length); p < 0.001; r2 (adjusted) = 0.245) ‡N = 245 observations on 187 white-morph males for weight, 208 observations on 166 white-morph males for tarsus length, 210 observations on 170 white-morph males for wing chord, 119 observations on 104 males for mass-size residuals (calculated from the equation: Mass~12.9 (± 3.31 SE)+0.514 (± 0.134 SE)(tarsus length); p < 0.001; r2 (adjusted) = 0.102) | |||||||








