The following is the established format for referencing this article:
Takagi, M. 2022. Replacement of rectrices by wintering White Wagtails: an experiment to examine effects of sex, age, and body condition. Journal of Field Ornithology 93(3):1.ABSTRACT
Adventitious replacement is regrowth of accidentally lost feathers outside usual molt periods. To examine the intrinsic factors affecting adventitious replacement, I conducted feather-plucking experiments on White Wagtail (Motacilla alba lugens) during the coldest time of the year. I examined sex, age class, and body condition of wagtails as potential factors affecting the ratio of the lengths of regrown rectrices (T4) to their original lengths. I captured and ringed 288 wagtails in January, plucked and measured the length of the right T4, and measured tarsus length and mass. I recaptured 121 of the wagtails approximately 6 weeks later, and plucked and measured their regrown T4s. In a modeling analysis of the ratio of original to fully grown T4, males regrew their T4s to lengths closer to those of their original T4s than did females. Males are presumed to need longer tail feathers than females. Differences in regrowth were not explained by age class. Both adults and yearlings must acquire sufficiently long rectrices for them to achieve full their functionality significance. Individuals in better condition tended to regrow their T4s to lengths closer to those of their original T4s than did individuals in poorer condition. Adventitious replacement may be influenced by body condition in those individuals that finished regrowing their rectrices early. In an analysis of the ratio of original to regrowing T4, the ratio for adults was greater than that for yearlings. Yearlings may be slower than adults to fill the gap left by losing a T4, or juveniles may have begun regrowth of their plucked T4s later than adults. Sex and body condition did not significantly contribute to the model. Because age-class difference in the regrowth speed of T4s might not be influenced by body condition, adventitious replacement is worthy of careful examination in the future in the context of life history strategies.RESUMEN
INTRODUCTION
Feathers provide insulation, which enables birds to maintain a high body temperature; they are also essential for physical protection and flight, and serve visual communication and camouflage purposes (Ginn and Melville 1983). Birds maintain good feather condition by preening and by bathing in water or dust (Mench and Blatchford 2013). Because feathers cannot self-repair, they are replaced regularly in a comprehensive way, both seasonally and with age, in the process known as “molt” (Gill 2007, Jenni and Winkler 2020). Molt serves to maintain the plumage in good condition and adjusts the plumage characteristics to the bird’s changing needs during its lifetime (Jenni and Winkler 1994). Molt is one of the three most energy-demanding processes in the life cycle of birds (Newton 2009). Birds regularly face the energetically demanding task of molting all their flight and body feathers (Jenni and Winkler 2020). Complete molt typically follows the breeding season in the warmest months of the year and precedes migration (Gill 2007). Molt also competes with migration in terms of time and energy, and the performance of the bird (Jenni and Winkler 2020).
Replacement of accidentally lost feathers outside the molt periods is known as adventitious replacement (Jenni and Winkler 2020). Experiments involving the plucking of rectrices have been conducted to explore whether original and replaced feathers show structural and mechanical differences (Grubb 1989, Reudink et al. 2008, De la Hera et al. 2010, 2015). In Downy Woodpeckers (Picoides pubescens), replaced rectrices are shorter than original feathers (Grubb 1989). In Eurasian Blackcaps (Sylvia atricapilla), replaced rectrices have wider rachides and greater density of barbs but are lighter, shorter, and less stiff than original feathers (De la Hera et al. 2010). Experiments on wintering American Redstart (Setophaga ruticilla) that compared stable hydrogen isotopes (δD) and color between original and regrown rectrices showed that regrown feathers had highly positive δD signatures and low red chroma (Reudink et al. 2008). Simultaneous loss of primary and tail feathers in Eurasian Blackcaps and European Robins (Erithacus rubecula) suggests that rapid repair of the most essential feather tracts is also evolutionarily advantageous during the adventitious replacement of plumage (De la Hera et al. 2015). Adventitious replacement would have been favored by natural selection because the energetic costs of regrowth are lower than the long-term costs of maintaining an incomplete plumage until the following molt (Grubb 2006, Møller et al. 2006). The effects of age class and body condition in rectrix-plucking experiments have been examined only in terms of feather color in American Redstart males, and resulted in both yearlings and adults regrowing rectrices with reduced carotenoid content (Tonra et al. 2014). In order to elucidate the ecological and evolutionary significance of adventitious replacement, it is essential to adequately examine which individual traits, such as sex, age, and body condition, are associated with adventitious replacement, although as yet, this has not been sufficiently achieved.
Experimental studies on adventitious replacement have been conducted mainly by plucking a single rectrix, for two main reasons. First, there is evidence that fault bars, which have long been known as common feather growth abnormalities in birds (Riddle 1908), occur more frequently on avian rectrices than on remiges (American Kestrel Falco sparverius [Bortolotti et al. 2002], Barn Swallow Hirundo rustica [Serrano and Jovani 2005]). This implies that rectrices play a less important functional role than remiges. I thus hypothesized that only individuals in better body condition or requiring more tail function would regrow a lost rectrix. Second, rectrix-plucking experiments mimic natural situations because tail feathers are highly susceptible to accidental loss (Møller et al. 2006, Reudink et al. 2008). Tail movements occur and are obvious in many bird species (Randler 2016). Some hypotheses have been proposed to explain the functional significance of morphometric tail traits and/or movements in intra- and inter-specific signaling contexts. Thus, tail movements might signal to a predator that the signaler has detected it or is particularly alert or otherwise difficult to catch; they may warn conspecifics of predators; or they might advertise quality as a mate, signal social status, or aid in flock cohesion (reviewed in Randler 2016).
I conducted plucking experiments on the rectrices of wintering White Wagtail (Motacilla alba lugens), in light of the ecological importance of rectrices for the species. The normal molt periods of this taxon are as follows: complete definitive prebasic molt during July–August; partial preformative molt (almost all feathers, excluding primary, secondary, and rectrices) by late summer; and first and definitive prealternate molt of all head and body feathers starting from February to April (Yamashina 1934, Bird Migration Research Center 1983). It may be difficult for White Wagtails to maintain their body condition during the coldest period of the year because the body masses of wintering wagtails are toward the light end of their range at that time of year (Hirano 2009, this study).
