The following is the established format for referencing this article:
Turner, A. M., M. E. Hauber, and D. G. Reichard. 2022. Twenty-two years of brood parasitism data do not support the mafia hypothesis in an accepter host of the Brown-headed Cowbird (Molothrus ater). Journal of Field Ornithology 93(4):4.ABSTRACT
Most host species of the obligate brood parasitic Brown-headed Cowbird (Molothrus ater) accept foreign eggs and offspring despite the associated reproductive costs. One theoretical explanation for this host tolerance is the potential for mafia-like behavior by the brood parasite, which involves the retaliatory destruction of a host’s eggs and/or nestlings in response to that host’s rejection of the parasitic progeny. Here, we use a long-term dataset of an accepter cowbird host, the Dark-eyed Junco (Junco hyemalis carolinensis), where researchers opportunistically removed parasitic cowbird eggs from the clutch, to test critical assumptions and predictions of the mafia hypothesis in this cowbird-host system. For the mafia hypothesis to be supported, at least some junco nestlings should survive in cowbird parasitized nests. In turn, if cowbirds parasitizing juncos engage in mafia behavior, then nests where the parasitic egg was removed should have lower nest success than nests where the cowbird egg remained. We found no support for the mafia hypothesis, because fledgling success was higher in nests where the cowbird egg was removed compared to nests where the cowbird egg remained. If mafia behavior is present in this cowbird population, as suggested by anecdotal observations, it is likely inconsistent over time and space or localized to a small number of individual female parasites rather than a population-wide phenomenon. Further studies are needed to assess how widespread cowbird mafia behavior is across host species and populations and what its implications and impacts may be on host fitness.RESUMEN
INTRODUCTION
Obligate brood parasites lay their eggs in the nests of other species, effectively tricking most host parents into raising their young (Davies 2000, Di Liberto 2021). Brown-headed Cowbirds (Molothrus ater; hereafter, cowbird) are a common obligate avian brood parasite in North America that often impose moderate-to-severe reproductive costs on their hosts (Rothstein 1975, Hauber 2003, Kilner 2003). In turn, some hosts of the cowbird have evolved defenses to lessen the costs associated with parasitism (Soler 2014). For example, egg ejection, which involves successfully identifying and removing parasite eggs from the nest, is performed by a handful of cowbird host species, such as the Gray Catbird (Dumetella carolinensis) and the American Robin (Turdus migratorius; Rothstein 1982, Peer and Sealy 2004, Turner and Hauber 2021).
In contrast to the few hosts that have evolved defense behaviors against cowbird parasitism, most hosts accept foreign eggs and/or nestlings (Winfree 1999). Oftentimes, accepting brood parasitic young can greatly impact host nestling growth and survival because cowbird nestlings are typically larger and hatch earlier than their host siblings and can outcompete them for parental resources (Hauber 2003, Kilner 2003). Indigo Bunting (Passerina cyanea) nestlings, for example, when raised with a cowbird nestling, have slower mass gain, likely related to the decrease in food acquisition because of the host parents disproportionately feeding the parasite (Dearborn et al. 1998). Accordingly, a review analyzing the effect of cowbird parasitism on 29 different host species found that cowbird parasitism significantly negatively affected host reproductive success (Lorenzana and Sealy 1999).
However, potential costs associated with rejecting parasitic eggs may also exist for hosts. Recognition errors, which eliminate the host’s own egg, could occur during rejection (Davies and Brooke 1988, Stokke et al. 2016), especially when the host and parasite eggs are similar in appearance (Peer et al. 2000). Further, through rejection costs, the host species could damage their own eggs while attempting to reject the foreign parasite’s egg. For example, Røskaft et al. (1993) found that Baltimore Orioles (Icterus galbula) often damaged their own eggs when rejecting cowbird eggs, decreasing hatchability and fitness.
Another potential, though rare, cost for host species capable of egg ejection is the possibility of mafia behavior by the parasite. Mafia behavior is defined as the retaliatory killing of host progeny (eggs or nestlings) by the same female cowbird after the host has rejected the parasitic egg/offspring (Zahavi 1979). The mafia hypothesis predicts that in nests where the parasite’s progeny has been removed, the host clutch or brood should experience higher predation. Experimental evidence for mafia behavior has been documented in Great Spotted Cuckoos (Clamator glandarius; Soler et al. 1995) and Brown-headed Cowbirds (Hoover and Robinson 2007). In both studies, nests where the parasite’s egg was removed had higher rates of predation compared to control nests. There is also evidence to suggest that cowbirds depredate host nests to initiate a renest that can later be parasitized, an idea known as the farming hypothesis (Arcese et al. 1996, Clotfelter and Yasukawa 1999, Hauber 2014). Mafia behavior differs from farming because it requires the same female cowbird to check up on the status of her reproductive investment. However, more studies are needed to assess how common mafia and farming behavior are across diverse populations and species of brood parasites.