I examined sex, age class, and body condition of wagtails as potential factors that affect the ratio of the lengths of regrown and original rectrices. Furthermore, wagtails are clearly representative of birds that frequently wag their tails (Alström and Mild 2003), and it has been suggested that such tail wagging may be an honest signal of vigilance during both feeding and preening when the wagtail is aware of an ambushing predator (Randler 2006). Male wagtails approach females while lowering their body and spreading and slightly drooping their wings and tail, and court females by widely spreading their tail feathers(Alström and Mild 2003, Kawabe 2006). Thus, it is inferred that tail traits are important for both natural and sexual selection in wagtails. After showing how sex, age, and body condition are related to rectrix regrowth, I discuss the behavioral ecological aspects of adventitious replacement in wagtails.
METHODS
Materials and study site
White Wagtails breed throughout the Japanese archipelago (Ornithological Society of Japan 2012). Northern breeding populations, especially those of Hokkaido, winter in southwest central Honshu from October to April (Yamashina 1934, Yamashina Institute for Ornithology 1996) and join communal roosts (Broom et al. 1976, Watanabe and Maruyama 1977). A communal roost was located and studied at Yamato River Bridge on Route 26, on the Yamato River that divides Sakai and Osaka cities in Honshu, Japan (34°35'53" N, 135°28'51" E). The average minimum temperature at the roosting site was 1.1–1.3ºC during January and February (the coldest period of the winter) from 1981 to 2010 (Japan Meteorological Agency 2010).
Field procedure
I captured wagtails at the communal roost during two periods: 13, 15, and 17 January 2002; and 26 February and 1 and 5 March 2002. To capture several wagtails simultaneously, I opened two mist-nets (one LTX 20 m long 36-mm mesh net, and one ATX 12 m long 36-mm mesh net) below the bridge at 0500, approximately 1 or 2 hours before dawn, when wagtails leave the communal roost for the day.
Data collection
All the captured wagtails were safely transported by car to Osaka City University, 2.7 km away from the communal roost where they had been captured, and all the following procedures were carried out in the laboratory. In the first period, wagtails were ringed individually with numbered metal leg rings, were aged as either yearling (first winter) or adult (older), and were sexed using plumage characteristics as described by Bird Migration Research Center (1983). I measured tarsus length to the nearest 0.01 mm using digital calipers (Mitutoyo ABS digimatic calipers CD-15CP), according to Svensson (1992). Tarsus length is most suitable as the covariate because it is one of the best predictors of body size (Senar and Pascual 1997). Body mass was measured to the nearest 0.1 g using a digital balance (Tanita pocketable scale KP-104). I plucked right rectrix T4 and measured its length (from the inferior umbilicus to the tip of the rachis) to the nearest 0.01 mm using the digital caliper. This was recorded as the original T4 length (OLT4). I did not detect any difference in feather wear between age and sex classes among the originally plucked feather samples.
During the second period, I recaptured banded wagtails to reweigh and inspect them. The rectrices were examined to confirm if a new T4 was regrowing from the position of the plucked T4. All T4s were plucked again regardless of whether they were regrowing or had already been fully regrown during adventitious replacement. Those exhibiting a T4 root sheath were classified as regrowing, while those with T4 present but without sheaths were classified as fully regrown and were remeasured. I plucked new, replacement right T4s and remeasured them (RLT4) as before. Individuals that had not regrown a new T4 were included in the following analyses, using 0 cm as the length of the T4 length. All wagtails were released on the riverbed, 2.57 km away in a straight line from the roost, as soon as possible after the treatment was completed.
The experiment was approved by the Japanese Ministry of Environment under the Wildlife Protection, Management and Hunting Act, with considerations including animal welfare (permission number was #7-17 issued on 20 December 2001).
Statistical analyses
The normality of the distribution of OLT4 was tested using the Shapiro-Wilk normality test in the R package. I used a two-way analysis of variance (ANOVA) to assess whether sex and age class (both as two-level factors) influence OLT4, using the R package psych (Revelle 2021). To compare OLT4 and fully grown RLT4, I made comparisons in each combination of sex and age class (i.e., yearling female, adult female, yearling male, and adult male), using the Wilcoxon signed rank test in the R package exact Rank Tests (Hothorn and Hornik 2021). In the comparison, no Bonferroni correction was applied due to the sharp drop in statistical power and the possibility of Type II error (Nakagawa 2004).
For both fully grown and growing T4 feathers, I assessed separately whether the ratio of RLT4 to OLT4 was influenced by each individual trait. I constructed a generalized linear model with a Gaussian distribution and identity link function. The shortest interval between first capture and recapture was 40 days and the longest was 51 days, a difference of 11 days. In the analyses that follow, the effect of the difference in the length of time between capture and recapture was considered as the capturing days interval (CDI). I adopted a body condition index (BCI), wherein body mass (g) *100 was divided by tarsus length3 (cm3). In a completely different context from this study, Takagi (2022) conducted statistical tests that compared sex and age class differences in BCI based on the same data set. Thus, to avoid duplication of analyses in both papers, only descriptive statistics are presented here, and no comparative statistical analysis is described. When comparative information was necessary for this paper, I referred to Takagi (2022). I also adopted a change index in body condition index (BCCI), which was BCI in the first period minus BCI in the second period, for each individual.
I designated RLT4 as the response variable with two categorical variables, sex and age class (both as two-level factors), and CDI, BCI, and BCCI (as three continuous values) as the five predictor variables, along with the offset term OLT4 for each wagtail. The “offset” method is a suitable means of creating a model without distorting the distribution by division when the ratio is the response variable (Kubo 2012). The model was simplified by eliminating independent variables with P > 0.05. Likelihood ratio tests were used to select the parsimonious final model (Burnham and Anderson 2002). Because I constructed the generalized linear model using the offset term, the resulting figure was based on RLT4 divided by OLT4.
All data are expressed as mean ± SD (n) unless stated otherwise. All statistical analyses were conducted in R software version 3.4.4 (R Core Team 2018).
RESULTS
Out of four combinations of sex and age classes (Fig. 1), three followed a normal distribution (Shapiro-Wilk normality test: adult male, W = 0.991, P = 0.821; yearling male, W = 0.976, P = 0.288; adult female, W = 0.990, P = 0.843), whereas the frequency distribution of yearling females deviated slightly from the normal distribution (W = 0.970, P = 0.0452). OLT4s (mm) were 102.50 ± 2.58 (n = 88) for adult males, 101.66 ± 2.60 (n = 46) for yearling males, 97.54 ± 2.82 (n = 70) for adult females, and 98.08 ± 3.43 (n = 84) for yearling females. The OLT4 of males was significantly longer than that of females (two-way ANOVA, F1, 284 = 161.67, P < 0.0001), and there was no significant difference between the two age classes (F1, 284 = 0.039, P = 0.084). The significance level of the interaction between sex and age class was marginal due to including a few yearling females with longer OLT4 (F1, 284 = 3.836, P = 0.051) (Fig. 1). BCIs in the first period were 0.183 ± 0.015 (n = 88) for adult males, 0.177 ± 0.016 (n = 46) for yearling males, 0.180 ± 0.014 (n = 70) for adult females, and 0.184 ± 0.018 (n = 84) for yearling females.
Of 288 wagtails that were captured in the first period, 121 were recaptured in the second period. All the adult males had unfurled the mature part of the replaced T4 by the second period (Table 1). One adult female and one yearling male did not regrow a new T4, and 61% of yearling males and 56% of yearling females had fully regrown their T4s (Table 1). Although 13% of yearling females did not grow a new T4, 56% had fully regrown their T4s (Table 1). A higher proportion of yearlings (58%) than adults (45%) had fully regrown their T4s, but a higher proportion of yearlings (11%) also showed no new growth compared with adults (2%). There was a bimodal response to plucking among the yearlings, and there was a significant difference in the distributions of feather states between adults and juveniles (Fisher’s exact test, P = 0.014).
There was no significant difference in BCI among the three groups with fully grown and growing T4s and those lacking a T4 after plucking (one-way ANOVA: in the first period, F1, 119 = 0.039, P = 0.845; in the second period, F1, 119 = 0.98, P = 0.32).
Some individuals with regrowing T4s had reached T4 lengths equivalent to those of individuals that had already finished regrowing their T4s (Fig. 2). While many individuals had fully grown T4s, there was a small number of individuals with various lengths of growing T4s (Fig. 2).
RLT4s (mm) that had already finished regrowing were 99.27 ± 3.59 (n = 18) for adult males, 98.81 ± 3.59 (n = 11) for yearling males, 93.69 ± 2.98 (n = 11) for adult females, and 95.39 ± 2.98 (n = 22) for yearling females. In each of the four sex–age classes, fully grown RLT4 was significantly shorter than OLT4 (Wilcoxon signed rank test: adult male, V = 163, P < 0.001; yearling male, V = 65, P = 0.0019; adult female, V = 66, P < 0.001; yearling female, V = 65; P = 0.002).
BCIs in the second period were 0.182 ± 0.015 (n = 41) for adult males, 0.173 ± 0.012 (n = 18) for yearling males, 0.174 ± 0.016 (n = 23) for adult females, and 0.176 ± 0.013 (n = 39) for yearling females. BCCIs were 0.000519 ± 0.00959 (n = 41) for adult males, 0.00289 ± 0.00988 (n = 18) for yearling males, 0.000796 ± 0.00931 (n = 23) for adult females, and 0.00569 ± 0.00869 (n = 39) for yearling females. There was a significant difference in BCCI only between age classes (two-way ANOVA; sex: F1, 117 = 0.637, P = 0.43; age: F1, 117 = 4.33, P = 0.040; interaction: F1, 117 = 0.495, P = 0.48). Thus, the body condition of yearlings decreased significantly more between the first and second captures compared with that of adults.
The most parsimonious models in the generalized linear model analysis of the ratio of fully grown RLT4 with BCI in the first period or BCCI included only sex (log likelihood in sex: c2 = 22.63, df = 1, P < 0.0001) (Table 2), and the model with BCI in the second period included both sex and BCI (likelihood ratio test in sex: c2 = 4.259, df = 1, P = 0.040; in BCI: c2 = 22.77, df = 1, P < 0.0001) (Table 2, Fig. 3AB). A difference in the ratios of RLT4 to OLT4 was detected between males and females (Fig. 3A), and the ratio increased with BCI (Fig. 3B).
All of the most parsimonious models included only age class in the analysis of the ratio of growing RLT4 with BCIs in the first period (age class [male], estimate = 28.45, SE = 8.12, t = 3.51, P = 0001) or second period (estimate = 33.35, SE = 7.69, t = 4.34, P < 0.0001) or BCCI (estimate = 28.07, SE = 8.22, t = 3.41, P = 0.0012; likelihood ratio test: c2 = 16.841, df = 1, P < 0.0001) (Fig. 3C). The ratio of RLT4 to OLT4 in adults was clearly greater than in yearlings. A bimodal pattern among yearlings is also shown in Fig. 3C, with seven of them exhibiting ratios near 1.0 (similar to many adults), very few exhibiting ratios between 0.5 and 0.9 (unlike adults), and another large proportion exhibiting ratios between 0.2 and 0.4.
DISCUSSION
Altogether, the categorical analysis (Table 1), the frequency distribution of RLT4 (Fig. 2), and the comparison of the ratio of growing RLT4 to OLT4 in relation to age class (Fig. 3C) suggest that there are two groups of yearlings: one that is a quick-replacer and one that is a slow- or non-replacer. The difference could be derived from the timing of their hatching because White Wagtails (subspecies alba) have been reported to show faster molt in late broods than in early broods (Yakovleva et al. 1987). This could be tied to carry-over effects from the fall migration and/or fall molt of young birds. Carry-over effects occur when processes in one season influence the success of an individual in the following season (Inger et al. 2010, Harrison et al. 2011). Thus, if there are carry-over effects of molt timing from the summer to the winter, this could create a bimodal distribution of adventitious replacement in the winter. This also raises the possibility of carry-over effects among young birds, with one group being much more prepared for spring migration/breeding than the other group.