In this study, we tested key assumptions and predictions of the mafia hypothesis by using long-term data on cowbird parasitism in the Dark-eyed Junco (Junco hyemalis carolinensis; hereafter, junco), conducted at Mountain Lake Biological Station (MLBS). Juncos are typically a ground nesting songbird that often experience high cowbird parasitism rates (Wolf 1987). Normally, juncos accept cowbird eggs and have been shown to successfully raise cowbird nestlings alongside their own (Wolf 1987). However, during the 22 years of data that we have mined from research conducted at MLBS, cowbird eggs were opportunistically removed by researchers. Under the mafia hypothesis’ assumptions and predictions, (1) at least some junco nestlings should fledge when a cowbird egg is left inside the nest for tolerance of cowbird parasitism to evolve (Hauber 2014), and (2) cowbird egg removal nests should have lower nest success than nests where the cowbird egg remained in the nest.
METHODS
Dark-eyed Juncos as cowbird hosts
Between 1990 and 2012, junco nests were found and monitored at the Mountain Lake Biological Station (MLBS) in Pembroke, Virginia, USA (37°22’ N, 80°32’ W; Ketterson and Atwell 2016). This research program was established by Drs. Ellen Ketterson and Val Nolan, Jr., and included annual surveys during the breeding season and experiments with junco adults and nests (Reed et al. 2006). The habitat is characterized by a mixed deciduous and coniferous forest spanning an elevational range of 1000–1330 m. The forest is divided by roads, hiking trails, power lines, and human structures associated with the biological station and nearby Mountain Lake Hotel. Juncos are territorial and nest throughout the study site, including directly on buildings when feasible (see Chandler et al. 1994 for more details).
Juncos accept cowbird eggs and lay beige eggs with brown maculation similar to those of a cowbird. Junco clutch sizes are 3–5 eggs, with a modal clutch size of 4 (Nolan et al. 2020). At MLBS, an average of 1.7 cowbird eggs were laid in parasitized junco nests and 1.2 junco eggs were removed by cowbirds (Nolan et al. 2020). Cowbird parasitism negatively impacts junco reproductive success because parasitized nests have lower reproductive success compared to non-parasitized nests (Nolan et al. 2020). However, cowbird young survival is high (85% survival) in junco nests when at least 1 junco nestling survives to fledge (Wolf 1987).
Data mining
We accessed a publicly available dataset associated with this long-term study (https://scholarworks.iu.edu/dspace/handle/2022/15260/browse) and extracted data on junco nest success, cowbird parasitism, cowbird egg removal, nest content, and reproductive outcome (i.e., number of junco eggs laid and hatched and number of nestlings fledged). Cowbird eggs were removed opportunistically across the study years (N = 116 nests), independent of directly testing the mafia hypothesis. As such, it was not a proper experimental design because the study was missing a junco egg removal treatment; instead, we used non-egg removal nests with (N = 46 nests) or without (N = 1539 nests) cowbird eggs as our statistical comparisons (sensu Hoover and Robinson 2007).
We focused on junco nests that had a first egg date on or before 1 July because of the consistent decline in cowbird parasitism later in the breeding season in this and other data sets (e.g., Wolf 1987, Hauber 2001). Nest data from 1993, 1997, and 2005 were used to calculate the parasitism rate only and were excluded from the mafia analyses because fledging success was manipulated for other projects in those years.
Last, we examined every nest log and made note of anecdotal evidence for cowbird predation on junco nests (Table 1). This evidence was characterized by puncture wounds on junco eggs and/or nestlings or direct mentions of cowbirds responsible for nest failure.
Statistics
We built a generalized mixed model (GLMER) in R version 4.1.1 (R Core Team 2021) by using nest fate (failed or fledged) as the response variable and cowbird removal status (cowbird egg remained in the nest, cowbird egg removed from the nest, or non-parasitized nest) as a fixed predictor variable (package “lme4”). Year and female identification were included as a random effect to control for repeated sampling of the same female within and between years.
To analyze the effect of annual cowbird parasitism on annual junco nest success, we used a linear regression with annual junco nest fate and annual cowbird parasitism. Last, to analyze the effect of year on cowbird parasitism, we generated a GLM with cowbird parasitism as the response variable and year as a fixed predictor variable. The package “emmeans” was used for post hoc comparisons.