In this plucking experiment, males had a significantly greater proportion of individuals with fully grown T4 feathers than did females (Table 1). In the analysis of wagtails with fully grown T4 feathers, males regrew T4s to lengths closer to those of the originals than did females (Table 2, Fig. 2). Males are presumed to need longer tail feathers than females because males of the Japanese population court females by fanning their rectrices wide open (Kawabe 2006). Unequal-length rectrices may convey a poorer impression of the quality of males to females (reviewed in Romano et al. 2017).
Even though preformative molt does not change rectrices, there was no difference in OLT4 between age classes for both sexes. Remiges and rectrices of juveniles are generally shorter than those of adults, including M. alba alba (Demongin 2016). Because wing length of M. alba lugens was significantly longer in adults than in juveniles, in a study conducted on the same data set as this research (Takagi, in preparation), the absence of the difference in OLT4 between juveniles and adults in this study deviates from the general tendency.
The difference in length (as an estimate of investment) between OLT4 and RLT4 was not explained by age class in the model. This result seems in agreement with Randler’s (2006) suggestion that adult and juvenile wagtails do not differ in their frequency of tail wagging. Both adults and juveniles must acquire sufficiently long rectrices for them to achieve their functional significance if, as Randler (2006) suggests, tail wagging is an honest signal of vigilance during both feeding and preening. It is inferred that juveniles benefit from having T4s as long as those of adults for survival.
Body condition in the first period, when T4 was first plucked, was not related to the ratio of RLT4 to OLT4, but individuals in better condition in the second period tended to regrow their T4s to lengths closer to those of their original T4s (Table 2, Fig. 3B). It appears that adventitious replacement is influenced by body condition in individuals that finish regrowing their T4s early.
In the analysis of wagtails with regrowing T4 feathers, partial regrowth was achieved by the second period, indicating that there are differences in regrowth speed or in the timing of regrowth after losing a T4. Yearlings were slower to fill the gap left by losing a T4, and/or started later than adults (Fig. 3C). Since yearlings had significantly lower body condition compared with adults, it can be inferred that the physiological cost of T4 regrowth in yearlings is greater than in adults. No contribution of sex or BCI to the models was detected in this analysis. It is reasonable to infer that preparation for migration from the wintering grounds to the breeding grounds may have affected the results because among many avian species, juveniles typically arrive at their breeding sites later than adults (Stewart et al. 2002, Payevsky 2016). Because the number of White Wagtails using communal roosts begins to decline in mid to late March (Kumagai et al. 1983), the second period of this study was just before the start of the northward migration. Being in good body condition by the end of the wintering period and reaching the breeding grounds early are critical requirements for birds to acquire territories and achieve high breeding performance (Crick et al. 1993, Verhulst and Nilsson 2008, Harriman et al. 2017).
The fact that regrowth speed of T4 was not dependent on body condition, even though it was dependent on age class, is an interesting result in wagtails that did not finish regrowing their T4s. Although all adult males (n = 41) were found to have begun regrowing their T4s, among yearling females (n = 5), which did not differ significantly in BCI from adult males (Takagi 2022), none had regrown their T4s (Table 1, Fig. 2D). BCIs of those wagtails that lacked a new T4 in the position of the plucked T4 (n = 7) did not differ from wagtails with fully grown and growing T4s (n = 114). That is, T4-plucking experiments revealed that adventitious replacement is not simply dependent on body condition in each individual. In terms of life history theory, the slow regrowth rate of T4s, or the slow start of regrowth should be due to evolutionary restraints rather than mere physiological constraints (Stearns 1992). It is important to note that adventitious replacement was influenced by age-dependent traits that are embedded in the life history of each individual, and which also involve the carry-over effect.
CONCLUSION
Individual differences in the duration of complete adventitious replacement were found to be substantial. In individuals that completed adventitious replacement early, males regrew their lost T4s to nearer original length than did females, independent of body condition. In individuals that underwent slow adventitious replacement, yearlings were slower to regrow their T4s than were adults, independent of body condition. These results can be understood in terms of sexual selection and natural selection, respectively. Since age class difference in regrowth speed of T4s was not related to body condition, adventitious replacement is worthy of careful examination in the future in the context of life history strategies.
RESPONSES TO THIS ARTICLE
Responses to this article are invited. If accepted for publication, your response will be hyperlinked to the article. To submit a response, follow this link. To read responses already accepted, follow this link.AUTHOR CONTRIBUTIONS
The author designed the study, conducted the fieldwork, analyzed the results, and wrote the paper.
ACKNOWLEDGMENTS
I deeply appreciate the assistance of Shiro Murahama and Yoshihiro Fukuda for their help with banding wagtails, and the members of the Laboratory of Animal Sociology, Osaka City University, Nobuhiko Kotaka, Tomohiro Takeyama, Satoshi Awata, Yu Chin, Kana Akatani, Sayako Hikosaka, Akan Yoshikawa, and Miwa Nishimori for their kind support of my fieldwork and collection of individual data. The experiment was approved by the Japanese Ministry of Environment under the Wildlife Protection, Management and Hunting Act, with considerations including animal welfare, and the experimenters were trained by the Yamashina Institute for Ornithology and were certified as possessing ample knowledge and skill to safely handle birds and conduct experiments in the field. I would also like to thank Mark Brazil, Scientific Editing Services, for assistance in the preparation of the final draft of the manuscript.
DATA AVAILABILITY
I will make the data table available upon request.
LITERATURE CITED
Alström, P., and K. Mild. 2003. Pipits and wagtails. Princeton University Press, Princeton, USA.
Bird Migration Research Center. 1983. Bird banding manual. Identification guide to Japanese birds. No.7. Motacilla alba. Yamashina Institute for Ornithology, Shibuya, Japan.