RESULTS
We found a significant effect of parasitism status on the fledging success of junco nests (χ² = 7.32, df = 2, p = 0.03; Fig. 1). Fledging success was higher in nests where the cowbird egg was removed (n = 116) compared to nests where the cowbird egg remained in the nest (n = 46; z = -2.13, p = 0.03). Non-parasitized nests (n = 1539) had higher fledging success than nests where cowbird remained inside the nest (z = -2.63, p < 0.01); however, there was no significant difference between non-parasitized nests and nests where the cowbird egg was removed (z = -0.71, p = 0.48).
Cowbird parasitism rates heavily fluctuated from 1990 to 2012 (χ² = 45.31, df = 19, p < 0.001; Fig. 2). However, there was no detectable relationship between annual cowbird parasitism rate and annual junco nest success (F < 0.01, df = 18, p = 0.96). Table 1 shows anecdotal evidence of cowbird predation (mafia and/or farming behavior, n = 6) in this system.
DISCUSSION
A critical assumption of the mafia hypothesis is that at least some junco nestlings should fledge from parasitized nests, but this assumption has little support from our dataset. Throughout our entire multi-decade dataset, only 11% (5 out of 46) of nests where the cowbird remained had at least one junco nestling survive to fledge, with the median and modal reproductive success being zero in parasitized junco nests. Our result differs considerably from the findings of Wolf (1987), where 75% of juncos fledged from parasitized nests, although it is important to note that most of the cowbird eggs were not removed from nests in that study.
A central prediction of the mafia hypothesis is that nests with parasitic eggs removed should have lower success than nests with parasitic eggs that remain. Our findings do not support this prediction. At least in this junco population, fledging success was lowest in nests where the cowbird was left inside the nest. These findings are contrary to results from at least two other accepter cowbird hosts: the Song Sparrow (Melospiza melodia), where the number of sparrow eggs that produced nestlings decreased when the cowbird egg had been removed (Hauber 2009); and the Prothonotary Warbler (Protonotaria citrea), where the predictions of the mafia hypothesis were experimentally supported (Hoover and Robinson 2007). Cowbird parasitism decreases nest success in juncos (Wolf 1987) and in most other host species (Hauber 2003); therefore, in the absence of retaliatory cowbirds, the removal of parasitic eggs would not negatively impact the junco’s reproductive success, which may be relevant if juncos should ever become of conservation concern locally or range wide (Domínguez et al. 2015).
Multiple explanations exist for why mafia behavior does not seem to be occurring in this population. First, the cowbirds and juncos at this study site have only had about 50 years of interactions since the arrival of cowbirds in the 1960s, and during that time juncos have accepted foreign eggs and raised cowbird nestlings (Wolf 1987), which is a host-parasite relationship that ought not favor parasitic mafia behavior (Hoover and Robinson 2007). The research conducted at MLBS that artificially changed these juncos into “apparent” cowbird egg rejectors has only occurred since the late 1980s and is so recent that not enough time may have passed on a behavioral evolutionary timescale to favor the cowbird’s retaliatory mafia behaviors. In other words, the absence of cowbird mafia behavior might be a result of evolutionary lag because of their relatively short coevolutionary relationship with “rejecter” juncos at this site. This hypothesis is often used as a possible explanation for why so many hosts of Brown-headed Cowbirds accept foreign eggs (Winfree 1999).
Second, cowbird mafia behavior may be a localized or even individually driven behavior among cowbirds (Peer et al. 2013). Theoretically, if cowbird mafia behavior were widespread throughout North America, it should maintain hosts’ acceptance of parasitic eggs (Peer et al. 2013). However, some cowbird hosts can and do successfully reject cowbird eggs from their nests (Rothstein 1982, Peer and Sealy 2004, Turner and Hauber 2021), further suggesting that cowbird mafia behavior could be a regional, host-species specific, or otherwise localized behavior. Specifically, cowbirds may exhibit mafia behavior with host species that have a longer, shared coevolutionary history with cowbirds (Peer et al. 2013). In contrast, however, Peer and Sealy (2004) found that hosts with longer evolutionary histories with cowbirds were in fact more likely to be rejecters. Furthermore, as far as we know, cowbird mafia has only been experimentally documented in the Prothonotary Warbler (Hoover and Robinson 2007), which is an accepter host that has likely experienced cowbird parasitism for less time compared to cowbird-host systems in the western part of the North American continent (Peer et al. 2013). Last, it is possible that cowbirds do not exhibit mafia behavior and more experimental evidence is necessary to tease apart evidence for mafia versus farming strategies.