Bortolotti, G. R., R. D. Dawson, and G. L. Murza. 2002. Stress during feather development predicts fitness potential. Journal of Animal Ecology 71:333-342. https://doi.org/10.1046/j.1365-2656.2002.00602.x
Broom, D. M., W. J. A. Dick, C. E. Johnson, D. I. Sales, and A. Zahavi 1976. Pied Wagtail roosting and reeding behaviour. Bird Study 23:267-279. https://doi.org/10.1080/00063657609476513
Burnham, K. P., and D. R. Anderson. 2002. Model selection and multimodel inference. Springer, New York, USA. https://doi.org/10.1007/b97636
Crick, H. Q. P., D. W. Gibbons, and R. D. Magrath. 1993. Seasonal changes in clutch size in British birds. Journal of Animal Ecology 62:263-273. https://doi.org/10.2307/5357
De la Hera, I., J. Pérez-Tris, and J. L. Tellería. 2010. Structural and mechanical differences between original and replaced feathers in blackcaps Sylvia atricapilla. Ardeola 57:431-436.
De la Hera, I., J. Pérez-Tris, and J. L. Tellería. 2015. Adventitious feather replacement favours a more rapid regeneration of primaries over rectrices in two passerine bird species. Ibis 157:883-887. https://doi.org/10.1111/ibi.12302
Demongin, L. 2016. Identification guide to birds in the hand. Beauregard-Vendon, France.
Gill, F. B. 2007. Ornithology. Third edition. WH Freeman Company, New York, USA.
Ginn, H. B., and D. S. Melville. 1983. Moult in birds. British Trust for Ornithology, Tring, UK.
Grubb, T. C., Jr. 1989. Ptilochronology: feather growth bars as indicators of nutritional status. Auk 106:314-320.
Grubb, T. C., Jr. 2006. Ptilochronology. Feather time and the biology of birds. Oxford University Press, Oxford, UK.
Harriman, V. B., R. D. Dawson, L. E. Bortolotti, and R. G. Clark. 2017. Seasonal patterns in reproductive success of temperate-breeding birds: experimental tests of the date and quality hypotheses. Ecology and Evolution 7:2122-2132. https://doi.org/10.1002/ece3.2815
Harrison, X. A., J. D. Blount, R. Inger, D. R. Norris, and S. Bearhop. 2011. Carry-over effects as drivers of fitness differences in animals. Journal of Animal Ecology 80:4-18. https://doi.org/10.1111/j.1365-2656.2010.01740.x
Hirano, T. 2009. White Wagtail. Bird Research News 6:4-5.
Hothorn, T., and K. Hornik. 2021. Exact distributions for rank and permutation tests. R package version 0.8-32. https://cran.r-project.org/web/packages/exactRankTests/exactRankTests.pdf
Inger, R., X. A. Harrison, G. D. Ruxton, J. Newton, K. Colhoun, G. A. Gudmundsson, G. McElwaine, M. Pickford, D. Hodgson, and S. Bearhop. 2010. Carry-over effects reveal reproductive costs in a long-distance migrant. Journal of Animal Ecology 79:974-982. https://doi.org/10.1111/j.1365-2656.2010.01712.x
Japan Meteorological Agency. 2010. Weather data search 1981–2010 at Sakai. Japan Meteorological Agency, Otemachi, Japan. http://www.data.jma.go.jp/obd/stats/etrn/view/nml_amd_ym.php?prec_no=62&block_no=1062&year=&month=&day=&view=a2
Jenni, L., and R. Winkler. 1994. Moult and ageing of European passerines. Academic Press, London, UK.
Jenni, L., and R. Winkler. 2020. The biology of moult in birds. Helm, London, UK.
Kawabe, M. 2006. Factors of interspecific pair formation between Japanese Wagtail, Motacilla grandis and White Wagtail, M. alba lugens. Bulletin of the Higashi Taisetsu Museum of Natural History 28:23-38 (in Japanese with English summary).
Kubo, T. 2012. Introduction to statistical modelling for data analysis-generalized linear model, Hierarchical Bayesian model, and MCMC. Iwanami, Tokyo, Japan (in Japanese).
Kumagai, J., T. Inuyama, K. Usui, A. Saito, T. Hamaguchi, A. Mishima, and K. Yashi. 1983. On communal roost of White Wagtail Motacilla alba in Kanagawa prefecture. Strix 2:33-40 (in Japanese with English summary).
Mench, J. A., and R. A. Blatchford. 2013. Birds as laboratory animals. Pages 279-299 in K. Bayne and P. V. Turner, editors. Laboratory animal welfare. Elsevier, Amsterdam, Netherlands. https://doi.org/10.1016/B978-0-12-385103-1.00016-6
Møller, A. P., J. T. Nielsen, and J. Erritzoe. 2006. Losing the last feather: feather loss as an antipredator adaptation in birds. Behavioral Ecology 17:1046-1056. https://doi.org/10.1093/beheco/arl044
Nakagawa, S. 2004. A farewell to Bonferroni: the problems of low statistical power and publication bias. Behavioral Ecology 15:1044-1045. https://doi.org/10.1093/beheco/arh107
Newton, I. 2009. Moult and plumage. Ringing & Migration 24:220-226. https://doi.org/10.1080/03078698.2009.9674395
Ornithological Society of Japan. 2012. Check-list of Japanese birds. Seventh revised edition. Ornithological Society of Japan, Sanda, Japan.
Payevsky, V. A. 2016. Sex-biased survival and philopatry in birds: Do they interact? Biology Bulletin 43:804-818. https://doi.org/10.1134/S1062359016080136
R Core Team. 2018. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.r-project.org/
Randler, C. 2006. Is tail wagging in White Wagtails, Motacilla alba, an honest signal of vigilance? Animal Behaviour 71:1089-1093. https://doi.org/10.1016/j.anbehav.2005.07.026
Randler, C. 2016. Tail movements in birds—current evidence and new concepts. Ornithological Science 15:1-14. https://doi.org/10.2326/osj.15.1
Reudink, M. W., P. P. Marra, K. M. Langin, C. E. Studds, T. K. Kyser, and L. M. Ratcliffe. 2008. Molt-migration in the American Redstart (Setophaga ruticilla) revisited: explaining variation in feather δD signatures. Auk 125:742-746. https://doi.org/10.1525/auk.2008.2708.1
Revelle, W. 2021. psych: Procedures for psychological, psychometric, and personality research. R Package 2.1.3. https://cran.r-project.org/web/packages/psych/index.html
Riddle, O. 1908. The genesis of fault-bars in feathers and the cause of alternation of light and dark fundamental bars. Biological Bulletin 14:328-321. https://doi.org/10.2307/1535869
Romano, A., A. Costanzo, D. Rubolini, N. Saino, and A. P. Møller. 2017. Geographical and seasonal variation in the intensity of sexual selection in the Barn Swallow Hirundo rustica: a meta‐analysis. Biological Reviews 92:1582-1600. https://doi.org/10.1111/brv.12297
Senar, J. E., and J. Pascual. 1997. Keel and tarsus length may provide a good predictor of avian body size. Ardea 85:269-274.