Despite a lack of statistical support for persistent retaliatory behavior, there is some anecdotal evidence that suggests cowbirds may have depredated select junco nests in our study population (see Table 1). The main predators of junco nests are rodents, which tend to remove the entire egg/nestling from the nest and often pull the nest out or compromise the nest lining (Wolf et al. 1988). Punctured eggs/nestlings that are left in the nest may be indicative of cowbird predation (Hauber 1998), and to our knowledge, there are no other avian predators documented that puncture junco eggs. Igl (2003) has also documented a case of a male cowbird pecking a host nestling to death. However, not all of the nests in this study with possible cowbird predation had a cowbird egg that was removed (Table 1), suggesting that cowbirds could be depredating host nests for non-retaliatory reasons. For instance, the farming hypothesis predicts that cowbirds depredate when encountering a non-parasitized host nest to generate more opportunities for future parasitism attempts (Hauber 2014), which has been documented in several cowbird hosts (Arcese et al. 1996, Clotfelter and Yasukawa 1999, Hoover and Robinson 2007, Swan et al. 2015). Additionally, the Ketterson and Nolan lab have a video recording of a female cowbird pecking junco nestlings in the nest (https://doi.org/10.6084/m9.figshare.19222953). The nest log confirms that there were no cowbird eggs laid in the nest, evidence supporting the farming hypothesis (sensu Hauber 2014). Other cowbird species, such as the Shiny Cowbird (M. bonariensis) and the Screaming Cowbird (M. rufoaxillaris), also peck their host eggs to reduce future nestling competition rather than farming (Cossa et al. 2017). Further studies are needed to examine the prevalence of farming versus mafia behaviors across cowbird and their host populations.
Cowbird parasitism rates varied heavily over the 22 years. During some years, juncos experienced high parasitism pressure, upward of 20% (e.g., 1993, 1997, 2011), whereas other years resulted is very little or no parasitism (e.g., 2003, 2004). Cowbird abundance on a local scale can be impacted by factors such as habitat, vegetation and food sources, predators, and host abundance (Chace et al. 2005). However, more studies are needed to investigate which factors are most prominent in influencing cowbird abundance and parasitism rates in the cowbird-junco system at Mountain Lake.
Because these data were mined from an existing database, our study lacks some experimental and control treatments, such as a junco egg removal group, which would allow us to fully analyze how cowbirds respond to egg removal. For example, an alternative explanation for our results is that disruption of the nest by researchers during egg removal influenced nest success, and we cannot statistically test for this potential confound. However, we find this interpretation to be unlikely for two reasons. First, all junco nests, regardless of parasitism/removal status, were handled and/or disturbed by researchers for various experiments; therefore, disturbance was not limited to the egg removal treatment. Second, we would predict egg removal and/or disturbance to increase the likelihood of nest failure (Rothstein 1986, Kosciuch et al. 2006), but our results contradict this prediction. Nevertheless, future experimental studies analyzing cowbird mafia should seek to include such a treatment.
CONCLUSIONS
Our findings in a cowbird-junco system do not statistically support a key tenet of the mafia hypothesis. Junco nests where the cowbird egg had been removed had higher fledging success than nests where the cowbird egg remained inside the nest. Brown-headed Cowbird mafia behavior has been experimentally documented in only one host species, the Prothonotary Warbler (Hoover and Robinson 2007). Future studies are needed to investigate the presence and effect of cowbird mafia behavior, particularly in cowbird-host systems with longer coevolutionary histories. If cowbird mafia is individually specific, tracking tagged female cowbirds shall provide an optimal way to monitor how widespread mafia and/or farming behaviors are among different cowbird populations.
RESPONSES TO THIS ARTICLE
Responses to this article are invited. If accepted for publication, your response will be hyperlinked to the article. To submit a response, follow this link. To read responses already accepted, follow this link.ACKNOWLEDGMENTS
For access to data and nest logs we thank Ellen Ketterson and her research team at Indiana University. Funding was provided by a University of Illinois Graduate College Fellowship (to AMT) and the Humboldt Foundation through the University of Bielefeld (to MEH). For discussions, we thank Ellen Ketterson, Jeff Hoover, Wendy Schelsky, Nick Antonson, Hannah Scharf, and other colleagues.
DATA AVAILABILITY
Our dataset and R code are published on Figshare.com and can be accessed here: https://doi.org/10.6084/m9.figshare.19319843.v1
LITERATURE CITED
Arcese, P., J. N. Smith, and M. I. Hatch. 1996. Nest predation by cowbirds and its consequences for passerine demography. Proceedings of the National Academy of Sciences 93(10):4608-4611. https://doi.org/10.1073/pnas.93.10.4608
Chace, J. F., C. Farmer, R. Winfree, D. R. Curson, W. E. Jensen, C. B. Goguen, and S. K. Robinson. 2005. Cowbird (Molothrus spp.) ecology: a review of factors influencing distribution and abundance of cowbirds across spatial scales. Ornithological Monographs 57:45-70. https://doi.org/10.2307/40166814
Chandler, C. R., E. D. Ketterson, V. Nolan, Jr., and C. Ziegenfus. 1994. Effects of testosterone on spatial activity in free-ranging male Dark-eyed Juncos, Junco hyemalis. Animal Behaviour 47(6):1445-1455. https://doi.org/10.1006/anbe.1994.1191
Clotfelter, E. D., and K. Yasukawa. 1999. Impact of brood parasitism by Brown-headed Cowbirds on Red-winged Blackbird reproductive success. Condor 101(1):105-114. https://doi.org/10.2307/1370451
Cossa, N. A., D. T. Tuero, J. C. Reboreda, and V. D. Fiorini. 2017. Egg pecking and puncturing behaviors in Shiny and Screaming Cowbirds: effects of eggshell strength and degree of clutch completion. Behavioral Ecology and Sociobiology 71:60. https://doi.org/10.1007/s00265-017-2289-1
Davies, N. B. 2000. Cuckoos, cowbirds, and other cheats. T. & A. D. Poyser, London, UK.
Davies, N. B., and M. d. L Brooke. 1988. Cuckoos versus Reed Warblers: adaptations and counteradaptations. Animal Behaviour 36(1):262-284. https://doi.org/10.1016/S0003-3472(88)80269-0
Dearborn, D. C., A. D. Anders, F. R. Thompson III, and J. Faaborg. 1998. Effects of cowbird parasitism on parental provisioning and nestling food acquisition and growth. Condor 100(2):326-334. https://doi.org/10.2307/1370273
Di Liberto, J. F. 2021. Interspecific brood parasitism. Pages 1-7 in J. Vonk and T. Shackelford, editors. Encyclopedia of animal cognition and behavior. Springer, Cham, Switzerland. https://doi.org/10.1007/978-3-319-47829-6_1339-1
Domínguez, M., J. C. Reboreda, and B. Mahler. 2015. Impact of Shiny Cowbird and botfly parasitism on the reproductive success of the globally endangered Yellow Cardinal Gubernatrix cristata. Bird Conservation International. 25(3):294-305. https://doi.org/10.1017/S095927091400015X
Hauber, M. E. 1998. Single-egg removal from an artificial nest by the Gray Catbird. Wilson Bulletin 110(3):426-429.
Hauber, M. E. 2001. Site selection and repeatability in Brown-headed Cowbird (Molothrus ater) parasitism of Eastern Phoebe (Sayornis phoebe) nests. Canadian Journal of Zoology 79(8):1518-1523. https://doi.org/10.1139/z01-091
Hauber, M. E. 2003. Hatching asynchrony, nestling competition, and the cost of interspecific brood parasitism. Behavioral Ecology 14(2):227-235. https://doi.org/10.1093/beheco/14.2.227
Hauber, M. E. 2009. Does the removal of an avian brood parasite eggs increase host productivity? A case study with Brown-headed Cowbirds Molothrus ater and Song Sparrows Melospiza melodia near Ithaca, New York, USA. Conservation Evidence 6:83-88.
Hauber, M. E. 2014. Mafia or farmer? Coevolutionary consequences of retaliation and farming as predatory strategies upon host nests by avian brood parasites. Coevolution 2(1):18-25. https://doi.org/10.1080/23256214.2014.913974
Hoover, J. P., and S. K. Robinson. 2007. Retaliatory mafia behavior by a parasitic cowbird favors host acceptance of parasitic eggs. Proceedings of the National Academy of Sciences 104(11):4479-4483. https://doi.org/10.1073/pnas.0609710104
Igl, L. D. 2003. Male Brown-headed Cowbird attacks and kills a nestling. Wilson Bulletin 115(2):210-212. https://doi.org/10.1676/02-109
Ketterson, E. D., and J. W. Atwell, editors. 2016. Snowbird: integrative biology and evolutionary diversity in the junco. University of Chicago Press, Chicago, Illinois, USA.
Kilner, R. M. 2003. How selfish is a cowbird nestling? Animal Behaviour 66(3):569-576. https://doi.org/10.1006/anbe.2003.2204
Kosciuch, K. L., T. H. Parker, and B. K. Sandercock. 2006. Nest desertion by a cowbird host: an antiparasite behavior or a response to egg loss? Behavioral Ecology 17(6):917-924. https://doi.org/10.1093/beheco/arl025
Lorenzana, J. C., and S. G. Sealy. 1999. A meta-analysis of the impact of parasitism by the Brown-headed Cowbird on its hosts. Studies in Avian Biology 18:241-253.