Serrano, D., and R. Jovani. 2005. Adaptive fault bar distribution in a long-distance migratory, aerial forager passerine? Biological Journal of the Linnean Society 85:455-461. https://doi.org/10.1111/j.1095-8312.2005.00509.x
Stearns, S. C. 1992. The evolution of life histories. Oxford University Press, Oxford, UK.
Stewart, R. L. M., C. M. Francis, and C. Masse. 2002. Age-related differential timing of spring migration within sexes in passerines. Wilson Bulletin 114:264-271. https://doi.org/10.1676/0043-5643(2002)114[0264:ARDTOS]2.0.CO;2
Svensson, L. 1992. Identification guide to the European passerines. Svensson, Stockholm, Sweden.
Takagi, M. 2022. Sex and age-class differences in feather mite loads in White Wagtails Motacilla alba lugens suggest self-regulation of feather mites by birds. Journal of Avian Biology 2022(7):e02943. https://doi.org/10.1111/jav.02943
Tonra, C. M., K. L. D. Marini, P. P. Marra, R. R. Germain, R. L. Holberton, and M. W. Reudink. 2014. Color expression in experimentally regrown feathers of an overwintering migratory bird: implications for signaling and seasonal interactions. Ecology and Evolution 4:1222-1232. https://doi.org/10.1002/ece3.994
Verhulst, S., and J-Å. Nilsson. 2008. The timing of birds’ breeding seasons: a review of experiments that manipulated timing of breeding. Philosophical Transactions of the Royal Society B 363:399-410. https://doi.org/10.1098/rstb.2007.2146
Watanabe, M., and N. Maruyama. 1977. Wintering ecology of White Wagtail Motacilla alba lugens in the middle stream of Tama River. Journal of the Yamashina Institute for Ornithology 9:20-43. https://doi.org/10.3312/jyio1952.9.20
Yamashina, Y. 1934. A natural history of Japanese birds. Azusa shobo, Tokyo, Japan.
Yamashina Institute for Ornithology. 1996. Atlas of Japanese migratory birds from 1961 to 1995. Yamashina Institute for Ornithology, Abiko, Japan.
Yakovleva, G. A., T. A. Rimkevich, and G. A. Noskov. 1987. Comparative characteristics of post-embriotic development of post-juvenal molt of White Wagtails (Motacilla alba L.) from early and late broods. Vestnik Leningrad University 2:12-20.
Fig. 1
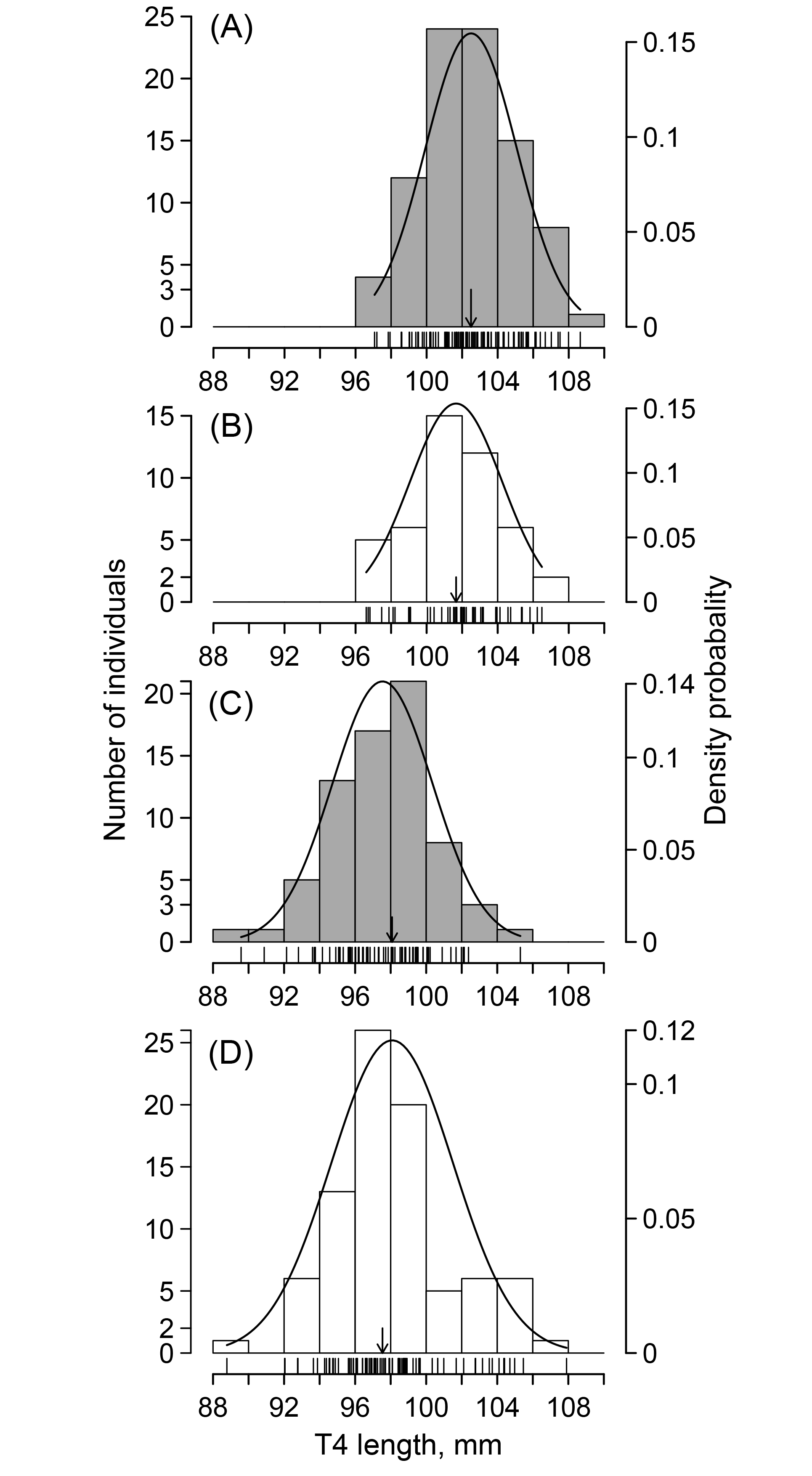
Fig. 1. Frequency distributions of the original T4 length (OLT4) and the normal probability density distributions of OLT4 in (A) adult males, (B) yearling males, (C) adult females, and (D) yearling females. Vertical lines on the x-axis show OLT4 for all individuals. The solid vertical downward arrows indicate the mean values category.