Nolan, V., Jr., E. D. Ketterson, D. A. Cristol, C. M. Rogers, E. D. Clotfelter, R. C. Titus, S. J. Schoech, and E. Snajdr. 2020. Dark-eyed Junco (Junco hyemalis). Version 1.0. In A. F. Poole and F. B. Gill, editors. Birds of the world. Cornell Lab of Ornithology, Ithaca, New York, USA. https://doi.org/10.2173/bow.daejun.01
Peer, B. D., J. W. Rivers, and R. I. Rothstein. 2013. Cowbirds, conservation, and coevolution: potential misconceptions and directions for future research. Chinese Birds 4(1):15-30.
Peer, B. D., S. K. Robinson, and J. R. Herkert. 2000. Egg rejection by cowbird hosts in grasslands. Auk 117(4):892-901. https://doi.org/10.1093/auk/117.4.892
Peer, B. D., and S. G. Sealy. 2004. Correlates of egg rejection in hosts of the Brown-headed Cowbird. Condor 106(3):580-599. https://doi.org/10.1093/condor/106.3.580
R Core Team. 2021. R: a language and environment for statistical computing. Version 4.1.1. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org/
Reed, W. L., M. E. Clark, P. G. Parker, S. A. Raouf, N. Arguedas, D. S. Monk, E. Snajdr, V. Nolan, Jr., and E. D. Ketterson. 2006. Physiological effects on demography: a long-term experimental study of testosterone’s effects on fitness. American Naturalist 167(5):667-683. https://doi.org/10.1086/503054
Røskaft, E., S. Rowher, and C. D. Spaw. 1993. Cost of puncture ejection compared with costs of rearing cowbird chicks for Northern Orioles. Ornis Scandinavica 24:28-32. https://doi.org/10.2307/3676406
Rothstein, S. I. 1975. An experimental and teleonomic investigation of avian brood parasitism. Condor 77(3):250-271. https://doi.org/10.2307/1366221
Rothstein, S. I. 1982. Mechanisms of avian egg recognition: which egg parameters elicit responses by rejecter species? Behavioral Ecology and Sociobiology 11:229-239. https://doi.org/10.1007/BF00299299
Rothstein, S. I. 1986. A test of optimality: egg recognition in the Eastern Phoebe. Animal Behaviour 34(4):1109-1119. https://doi.org/10.1016/S0003-3472(86)80170-1
Soler, M. 2014. Long-term coevolution between avian brood parasites and their hosts. Biological Reviews 89(3):688-704. https://doi.org/10.1111/brv.12075
Soler, M., J. J. Soler, J. G. Martinez, and A. P. Møller. 1995. Magpie host manipulation by Great Spotted Cuckoos: evidence for an avian mafia? Evolution 49(4):770-775. https://doi.org/10.1111/j.1558-5646.1995.tb02312.x
Stokke, B. G., E. Røskaft, A. Moksnes, A. P. Møller, A. Antonov, F. Fossøy, W. Liang, G. Lòpez-Iborra, C. Moskát, J. A. Shykoff, M. Soler, J. R. Vikan, C. Yang, and F. Takasu. 2016. Disappearance of eggs from nonparasitized nests of brood parasite hosts: the evolutionary equilibrium hypothesis revisited. Biological Journal of the Linnean Society 118(2):215-225. https://doi.org/10.1111/bij.12733
Swan, D. C., L. Y. Zanette, and M. Clinchy. 2015. Brood parasites manipulate their hosts: experimental evidence for the farming hypothesis. Animal Behaviour 105:29-35. https://doi.org/10.1016/j.anbehav.2015.03.012
Turner, A. M., and M. E. Hauber. 2021. The American Robin (Turdus migratorius): a focal species for anti-parasitic egg rejection studies among hosts of the Brown-headed Cowbird (Molothrus ater). Ethology 127(6):490-503. https://doi.org/10.1111/eth.13158
Winfree, R. 1999. Cuckoos, cowbirds and the persistence of brood parasitism. Trends in Ecology & Evolution 14(9):338-343. https://doi.org/10.1016/S0169-5347(99)01643-2
Wolf, L. 1987. Host-parasite interactions of Brown-headed Cowbirds and Dark-eyed Juncos in Virginia. Wilson Bulletin 99(3):338-350.