Fig. 2
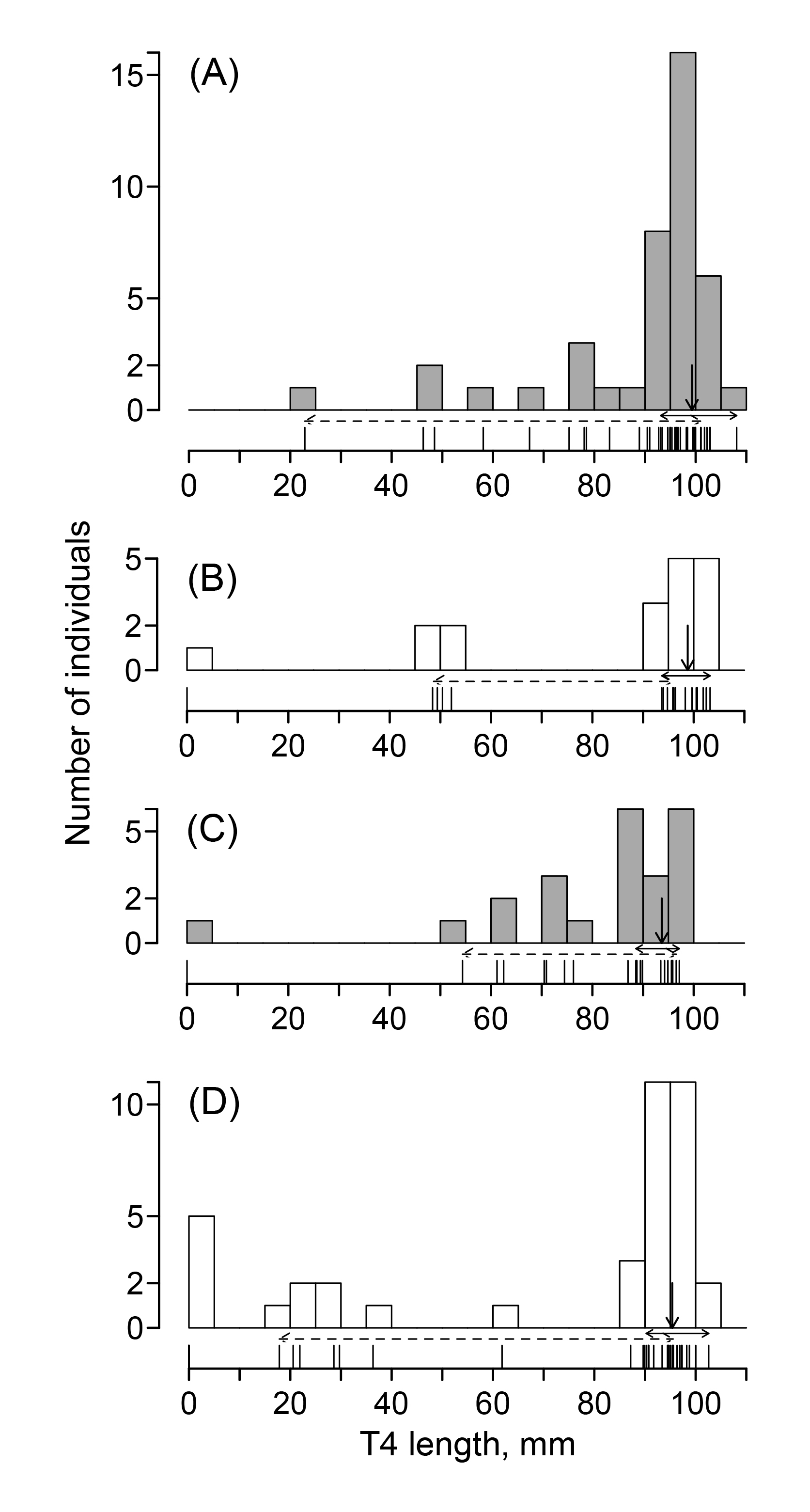
Fig. 2. Frequency distributions of the replacement T4 length (RLT4) in (A) adult males, (B) yearling males, (C) adult females, and (D) yearling females. Vertical lines on the x-axis show RLT4 for all individuals. The solid vertical downward arrows indicate the mean value of fully grown RLT4. The solid and dashed horizontal arrows indicate the ranges of the fully grown RLT4s and the growing RLT4, respectively.

Fig. 3
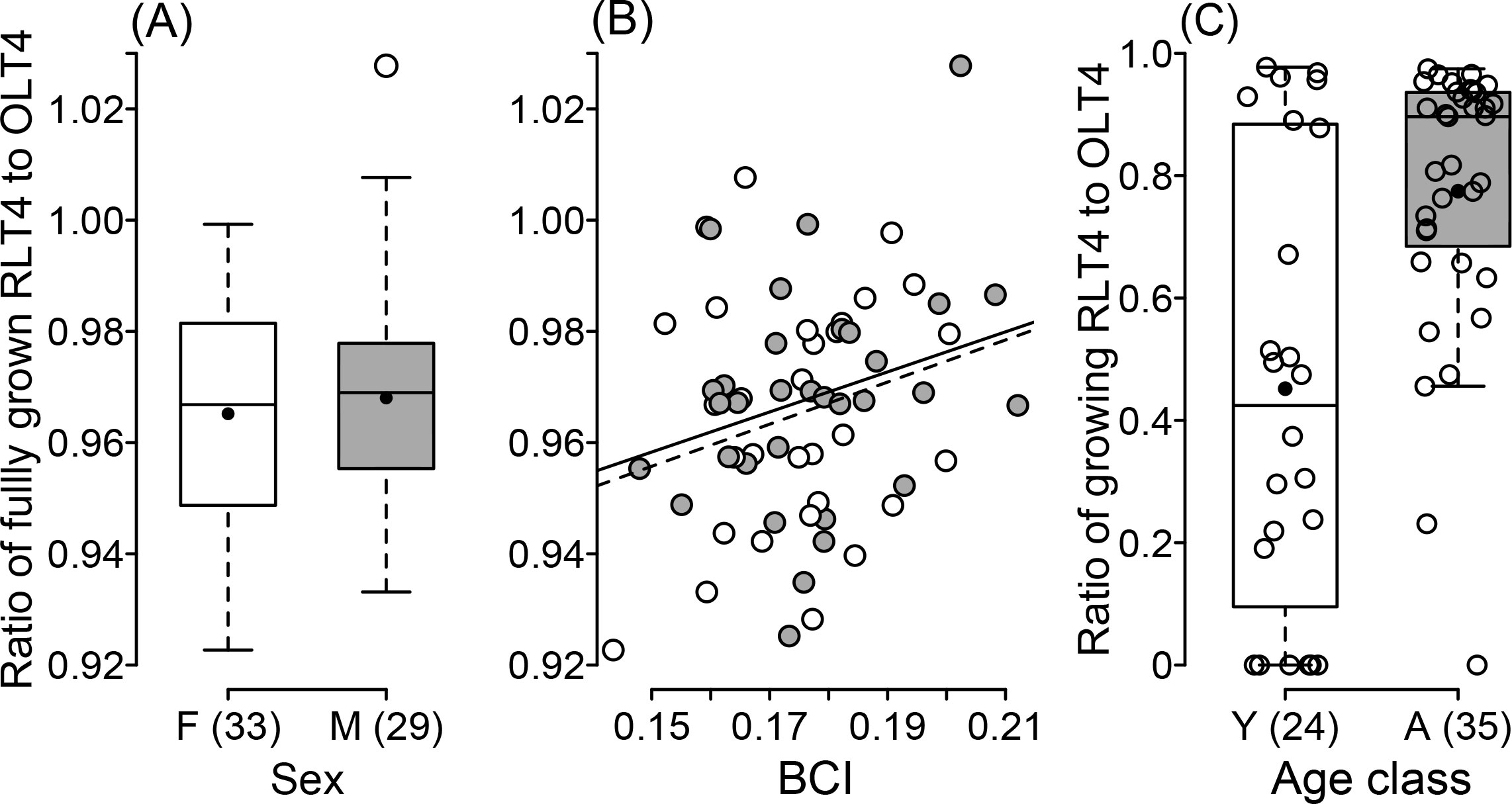
Fig. 3. (A) Comparison of the ratio of fully grown replaced length of T4 (RLT4) to original length of T4 (OLT4) in males and females. The lower and upper edges of the boxes represent the first and third quartiles, respectively, while the solid lines dividing each box are the medians, and the black dots represent means. The vertical whisker lines include the ranges of values. Parentheses on the x-axis show sample sizes. (B) Relationship between body condition index (BCI) and the ratio of fully grown RLT4 to OLT4. Dark grey dots indicate males; open dots indicate females. Linear regressions are for males (solid line y = 0.360x+0.904) and females (dotted line y = 0.379x+0.899), where y is the ratio of fully grown RLT4 to OLT4, and x is BIC. (C) Comparison of the ratio of growing RLT4 to OLT4 in relation to age class. Open dots represent individuals; the other symbols used are the same as in (A).

Table 1
Table 1. Number (percentages) of three statuses of T4 growth in relation to combination of age class and sex.
| Rectrix state (% individuals) | |||||
| Age class | Sex | Fully regrown | Growing | Ungrown | Total |
| Adult | Male | 18 (44) | 23 (56) | 0 | 41 |
| Female | 11 (48) | 11 (48) | 1 (4) | 23 | |
| Total | 29 (45) | 34 (53) | 1 (2) | 64 | |
| Yearling | Male | 11 (61) | 6 (33) | 1 (6) | 18 |
| Female | 22 (56) | 12 (31) | 5 (13) | 39 | |
| Total | 33 (58) | 18 (32) | 6 (11) | 57 | |
Table 2
Table 2. Regression coefficients of the ratio of the regrown T4 to the original T4 in the most parsimonious final models, in the results of generalized linear model with a Gaussian distribution and identity link function. Results for models that incorporated (A) body condition indexes (BCI) in the first period, (B) in the second period, and (C) the change index of body condition (BCCI) in the first and second periods. Numbers of adult males = 41, yearling males = 18, adult females = 23, and yearling females = 39.
| Variables | Estimate | SE | t value | P | |
| (A) | Intercept | 85.72 | 8.38 | 10.23 | < 0.0001 |
| Sex (male) | 4.33 | 0.86 | 5.04 | < 0.0001 | |
| (B) | Intercept | 82.43 | 8.73 | 9.44 | < 0.0001 |
| Sex (male) | 4.33 | 0.85 | 5.09 | < 0.0001 | |
| BCI | 6036.92 | 2842.27 | 2.12 | 0.038 | |
| (C) | Intercept | 94.09 | 7.01 | 13.43 | < 0.0001 |
| Sex (male) | 4.35 | 0.88 | 4.93 | < 0.0001 | |