Wolf, L., E. D. Ketterson, and V. Nolan, Jr. 1988. Parental influence on growth and survival of Dark-eyed Junco young: do parental males benefit? Animal Behaviour 36(6):1601-1618. https://doi.org/10.1016/S0003-3472(88)80102-7
Zahavi, A. 1979. Parasitism and nest predation in parasitic cuckoos. American Naturalist 113(1):157-159. https://doi.org/10.1086/283374
Fig. 1
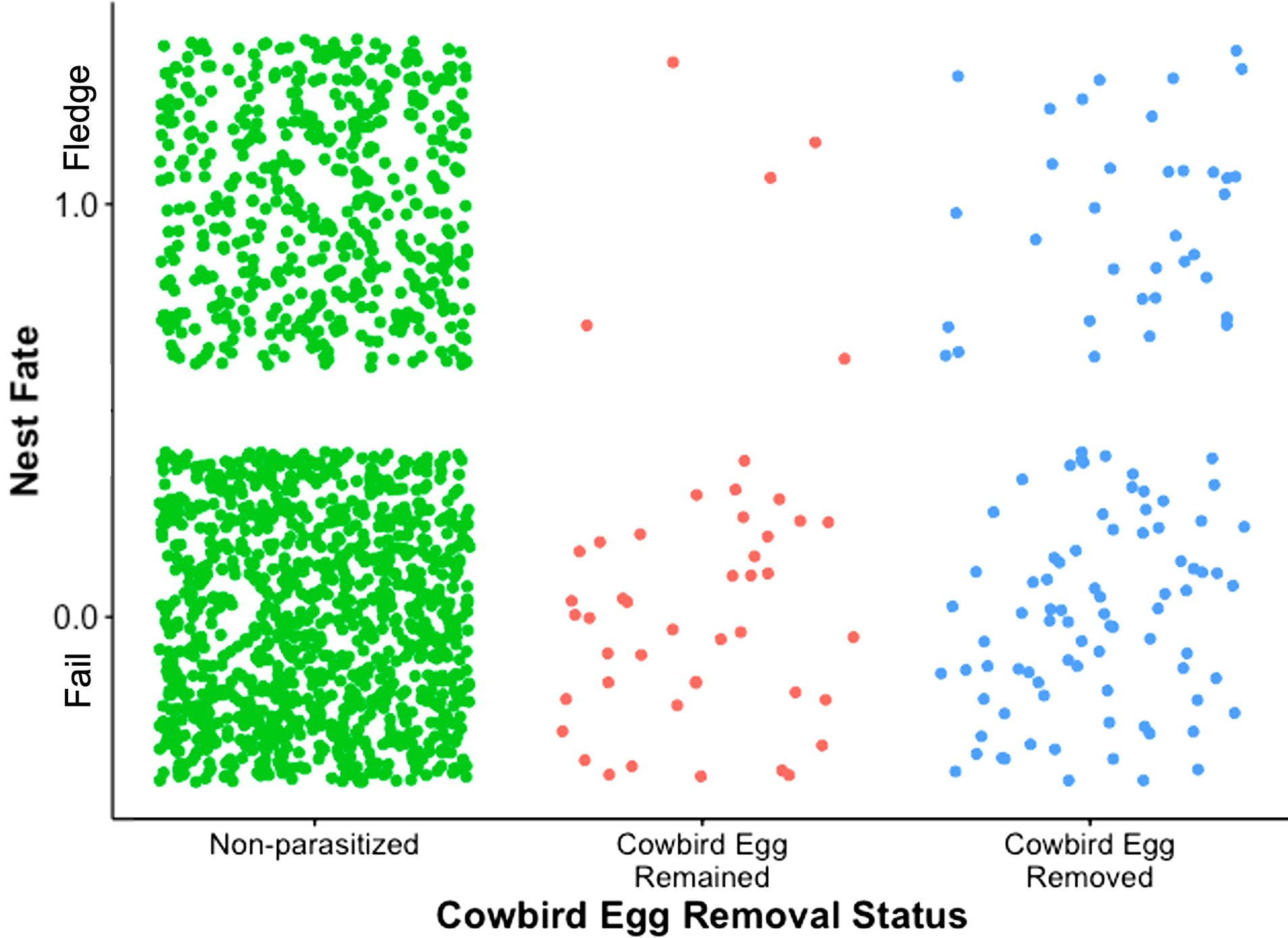
Fig. 1. Number of nests that failed or fledged in non-parasitized (n = 1539) nests, nests where the Brown-headed Cowbird (Molothrus ater) egg remained inside the nest (n = 46), and nests where the cowbird egg was removed (n = 116).

Fig. 2
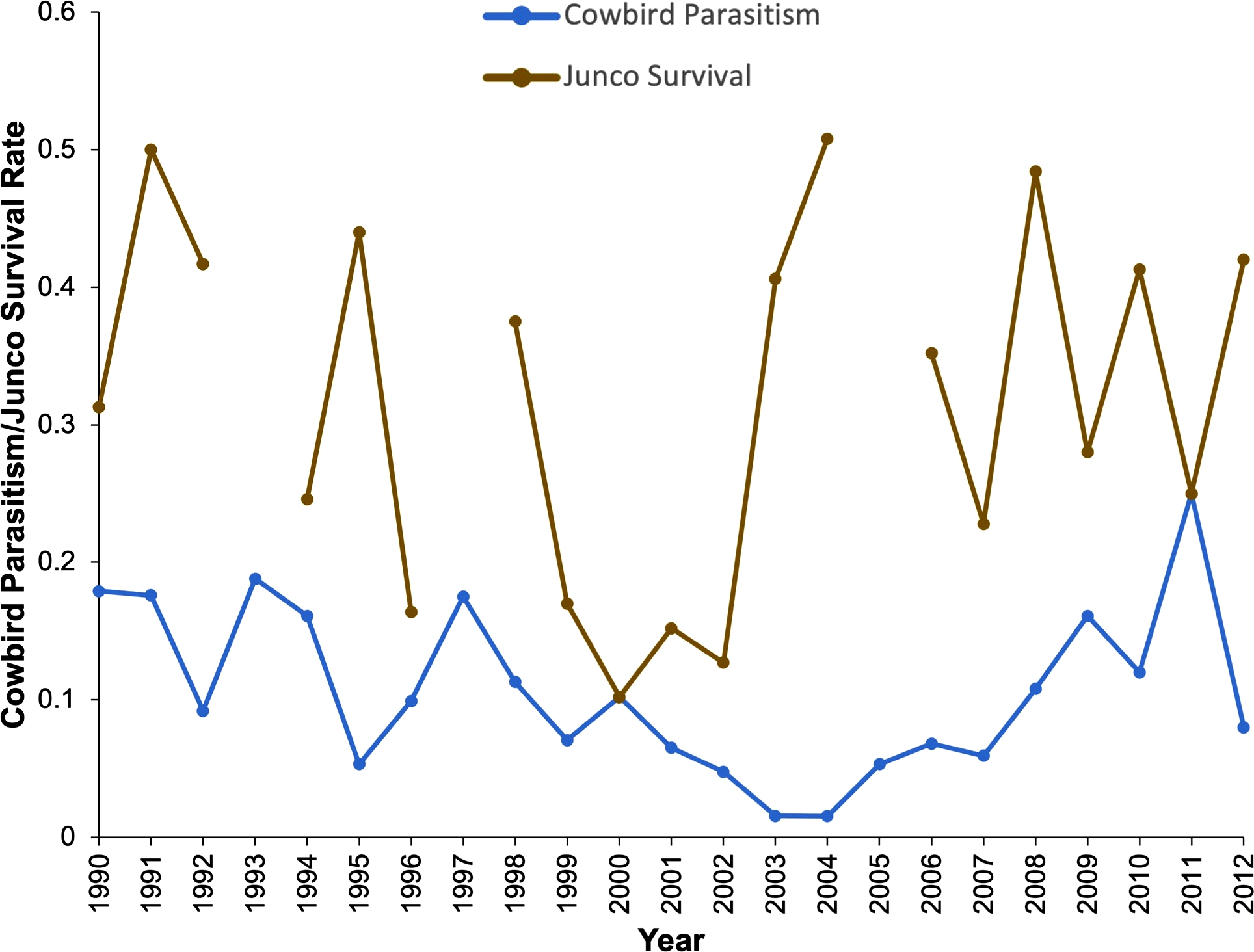
Fig. 2. Brown-headed Cowbird (Molothrus ater) parasitism rates and the proportion of Dark-eyed Junco (Junco hyemalis carolinensis) nests that fledged at least one nestling at Mountain Lake Biological Station between 1990 and 2012. Nest data from 1993, 1997, and 2005 were only used to calculate parasitism rate; these years were excluded from calculations of junco survival as they were manipulated for other projects.

Table 1
Table 1. Anecdotal evidence (n = 6) of Brown-headed Cowbird (Molothrus ater) impacts on Dark-eyed Junco (Junco hyemalis carolinensis) nests from our data logs.
| Date Found | Parasitized Nest? | Cowbird Removed? | Comment on Nest Log |
| 5/14/90 | Yes | No | “Transplanted a junco egg from a different nest. The egg laid from the female associated with this nest had two puncture wounds probably from the cowbird.” |
| 5/22/99 | Yes | Yes | “Nest empty, one egg is broken in half lying about six inches from nest. Another egg with a small puncture hole about twelve inches away from the nest. I suspect a cowbird is to blame.” |
| 5/23/03 | No | - | “Dead nestlings, little puncture wounds.” |
| 6/17/08 | Yes | Yes | “Cowbird killed nestlings in her first nest?” |
| 4/30/10 | No | - | “Four junco nestlings dead in nest, injuries and blood apparent on all nestlings, suspect cowbird mafia, no juncos seen or heard.” |
| 5/4/10 | Yes | No | “All eggs cold. One junco egg obviously pecked by something (likely cowbird), no juncos seen/heard.” |









